 Found 32 hits with Last Name = 'wang' and Initial = 're'
Found 32 hits with Last Name = 'wang' and Initial = 're' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protein O-GlcNAcase
(Homo sapiens (Human)) | BDBM50323697
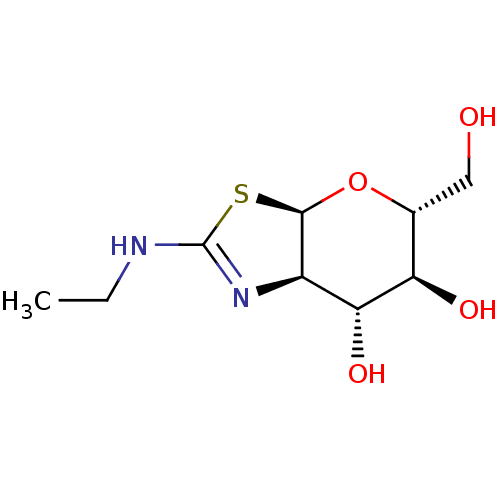
((3AR,5R,6S,7R,7AR)-2-(ETHYLAMINO)-5-(HYDROXYMETHYL...)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](CO)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O4S/c1-2-10-9-11-5-7(14)6(13)4(3-12)15-8(5)16-9/h4-8,12-14H,2-3H2,1H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human O-GlcNAcase using pNP-GlcNAc as substrate measured for 5 mins by UV-VIS spectrophotometer |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone acetyltransferase p300
(Homo sapiens (Human)) | BDBM50453820

(CHEMBL4208500)Show SMILES CC1=NN(C(=O)C1=Cc1ccc(o1)-c1cc(C)c(C)cc1[N+]([O-])=O)c1ccc(cc1)C(O)=O |t:1| Show InChI InChI=1S/C24H19N3O6/c1-13-10-20(21(27(31)32)11-14(13)2)22-9-8-18(33-22)12-19-15(3)25-26(23(19)28)17-6-4-16(5-7-17)24(29)30/h4-12H,1-3H3,(H,29,30)/b19-12- | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Competitive inhibition of VMA intein chitin binding domain-fused p300 HAT domain M1652G mutant (unknown origin) expressed in Escherichia coli BL21(RI... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50138764
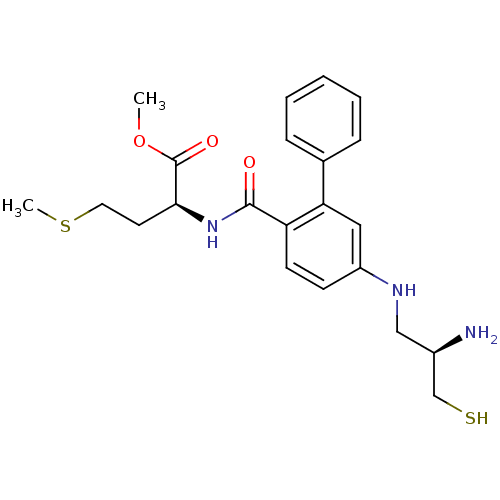
((S)-2-{[5-((R)-2-Amino-3-mercapto-propylamino)-bip...)Show SMILES COC(=O)[C@H](CCSC)NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1ccccc1 Show InChI InChI=1S/C22H29N3O3S2/c1-28-22(27)20(10-11-30-2)25-21(26)18-9-8-17(24-13-16(23)14-29)12-19(18)15-6-4-3-5-7-15/h3-9,12,16,20,24,29H,10-11,13-14,23H2,1-2H3,(H,25,26)/t16-,20+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase isolated from human Burkitt's lymphoma cells assessed as decrease in transfer of [3H]farnesyl from [3H]farnesyl PPi to H-Ras-CVLS |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50067584

((S)-2-{[5-((R)-2-Amino-3-mercapto-propylamino)-bip...)Show SMILES CSCC[C@H](NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H27N3O3S2/c1-29-10-9-19(21(26)27)24-20(25)17-8-7-16(23-12-15(22)13-28)11-18(17)14-5-3-2-4-6-14/h2-8,11,15,19,23,28H,9-10,12-13,22H2,1H3,(H,24,25)(H,26,27)/t15-,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase isolated from human Burkitt's lymphoma cells assessed as decrease in transfer of [3H]farnesyl from [3H]farnesyl PPi to H-Ras-CVLS |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50453819
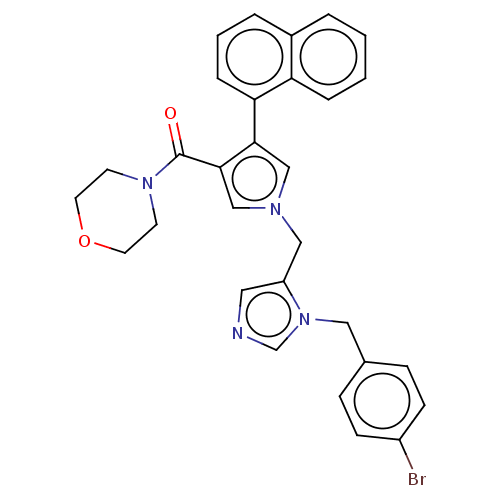
(CHEMBL4210419)Show SMILES Brc1ccc(Cn2cncc2Cn2cc(C(=O)N3CCOCC3)c(c2)-c2cccc3ccccc23)cc1 Show InChI InChI=1S/C30H27BrN4O2/c31-24-10-8-22(9-11-24)17-35-21-32-16-25(35)18-33-19-28(27-7-3-5-23-4-1-2-6-26(23)27)29(20-33)30(36)34-12-14-37-15-13-34/h1-11,16,19-21H,12-15,17-18H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase (unknown origin) using H-ras as substrate |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50453817
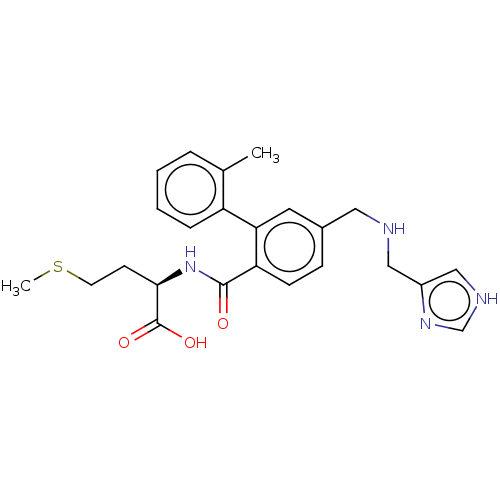
(CHEMBL4218727)Show SMILES CSCC[C@@H](NC(=O)c1ccc(CNCc2c[nH]cn2)cc1-c1ccccc1C)C(O)=O |r| Show InChI InChI=1S/C24H28N4O3S/c1-16-5-3-4-6-19(16)21-11-17(12-25-13-18-14-26-15-27-18)7-8-20(21)23(29)28-22(24(30)31)9-10-32-2/h3-8,11,14-15,22,25H,9-10,12-13H2,1-2H3,(H,26,27)(H,28,29)(H,30,31)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase isolated from human Burkitt's lymphoma cells assessed as decrease in transfer of [3H]farnesyl from [3H]farnesyl PPi to H-Ras-CVLS |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50092365
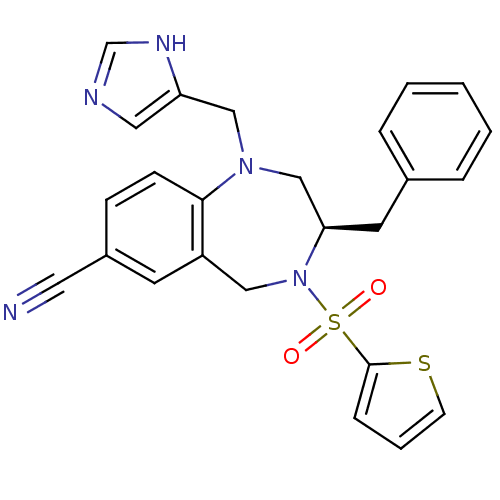
((R)-1-((1H-imidazol-5-yl)methyl)-3-benzyl-4-(thiop...)Show SMILES O=S(=O)(N1Cc2cc(ccc2N(Cc2cnc[nH]2)C[C@H]1Cc1ccccc1)C#N)c1cccs1 Show InChI InChI=1S/C25H23N5O2S2/c26-13-20-8-9-24-21(11-20)15-30(34(31,32)25-7-4-10-33-25)23(12-19-5-2-1-3-6-19)17-29(24)16-22-14-27-18-28-22/h1-11,14,18,23H,12,15-17H2,(H,27,28)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant FTase |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Rattus norvegicus-Rattus norvegicus (rat)) | BDBM14459
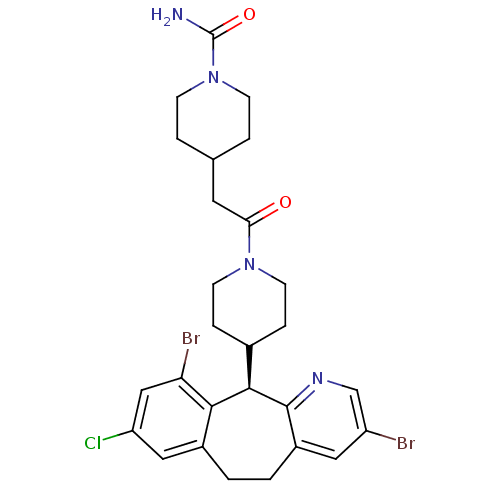
((+)-4-[2-[4-(8-Chloro-3,10-dibromo-6,11-dihydro-5H...)Show SMILES NC(=O)N1CCC(CC(=O)N2CCC(CC2)[C@H]2c3ncc(Br)cc3CCc3cc(Cl)cc(Br)c23)CC1 |r| Show InChI InChI=1S/C27H31Br2ClN4O2/c28-20-12-19-2-1-18-13-21(30)14-22(29)24(18)25(26(19)32-15-20)17-5-9-33(10-6-17)23(35)11-16-3-7-34(8-4-16)27(31)36/h12-17,25H,1-11H2,(H2,31,36)/t25-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of rat brain FTase assessed as decrease in transfer of [3H]farnesyl from [3H]farnesyl PPi to H-Ras-CVLS after 30 mins by liquid scintillat... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
NEDD8
(Homo sapiens) | BDBM50285607
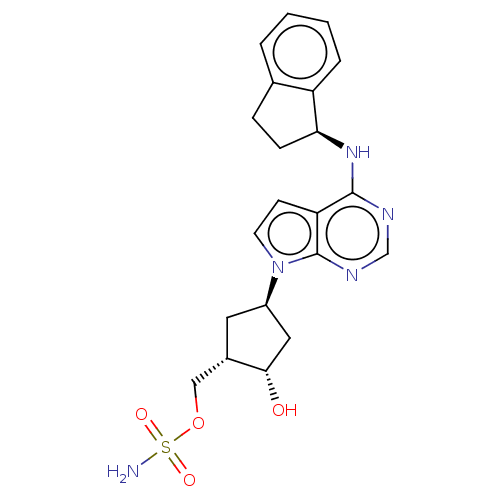
(MLN-4924 | MLN-4924003 | Pevonedistat)Show SMILES [H][C@]1(O)C[C@@]([H])(C[C@@]1([H])COS(N)(=O)=O)n1ccc2c(N[C@@]3([H])CCc4ccccc34)ncnc12 |r| Show InChI InChI=1S/C21H25N5O4S/c22-31(28,29)30-11-14-9-15(10-19(14)27)26-8-7-17-20(23-12-24-21(17)26)25-18-6-5-13-3-1-2-4-16(13)18/h1-4,7-8,12,14-15,18-19,27H,5-6,9-11H2,(H2,22,28,29)(H,23,24,25)/t14-,15+,18-,19-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of NEDD8 (unknown origin) assessed as decrease in formation of E2-UBL thioester reaction product |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta/Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076128
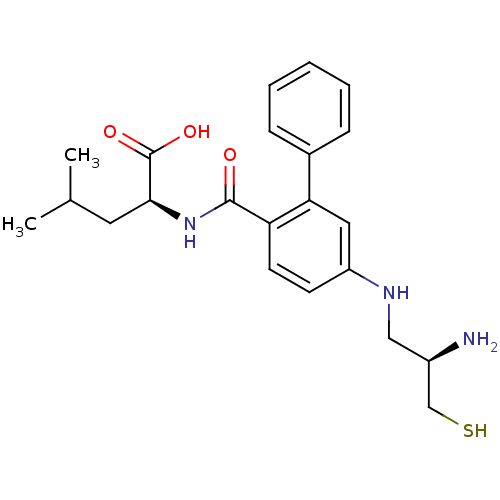
((S)-2-{[5-((R)-2-Amino-3-mercapto-propylamino)-bip...)Show SMILES CC(C)C[C@H](NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N3O3S/c1-14(2)10-20(22(27)28)25-21(26)18-9-8-17(24-12-16(23)13-29)11-19(18)15-6-4-3-5-7-15/h3-9,11,14,16,20,24,29H,10,12-13,23H2,1-2H3,(H,25,26)(H,27,28)/t16-,20+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of GGTase1 isolated from human Burkitt's lymphoma cells assessed as decrease in transfer of [3H]H]geranylgeranyl from [3H]geranylgeranyl P... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Receptor-interacting serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50322535
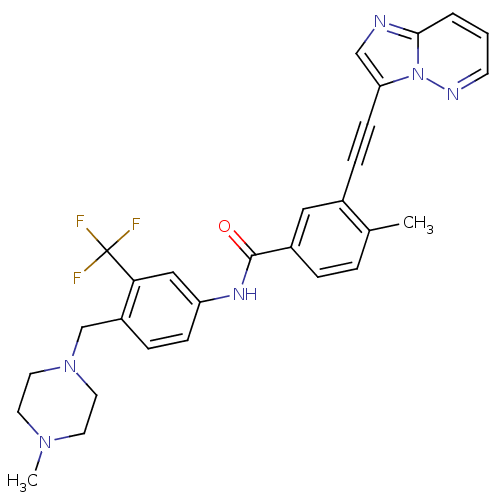
(3-(imidazo[1,2-b]pyridazin-3-ylethynyl)-4-methyl-N...)Show SMILES CN1CCN(Cc2ccc(NC(=O)c3ccc(C)c(c3)C#Cc3cnc4cccnn34)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C29H27F3N6O/c1-20-5-6-22(16-21(20)8-10-25-18-33-27-4-3-11-34-38(25)27)28(39)35-24-9-7-23(26(17-24)29(30,31)32)19-37-14-12-36(2)13-15-37/h3-7,9,11,16-18H,12-15,19H2,1-2H3,(H,35,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of human RIPK2 expressed in Sf9 cells after 1 hr by ADP-Glo assay |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50370385
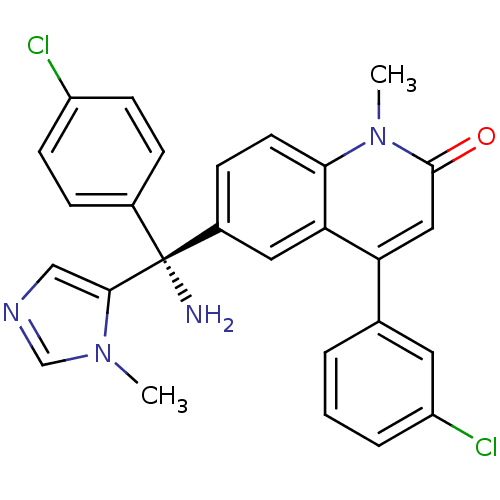
((R)-TIPIFARNIB | R-11577 | R-115777 | Tipifarnib |...)Show SMILES Cn1cncc1[C@@](N)(c1ccc(Cl)cc1)c1ccc2n(C)c(=O)cc(-c3cccc(Cl)c3)c2c1 |r| Show InChI InChI=1S/C27H22Cl2N4O/c1-32-16-31-15-25(32)27(30,18-6-9-20(28)10-7-18)19-8-11-24-23(13-19)22(14-26(34)33(24)2)17-4-3-5-21(29)12-17/h3-16H,30H2,1-2H3/t27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase isolated from Kirsten virus-transformed human osteosarcoma cells using K-rasB peptide as substrate in presence of [3H]farnesyl PP... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glycylpeptide N-tetradecanoyltransferase
(Aspergillus fumigatus) | BDBM50033418
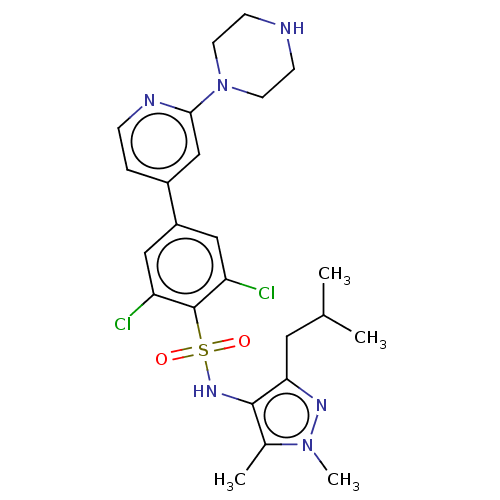
(CHEMBL3357685)Show SMILES CC(C)Cc1nn(C)c(C)c1NS(=O)(=O)c1c(Cl)cc(cc1Cl)-c1ccnc(c1)N1CCNCC1 Show InChI InChI=1S/C24H30Cl2N6O2S/c1-15(2)11-21-23(16(3)31(4)29-21)30-35(33,34)24-19(25)12-18(13-20(24)26)17-5-6-28-22(14-17)32-9-7-27-8-10-32/h5-6,12-15,27,30H,7-11H2,1-4H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aspergillus fumigatus NMT (Gln86 to Leu492 residues) expressed in Escherichia coli BL21(DE3) pLysS cells using [3H]-myristo... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50453816
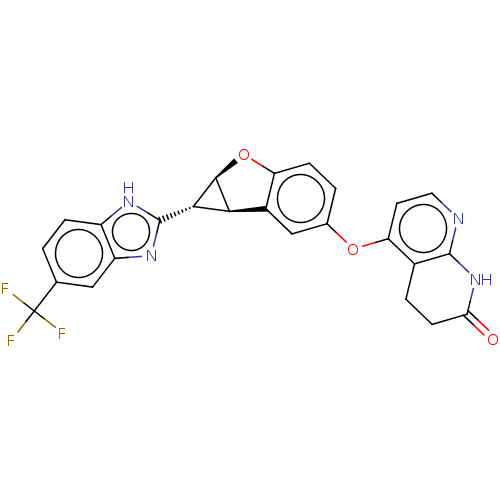
(BGB-283 | Bgb 283 | Lifirafenib)Show SMILES [H][C@]12Oc3ccc(Oc4ccnc5NC(=O)CCc45)cc3[C@@]1([H])[C@@H]2c1nc2cc(ccc2[nH]1)C(F)(F)F |r| Show InChI InChI=1S/C25H17F3N4O3/c26-25(27,28)11-1-4-15-16(9-11)31-24(30-15)21-20-14-10-12(2-5-17(14)35-22(20)21)34-18-7-8-29-23-13(18)3-6-19(33)32-23/h1-2,4-5,7-10,20-22H,3,6H2,(H,30,31)(H,29,32,33)/t20-,21-,22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin) using MEK1 K97R mutant as substrate preincubated for 60 to 120 mins followed by substrate addition measured after... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50453818

(CHEMBL4202636)Show SMILES COC(=O)[C@H](CCSC)NC(=O)CN1C[C@@H](N=C(c2ccccc2)c2ccccc12)N(C)C(=O)[C@H](N)CS |r,t:16| Show InChI InChI=1S/C27H35N5O4S2/c1-31(26(34)20(28)17-37)23-15-32(16-24(33)29-21(13-14-38-3)27(35)36-2)22-12-8-7-11-19(22)25(30-23)18-9-5-4-6-10-18/h4-12,20-21,23,37H,13-17,28H2,1-3H3,(H,29,33)/t20-,21+,23+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase (unknown origin) |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta/Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076141
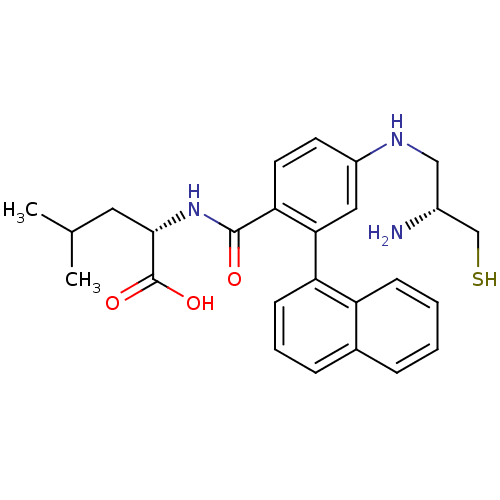
((S)-2-[4-((R)-2-Amino-3-mercapto-propylamino)-2-na...)Show SMILES CC(C)C[C@H](NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1cccc2ccccc12)C(O)=O Show InChI InChI=1S/C26H31N3O3S/c1-16(2)12-24(26(31)32)29-25(30)22-11-10-19(28-14-18(27)15-33)13-23(22)21-9-5-7-17-6-3-4-8-20(17)21/h3-11,13,16,18,24,28,33H,12,14-15,27H2,1-2H3,(H,29,30)(H,31,32)/t18-,24+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of GGTase1 isolated from human Burkitt's lymphoma cells assessed as decrease in transfer of [3H]H]geranylgeranyl from [3H]geranylgeranyl P... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076141

((S)-2-[4-((R)-2-Amino-3-mercapto-propylamino)-2-na...)Show SMILES CC(C)C[C@H](NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1cccc2ccccc12)C(O)=O Show InChI InChI=1S/C26H31N3O3S/c1-16(2)12-24(26(31)32)29-25(30)22-11-10-19(28-14-18(27)15-33)13-23(22)21-9-5-7-17-6-3-4-8-20(17)21/h3-11,13,16,18,24,28,33H,12,14-15,27H2,1-2H3,(H,29,30)(H,31,32)/t18-,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase isolated from human Burkitt's lymphoma cells assessed as decrease in transfer of [3H]farnesyl from [3H]farnesyl PPi to H-Ras-CVLS |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase RNF31/RanBP-type and C3HC4-type zinc finger-containing protein 1/Sharpin
(Homo sapiens) | BDBM50134315
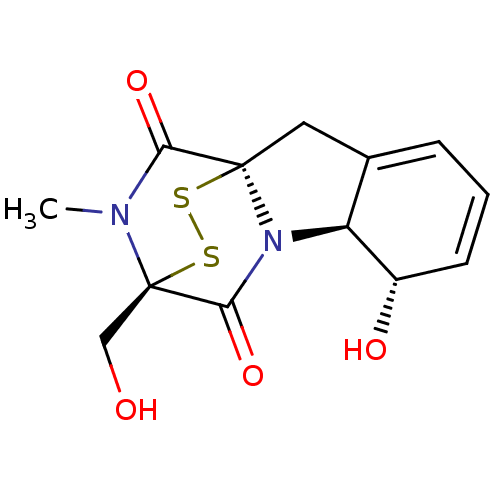
(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 510 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of petit-LUBAC (unknown origin)-mediated ubiquitylation expressed in Escherichia coli BL21 after 2 hrs in presence of E1, UbcH5c, E3 and u... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 10/8/9
(Homo sapiens (Human)) | BDBM29315

(2-amino-1,3,4-thiadiazole, 9)Show InChI InChI=1S/C5H3N5O2S3/c6-3-8-9-5(14-3)15-4-7-1-2(13-4)10(11)12/h1H,(H2,6,8) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of JNK (unknown origin) |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Ubiquitin-like modifier-activating enzyme ATG7
(Homo sapiens (Human)) | BDBM50285607

(MLN-4924 | MLN-4924003 | Pevonedistat)Show SMILES [H][C@]1(O)C[C@@]([H])(C[C@@]1([H])COS(N)(=O)=O)n1ccc2c(N[C@@]3([H])CCc4ccccc34)ncnc12 |r| Show InChI InChI=1S/C21H25N5O4S/c22-31(28,29)30-11-14-9-15(10-19(14)27)26-8-7-17-20(23-12-24-21(17)26)25-18-6-5-13-3-1-2-4-16(13)18/h1-4,7-8,12,14-15,18-19,27H,5-6,9-11H2,(H2,22,28,29)(H,23,24,25)/t14-,15+,18-,19-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of ATG7 (unknown origin) assessed as decrease in formation of E2-UBL thioester reaction product |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
SUMO-activating enzyme subunit 1/2
(Homo sapiens (Human)) | BDBM50285607

(MLN-4924 | MLN-4924003 | Pevonedistat)Show SMILES [H][C@]1(O)C[C@@]([H])(C[C@@]1([H])COS(N)(=O)=O)n1ccc2c(N[C@@]3([H])CCc4ccccc34)ncnc12 |r| Show InChI InChI=1S/C21H25N5O4S/c22-31(28,29)30-11-14-9-15(10-19(14)27)26-8-7-17-20(23-12-24-21(17)26)25-18-6-5-13-3-1-2-4-16(13)18/h1-4,7-8,12,14-15,18-19,27H,5-6,9-11H2,(H2,22,28,29)(H,23,24,25)/t14-,15+,18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of SAE (unknown origin) assessed as decrease in formation of E2-UBL thioester reaction product |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta
(Rattus norvegicus) | BDBM14459

((+)-4-[2-[4-(8-Chloro-3,10-dibromo-6,11-dihydro-5H...)Show SMILES NC(=O)N1CCC(CC(=O)N2CCC(CC2)[C@H]2c3ncc(Br)cc3CCc3cc(Cl)cc(Br)c23)CC1 |r| Show InChI InChI=1S/C27H31Br2ClN4O2/c28-20-12-19-2-1-18-13-21(30)14-22(29)24(18)25(26(19)32-15-20)17-5-9-33(10-6-17)23(35)11-16-3-7-34(8-4-16)27(31)36/h12-17,25H,1-11H2,(H2,31,36)/t25-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of rat brain GGTase1 assessed as decrease in transfer of [3H]H]geranylgeranyl from [3H]geranylgeranyl PPi to H-Ras-CVLL after 30 mins by l... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta/Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50370385

((R)-TIPIFARNIB | R-11577 | R-115777 | Tipifarnib |...)Show SMILES Cn1cncc1[C@@](N)(c1ccc(Cl)cc1)c1ccc2n(C)c(=O)cc(-c3cccc(Cl)c3)c2c1 |r| Show InChI InChI=1S/C27H22Cl2N4O/c1-32-16-31-15-25(32)27(30,18-6-9-20(28)10-7-18)19-8-11-24-23(13-19)22(14-26(34)33(24)2)17-4-3-5-21(29)12-17/h3-16H,30H2,1-2H3/t27-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of GGTase1 isolated from Kirsten virus-transformed human osteosarcoma cells using biotin-YRASNRSCAIL peptide as substrate after 120 mins i... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ubiquitin-like modifier-activating enzyme 6
(Homo sapiens (Human)) | BDBM50285607

(MLN-4924 | MLN-4924003 | Pevonedistat)Show SMILES [H][C@]1(O)C[C@@]([H])(C[C@@]1([H])COS(N)(=O)=O)n1ccc2c(N[C@@]3([H])CCc4ccccc34)ncnc12 |r| Show InChI InChI=1S/C21H25N5O4S/c22-31(28,29)30-11-14-9-15(10-19(14)27)26-8-7-17-20(23-12-24-21(17)26)25-18-6-5-13-3-1-2-4-16(13)18/h1-4,7-8,12,14-15,18-19,27H,5-6,9-11H2,(H2,22,28,29)(H,23,24,25)/t14-,15+,18-,19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of UBA6 (unknown origin) assessed as decrease in formation of E2-UBL thioester reaction product |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta/Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50453818

(CHEMBL4202636)Show SMILES COC(=O)[C@H](CCSC)NC(=O)CN1C[C@@H](N=C(c2ccccc2)c2ccccc12)N(C)C(=O)[C@H](N)CS |r,t:16| Show InChI InChI=1S/C27H35N5O4S2/c1-31(26(34)20(28)17-37)23-15-32(16-24(33)29-21(13-14-38-3)27(35)36-2)22-12-8-7-11-19(22)25(30-23)18-9-5-4-6-10-18/h4-12,20-21,23,37H,13-17,28H2,1-3H3,(H,29,33)/t20-,21+,23+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of GGTase1 (unknown origin) |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Histone acetyltransferase p300
(Homo sapiens (Human)) | BDBM50151658
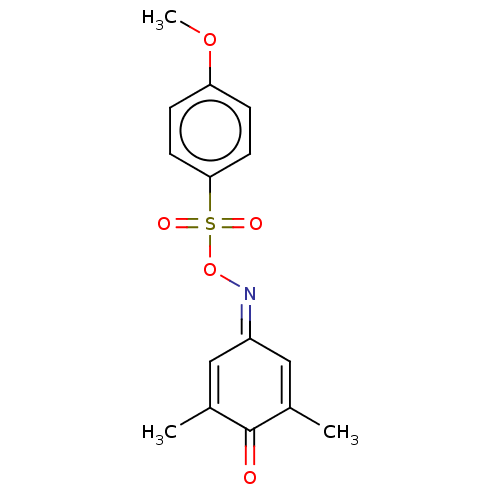
(CHEMBL3775671)Show SMILES [#6]-[#8]-c1ccc(cc1)S(=O)(=O)[#8]\[#7]=[#6]-1/[#6]=[#6](-[#6])-[#6](=O)-[#6](-[#6])=[#6]-1 |c:21,t:15| Show InChI InChI=1S/C15H15NO5S/c1-10-8-12(9-11(2)15(10)17)16-21-22(18,19)14-6-4-13(20-3)5-7-14/h4-9H,1-3H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of p300 catalytic domain (unknown origin) using histone H3 as substrate preincubated for 15 mins followed by substrate addition measured a... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta/Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076142
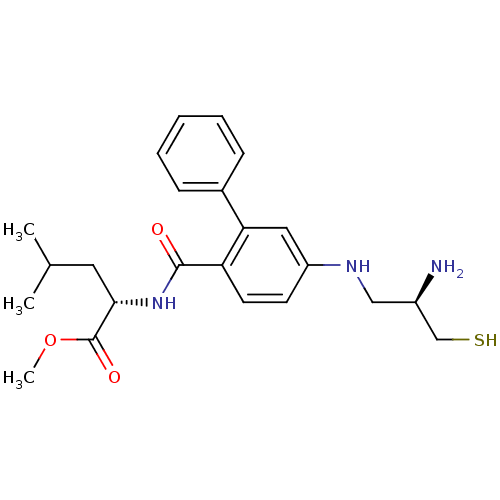
((S)-2-{[5-((R)-2-Amino-3-mercapto-propylamino)-bip...)Show SMILES COC(=O)[C@H](CC(C)C)NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1ccccc1 Show InChI InChI=1S/C23H31N3O3S/c1-15(2)11-21(23(28)29-3)26-22(27)19-10-9-18(25-13-17(24)14-30)12-20(19)16-7-5-4-6-8-16/h4-10,12,15,17,21,25,30H,11,13-14,24H2,1-3H3,(H,26,27)/t17-,21+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of GGTase1 (unknown origin) expressed in mouse NIH-3T3 cells transfected with k-Ras4B assessed as decrease in geranylgeranylation of k-Ras... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Calcium/calmodulin-dependent protein kinase type II subunit beta
(Homo sapiens (Human)) | BDBM7460

(2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...)Show InChI InChI=1S/C15H10O7/c16-7-4-10(19)12-11(5-7)22-15(14(21)13(12)20)6-1-2-8(17)9(18)3-6/h1-5,16-19,21H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University
Curated by ChEMBL
| Assay Description
Inhibition of CAMK2 using autocamtide-2 as substrate after 30 mins by PKLight assay |
Bioorg Med Chem 19: 4710-20 (2011)
Article DOI: 10.1016/j.bmc.2011.07.005
BindingDB Entry DOI: 10.7270/Q2959JM7 |
More data for this
Ligand-Target Pair | |
Histone acetyltransferase p300
(Homo sapiens (Human)) | BDBM50366305
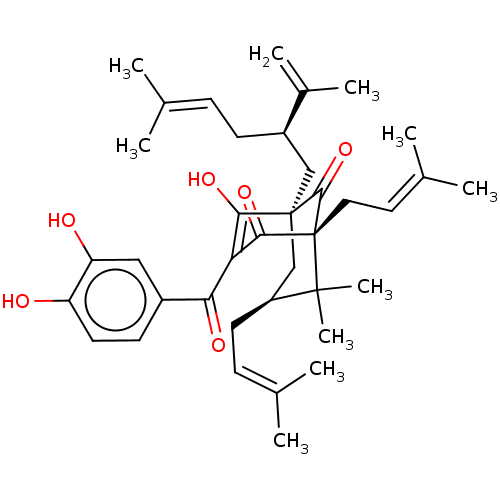
(CHEBI:70328 | CHEMBL445599)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-[#6@H](-[#6][C@]12[#6]-[#6@H](-[#6]\[#6]=[#6](\[#6])-[#6])C([#6])([#6])[C@]([#6]\[#6]=[#6](\[#6])-[#6])([#6](=O)-[#6](-[#6](=O)-c3ccc(-[#8])c(-[#8])c3)=[#6]1-[#8])[#6]2=O)-[#6](-[#6])=[#6] |r,c:37,TLB:40:39:26.37.24:15.8.9| Show InChI InChI=1S/C38H50O6/c1-22(2)11-13-27(25(7)8)20-37-21-28(15-12-23(3)4)36(9,10)38(35(37)44,18-17-24(5)6)34(43)31(33(37)42)32(41)26-14-16-29(39)30(40)19-26/h11-12,14,16-17,19,27-28,39-40,42H,7,13,15,18,20-21H2,1-6,8-10H3/t27-,28+,37+,38-/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of full length His6-tagged recombinant p300 (unknown origin) expressed in baculovirus infected Sf21 cells using human histone as substrate... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Calcium/calmodulin-dependent protein kinase type II subunit beta
(Homo sapiens (Human)) | BDBM50388936
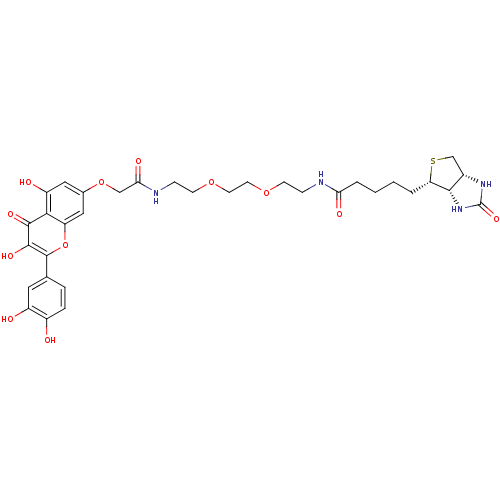
(CHEMBL2063416)Show SMILES Oc1ccc(cc1O)-c1oc2cc(OCC(=O)NCCOCCOCCNC(=O)CCCC[C@@H]3SC[C@@H]4NC(=O)N[C@H]34)cc(O)c2c(=O)c1O |r| Show InChI InChI=1S/C33H40N4O12S/c38-21-6-5-18(13-22(21)39)32-31(44)30(43)28-23(40)14-19(15-24(28)49-32)48-16-27(42)35-8-10-47-12-11-46-9-7-34-26(41)4-2-1-3-25-29-20(17-50-25)36-33(45)37-29/h5-6,13-15,20,25,29,38-40,44H,1-4,7-12,16-17H2,(H,34,41)(H,35,42)(H2,36,37,45)/t20-,25-,29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.59E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Washington University
Curated by ChEMBL
| Assay Description
Inhibition of CAMK2 using autocamtide-2 as substrate after 30 mins by PKLight assay |
Bioorg Med Chem 19: 4710-20 (2011)
Article DOI: 10.1016/j.bmc.2011.07.005
BindingDB Entry DOI: 10.7270/Q2959JM7 |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076142

((S)-2-{[5-((R)-2-Amino-3-mercapto-propylamino)-bip...)Show SMILES COC(=O)[C@H](CC(C)C)NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1ccccc1 Show InChI InChI=1S/C23H31N3O3S/c1-15(2)11-21(23(28)29-3)26-22(27)19-10-9-18(25-13-17(24)14-30)12-20(19)16-7-5-4-6-8-16/h4-10,12,15,17,21,25,30H,11,13-14,24H2,1-3H3,(H,26,27)/t17-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of FTase (unknown origin) expressed in mouse NIH-3T3 cells cells transfected with human H-Ras assessed as decrease in farnesylation of H-R... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Protein O-GlcNAcase
(Rattus norvegicus) | BDBM50323697

((3AR,5R,6S,7R,7AR)-2-(ETHYLAMINO)-5-(HYDROXYMETHYL...)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](CO)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O4S/c1-2-10-9-11-5-7(14)6(13)4(3-12)15-8(5)16-9/h4-8,12-14H,2-3H2,1H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of O-GlcNAcase in rat PC12 cells assessed as induction of OGlcNAcylation after 24 hrs by Western blot method |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data