 Found 443 hits with Last Name = 'wilson' and Initial = 'p'
Found 443 hits with Last Name = 'wilson' and Initial = 'p' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50094929
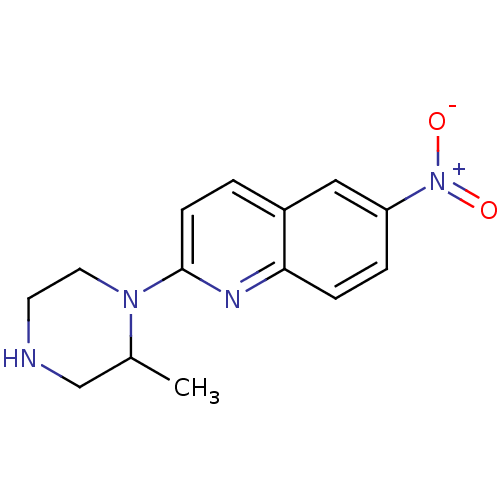
(2-(2-Methyl-piperazin-1-yl)-6-nitro-quinoline | CH...)Show InChI InChI=1S/C14H16N4O2/c1-10-9-15-6-7-17(10)14-5-2-11-8-12(18(19)20)3-4-13(11)16-14/h2-5,8,10,15H,6-7,9H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Washington University
Curated by ChEMBL
| Assay Description
In vitro radioligand [3H]-paroxetine from rat cortical Serotonin transporter |
Bioorg Med Chem Lett 10: 2643-6 (2000)
BindingDB Entry DOI: 10.7270/Q2W37VJW |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50063266
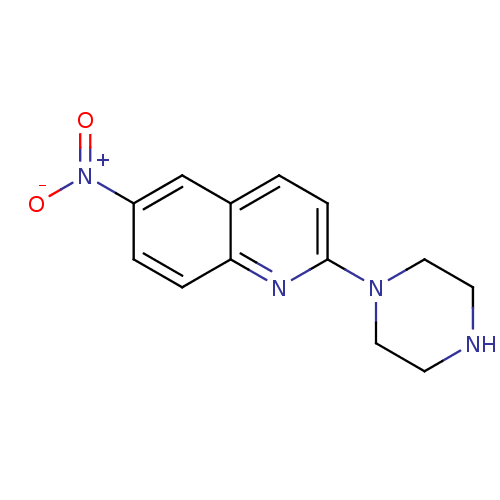
(6-Nitro-2-piperazin-1-yl-quinoline | 6-nitroquipaz...)Show InChI InChI=1S/C13H14N4O2/c18-17(19)11-2-3-12-10(9-11)1-4-13(15-12)16-7-5-14-6-8-16/h1-4,9,14H,5-8H2 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Washington University
Curated by ChEMBL
| Assay Description
In vitro radioligand [3H]-paroxetine from rat cortical Serotonin transporter |
Bioorg Med Chem Lett 10: 2643-6 (2000)
BindingDB Entry DOI: 10.7270/Q2W37VJW |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50063266

(6-Nitro-2-piperazin-1-yl-quinoline | 6-nitroquipaz...)Show InChI InChI=1S/C13H14N4O2/c18-17(19)11-2-3-12-10(9-11)1-4-13(15-12)16-7-5-14-6-8-16/h1-4,9,14H,5-8H2 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Washington University
Curated by ChEMBL
| Assay Description
In vitro radioligand [3H]-paroxetine from rat cortical Serotonin transporter |
Bioorg Med Chem Lett 10: 2643-6 (2000)
BindingDB Entry DOI: 10.7270/Q2W37VJW |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50094930
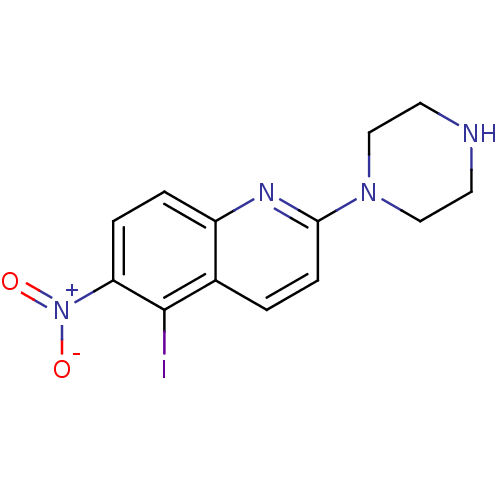
(5-Iodo-6-nitro-2-piperazin-1-yl-quinoline | CHEMBL...)Show InChI InChI=1S/C13H13IN4O2/c14-13-9-1-4-12(17-7-5-15-6-8-17)16-10(9)2-3-11(13)18(19)20/h1-4,15H,5-8H2 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Washington University
Curated by ChEMBL
| Assay Description
In vitro radioligand [3H]-paroxetine from rat cortical Serotonin transporter |
Bioorg Med Chem Lett 10: 2643-6 (2000)
BindingDB Entry DOI: 10.7270/Q2W37VJW |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219599
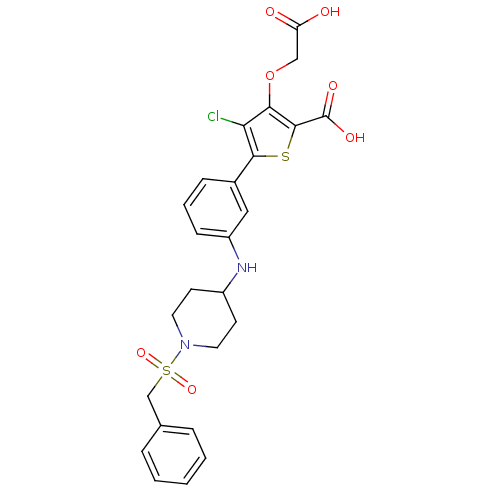
(5-(3-{[1-(benzylsulfonyl)piperidin-4-yl]amino}phen...)Show SMILES OC(=O)COc1c(Cl)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2)c1 Show InChI InChI=1S/C25H25ClN2O7S2/c26-21-22(35-14-20(29)30)24(25(31)32)36-23(21)17-7-4-8-19(13-17)27-18-9-11-28(12-10-18)37(33,34)15-16-5-2-1-3-6-16/h1-8,13,18,27H,9-12,14-15H2,(H,29,30)(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219588
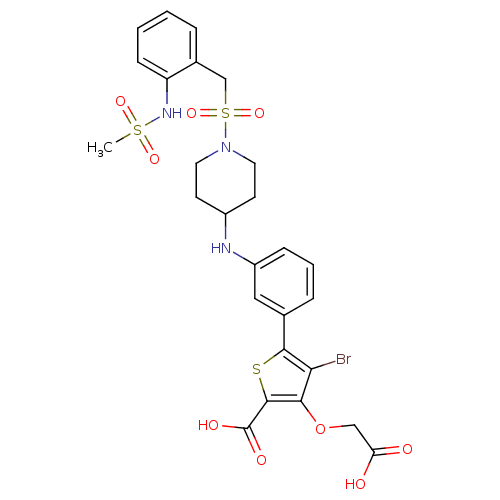
(4-bromo-3-(carboxymethoxy)-5-(3-{[1-({2-[(methylsu...)Show SMILES CS(=O)(=O)Nc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C26H28BrN3O9S3/c1-41(35,36)29-20-8-3-2-5-17(20)15-42(37,38)30-11-9-18(10-12-30)28-19-7-4-6-16(13-19)24-22(27)23(39-14-21(31)32)25(40-24)26(33)34/h2-8,13,18,28-29H,9-12,14-15H2,1H3,(H,31,32)(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219588

(4-bromo-3-(carboxymethoxy)-5-(3-{[1-({2-[(methylsu...)Show SMILES CS(=O)(=O)Nc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C26H28BrN3O9S3/c1-41(35,36)29-20-8-3-2-5-17(20)15-42(37,38)30-11-9-18(10-12-30)28-19-7-4-6-16(13-19)24-22(27)23(39-14-21(31)32)25(40-24)26(33)34/h2-8,13,18,28-29H,9-12,14-15H2,1H3,(H,31,32)(H,33,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219584
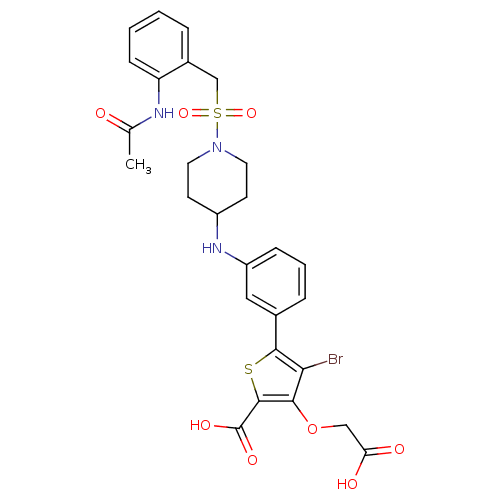
(5-{3-[(1-{[2-(acetylamino)benzyl]sulfonyl}piperidi...)Show SMILES CC(=O)Nc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C27H28BrN3O8S2/c1-16(32)29-21-8-3-2-5-18(21)15-41(37,38)31-11-9-19(10-12-31)30-20-7-4-6-17(13-20)25-23(28)24(39-14-22(33)34)26(40-25)27(35)36/h2-8,13,19,30H,9-12,14-15H2,1H3,(H,29,32)(H,33,34)(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219577
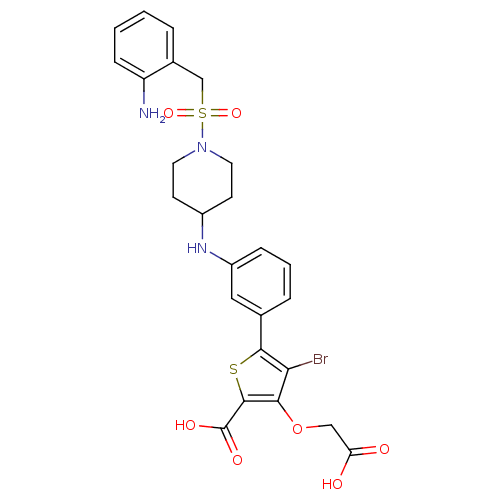
(5-[3-({1-[(2-aminobenzyl)sulfonyl]piperidin-4-yl}a...)Show SMILES Nc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C25H26BrN3O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)15-5-3-6-18(12-15)28-17-8-10-29(11-9-17)38(34,35)14-16-4-1-2-7-19(16)27/h1-7,12,17,28H,8-11,13-14,27H2,(H,30,31)(H,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219584

(5-{3-[(1-{[2-(acetylamino)benzyl]sulfonyl}piperidi...)Show SMILES CC(=O)Nc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C27H28BrN3O8S2/c1-16(32)29-21-8-3-2-5-18(21)15-41(37,38)31-11-9-19(10-12-31)30-20-7-4-6-17(13-20)25-23(28)24(39-14-22(33)34)26(40-25)27(35)36/h2-8,13,19,30H,9-12,14-15H2,1H3,(H,29,32)(H,33,34)(H,35,36) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219577

(5-[3-({1-[(2-aminobenzyl)sulfonyl]piperidin-4-yl}a...)Show SMILES Nc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C25H26BrN3O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)15-5-3-6-18(12-15)28-17-8-10-29(11-9-17)38(34,35)14-16-4-1-2-7-19(16)27/h1-7,12,17,28H,8-11,13-14,27H2,(H,30,31)(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219586
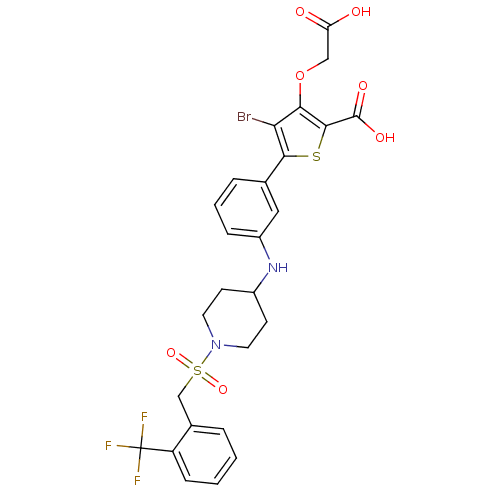
(4-bromo-3-(carboxymethoxy)-5-{3-[(1-{[2-(trifluoro...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2C(F)(F)F)c1 Show InChI InChI=1S/C26H24BrF3N2O7S2/c27-21-22(39-13-20(33)34)24(25(35)36)40-23(21)15-5-3-6-18(12-15)31-17-8-10-32(11-9-17)41(37,38)14-16-4-1-2-7-19(16)26(28,29)30/h1-7,12,17,31H,8-11,13-14H2,(H,33,34)(H,35,36) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219570
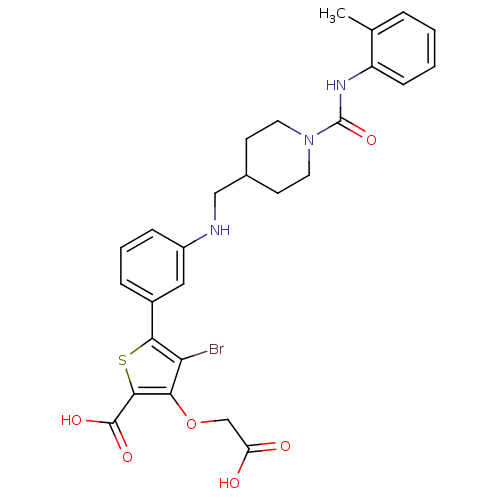
(4-bromo-3-carboxymethoxy-5-{3-[(1-o-tolylcarbamoyl...)Show SMILES Cc1ccccc1NC(=O)N1CCC(CNc2cccc(c2)-c2sc(C(O)=O)c(OCC(O)=O)c2Br)CC1 Show InChI InChI=1S/C27H28BrN3O6S/c1-16-5-2-3-8-20(16)30-27(36)31-11-9-17(10-12-31)14-29-19-7-4-6-18(13-19)24-22(28)23(37-15-21(32)33)25(38-24)26(34)35/h2-8,13,17,29H,9-12,14-15H2,1H3,(H,30,36)(H,32,33)(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219569
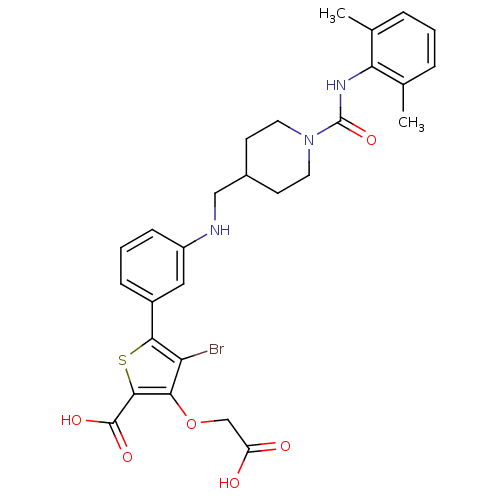
(4-bromo-3-carboxymethoxy-5-(3-{[1-(2,6-dimethylphe...)Show SMILES Cc1cccc(C)c1NC(=O)N1CCC(CNc2cccc(c2)-c2sc(C(O)=O)c(OCC(O)=O)c2Br)CC1 Show InChI InChI=1S/C28H30BrN3O6S/c1-16-5-3-6-17(2)23(16)31-28(37)32-11-9-18(10-12-32)14-30-20-8-4-7-19(13-20)25-22(29)24(38-15-21(33)34)26(39-25)27(35)36/h3-8,13,18,30H,9-12,14-15H2,1-2H3,(H,31,37)(H,33,34)(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219585
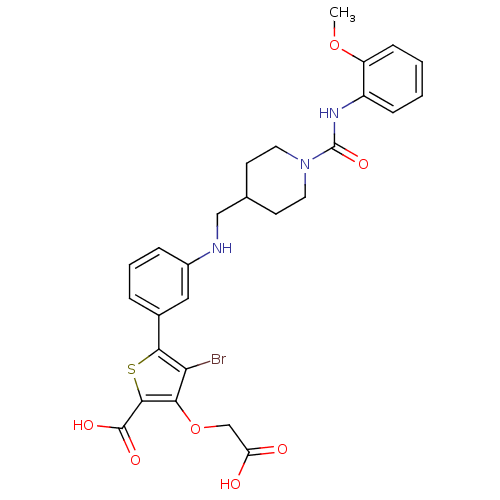
(4-bromo-3-carboxymethoxy-5-(3-{[1-(2-methoxyphenyl...)Show SMILES COc1ccccc1NC(=O)N1CCC(CNc2cccc(c2)-c2sc(C(O)=O)c(OCC(O)=O)c2Br)CC1 Show InChI InChI=1S/C27H28BrN3O7S/c1-37-20-8-3-2-7-19(20)30-27(36)31-11-9-16(10-12-31)14-29-18-6-4-5-17(13-18)24-22(28)23(38-15-21(32)33)25(39-24)26(34)35/h2-8,13,16,29H,9-12,14-15H2,1H3,(H,30,36)(H,32,33)(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219568

(4-bromo-3-carboxymethoxy-5-(3-{[1-(2-chlorophenylc...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NCC2CCN(CC2)C(=O)Nc2ccccc2Cl)c1 Show InChI InChI=1S/C26H25BrClN3O6S/c27-21-22(37-14-20(32)33)24(25(34)35)38-23(21)16-4-3-5-17(12-16)29-13-15-8-10-31(11-9-15)26(36)30-19-7-2-1-6-18(19)28/h1-7,12,15,29H,8-11,13-14H2,(H,30,36)(H,32,33)(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219566
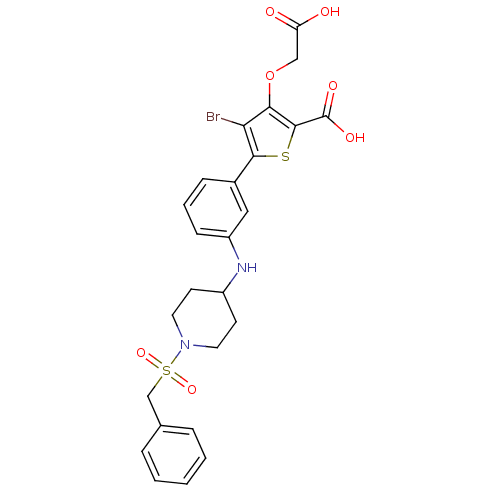
(4-bromo-3-carboxymethoxy-5-[3-(1-phenylmethanesulf...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2)c1 Show InChI InChI=1S/C25H25BrN2O7S2/c26-21-22(35-14-20(29)30)24(25(31)32)36-23(21)17-7-4-8-19(13-17)27-18-9-11-28(12-10-18)37(33,34)15-16-5-2-1-3-6-16/h1-8,13,18,27H,9-12,14-15H2,(H,29,30)(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219575
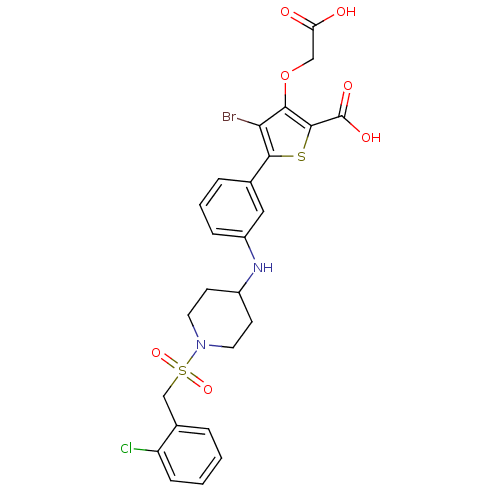
(3-carboxymethoxy-5-{3-[1-(2-chlorobenzenesulfonyl)...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2Cl)c1 Show InChI InChI=1S/C25H24BrClN2O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)15-5-3-6-18(12-15)28-17-8-10-29(11-9-17)38(34,35)14-16-4-1-2-7-19(16)27/h1-7,12,17,28H,8-11,13-14H2,(H,30,31)(H,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219567
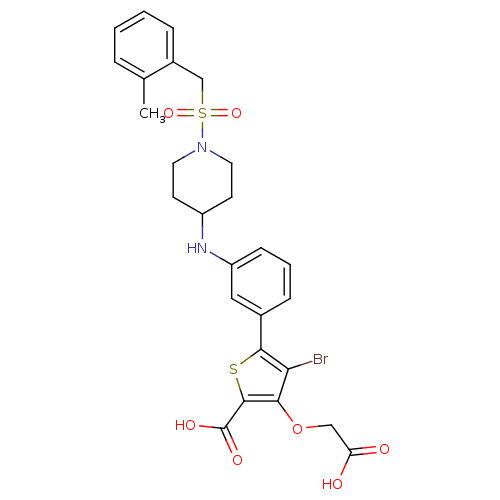
(4-bromo-3-(carboxymethoxy)-5-[3-({1-[(2-methylbenz...)Show SMILES Cc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C26H27BrN2O7S2/c1-16-5-2-3-6-18(16)15-38(34,35)29-11-9-19(10-12-29)28-20-8-4-7-17(13-20)24-22(27)23(36-14-21(30)31)25(37-24)26(32)33/h2-8,13,19,28H,9-12,14-15H2,1H3,(H,30,31)(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50090211
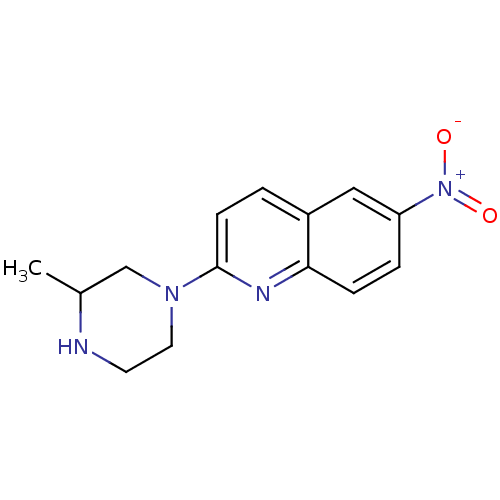
(2-(3-Methyl-piperazin-1-yl)-6-nitro-quinoline | CH...)Show InChI InChI=1S/C14H16N4O2/c1-10-9-17(7-6-15-10)14-5-2-11-8-12(18(19)20)3-4-13(11)16-14/h2-5,8,10,15H,6-7,9H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Central Washington University
Curated by ChEMBL
| Assay Description
In vitro radioligand [3H]-paroxetine from rat cortical Serotonin transporter |
Bioorg Med Chem Lett 10: 2643-6 (2000)
BindingDB Entry DOI: 10.7270/Q2W37VJW |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315675
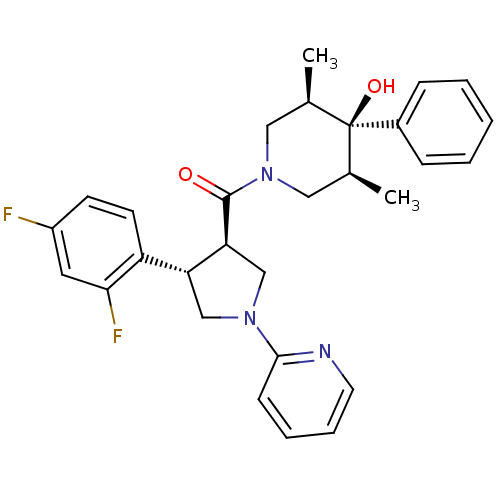
(((3R,4S)-4-(2,4-difluorophenyl)-1-(pyridin-2-yl)py...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)c1ccccn1 |r| Show InChI InChI=1S/C29H31F2N3O2/c1-19-15-34(16-20(2)29(19,36)21-8-4-3-5-9-21)28(35)25-18-33(27-10-6-7-13-32-27)17-24(25)23-12-11-22(30)14-26(23)31/h3-14,19-20,24-25,36H,15-18H2,1-2H3/t19-,20+,24-,25+,29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219566

(4-bromo-3-carboxymethoxy-5-[3-(1-phenylmethanesulf...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2)c1 Show InChI InChI=1S/C25H25BrN2O7S2/c26-21-22(35-14-20(29)30)24(25(31)32)36-23(21)17-7-4-8-19(13-17)27-18-9-11-28(12-10-18)37(33,34)15-16-5-2-1-3-6-16/h1-8,13,18,27H,9-12,14-15H2,(H,29,30)(H,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219570

(4-bromo-3-carboxymethoxy-5-{3-[(1-o-tolylcarbamoyl...)Show SMILES Cc1ccccc1NC(=O)N1CCC(CNc2cccc(c2)-c2sc(C(O)=O)c(OCC(O)=O)c2Br)CC1 Show InChI InChI=1S/C27H28BrN3O6S/c1-16-5-2-3-8-20(16)30-27(36)31-11-9-17(10-12-31)14-29-19-7-4-6-18(13-19)24-22(28)23(37-15-21(32)33)25(38-24)26(34)35/h2-8,13,17,29H,9-12,14-15H2,1H3,(H,30,36)(H,32,33)(H,34,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315688
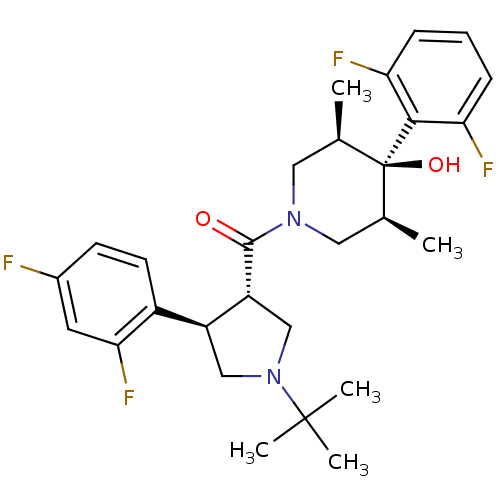
(((3S,4R)-1-tert-butyl-4-(2,4-difluorophenyl)pyrrol...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1c(F)cccc1F)C(=O)[C@@H]1CN(C[C@H]1c1ccc(F)cc1F)C(C)(C)C |r| Show InChI InChI=1S/C28H34F4N2O2/c1-16-12-33(13-17(2)28(16,36)25-22(30)7-6-8-23(25)31)26(35)21-15-34(27(3,4)5)14-20(21)19-10-9-18(29)11-24(19)32/h6-11,16-17,20-21,36H,12-15H2,1-5H3/t16-,17+,20-,21+,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219575

(3-carboxymethoxy-5-{3-[1-(2-chlorobenzenesulfonyl)...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2Cl)c1 Show InChI InChI=1S/C25H24BrClN2O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)15-5-3-6-18(12-15)28-17-8-10-29(11-9-17)38(34,35)14-16-4-1-2-7-19(16)27/h1-7,12,17,28H,8-11,13-14H2,(H,30,31)(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219585

(4-bromo-3-carboxymethoxy-5-(3-{[1-(2-methoxyphenyl...)Show SMILES COc1ccccc1NC(=O)N1CCC(CNc2cccc(c2)-c2sc(C(O)=O)c(OCC(O)=O)c2Br)CC1 Show InChI InChI=1S/C27H28BrN3O7S/c1-37-20-8-3-2-7-19(20)30-27(36)31-11-9-16(10-12-31)14-29-18-6-4-5-17(13-18)24-22(28)23(38-15-21(32)33)25(39-24)26(34)35/h2-8,13,16,29H,9-12,14-15H2,1H3,(H,30,36)(H,32,33)(H,34,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315674
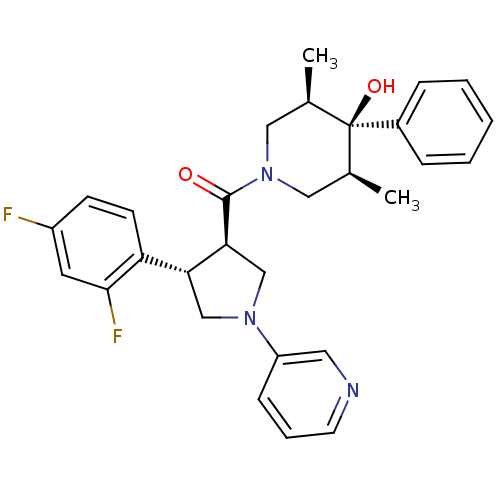
(((3R,4S)-4-(2,4-difluorophenyl)-1-(pyridin-3-yl)py...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)c1cccnc1 |r| Show InChI InChI=1S/C29H31F2N3O2/c1-19-15-34(16-20(2)29(19,36)21-7-4-3-5-8-21)28(35)26-18-33(23-9-6-12-32-14-23)17-25(26)24-11-10-22(30)13-27(24)31/h3-14,19-20,25-26,36H,15-18H2,1-2H3/t19-,20+,25-,26+,29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315676
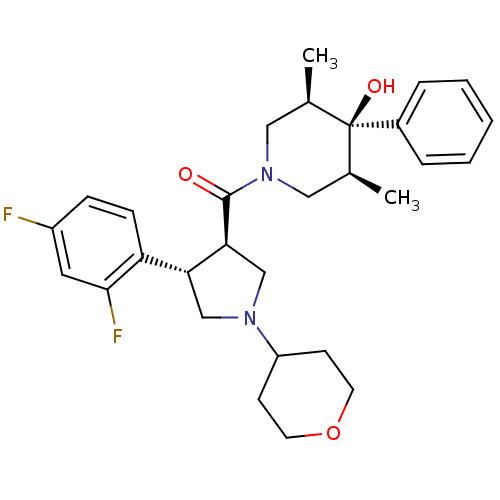
(((3R,4S)-4-(2,4-difluorophenyl)-1-(tetrahydro-2H-p...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)C1CCOCC1 |r| Show InChI InChI=1S/C29H36F2N2O3/c1-19-15-33(16-20(2)29(19,35)21-6-4-3-5-7-21)28(34)26-18-32(23-10-12-36-13-11-23)17-25(26)24-9-8-22(30)14-27(24)31/h3-9,14,19-20,23,25-26,35H,10-13,15-18H2,1-2H3/t19-,20+,25-,26+,29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315686
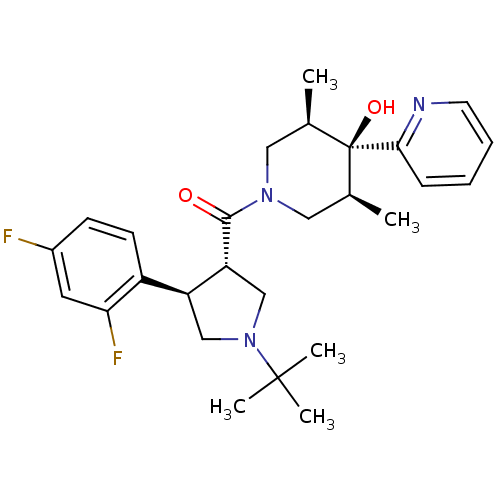
(((3S,4R)-1-tert-butyl-4-(2,4-difluorophenyl)pyrrol...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccn1)C(=O)[C@@H]1CN(C[C@H]1c1ccc(F)cc1F)C(C)(C)C |r| Show InChI InChI=1S/C27H35F2N3O2/c1-17-13-31(14-18(2)27(17,34)24-8-6-7-11-30-24)25(33)22-16-32(26(3,4)5)15-21(22)20-10-9-19(28)12-23(20)29/h6-12,17-18,21-22,34H,13-16H2,1-5H3/t17-,18+,21-,22+,27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 5.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315677

(((3R,4S)-1-cyclobutyl-4-(2,4-difluorophenyl)pyrrol...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)C1CCC1 |r| Show InChI InChI=1S/C28H34F2N2O2/c1-18-14-32(15-19(2)28(18,34)20-7-4-3-5-8-20)27(33)25-17-31(22-9-6-10-22)16-24(25)23-12-11-21(29)13-26(23)30/h3-5,7-8,11-13,18-19,22,24-25,34H,6,9-10,14-17H2,1-2H3/t18-,19+,24-,25+,28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219568

(4-bromo-3-carboxymethoxy-5-(3-{[1-(2-chlorophenylc...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NCC2CCN(CC2)C(=O)Nc2ccccc2Cl)c1 Show InChI InChI=1S/C26H25BrClN3O6S/c27-21-22(37-14-20(32)33)24(25(34)35)38-23(21)16-4-3-5-17(12-16)29-13-15-8-10-31(11-9-15)26(36)30-19-7-2-1-6-18(19)28/h1-7,12,15,29H,8-11,13-14H2,(H,30,36)(H,32,33)(H,34,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219567

(4-bromo-3-(carboxymethoxy)-5-[3-({1-[(2-methylbenz...)Show SMILES Cc1ccccc1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C26H27BrN2O7S2/c1-16-5-2-3-6-18(16)15-38(34,35)29-11-9-19(10-12-29)28-20-8-4-7-17(13-20)24-22(27)23(36-14-21(30)31)25(37-24)26(32)33/h2-8,13,19,28H,9-12,14-15H2,1H3,(H,30,31)(H,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219565
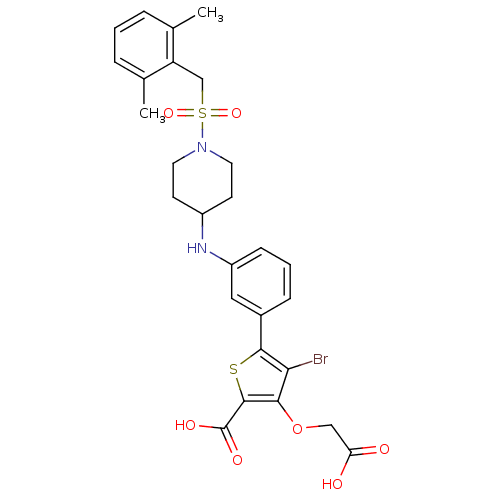
(4-bromo-3-(carboxymethoxy)-5-[3-({1-[(2,6-dimethyl...)Show SMILES Cc1cccc(C)c1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C27H29BrN2O7S2/c1-16-5-3-6-17(2)21(16)15-39(35,36)30-11-9-19(10-12-30)29-20-8-4-7-18(13-20)25-23(28)24(37-14-22(31)32)26(38-25)27(33)34/h3-8,13,19,29H,9-12,14-15H2,1-2H3,(H,31,32)(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219586

(4-bromo-3-(carboxymethoxy)-5-{3-[(1-{[2-(trifluoro...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2C(F)(F)F)c1 Show InChI InChI=1S/C26H24BrF3N2O7S2/c27-21-22(39-13-20(33)34)24(25(35)36)40-23(21)15-5-3-6-18(12-15)31-17-8-10-32(11-9-17)41(37,38)14-16-4-1-2-7-19(16)26(28,29)30/h1-7,12,17,31H,8-11,13-14H2,(H,33,34)(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315680
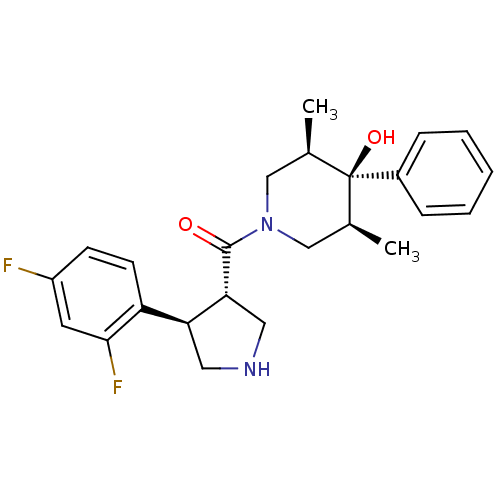
(((3S,4R)-4-(2,4-difluorophenyl)pyrrolidin-3-yl)((3...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@@H]1CNC[C@H]1c1ccc(F)cc1F |r| Show InChI InChI=1S/C24H28F2N2O2/c1-15-13-28(14-16(2)24(15,30)17-6-4-3-5-7-17)23(29)21-12-27-11-20(21)19-9-8-18(25)10-22(19)26/h3-10,15-16,20-21,27,30H,11-14H2,1-2H3/t15-,16+,20-,21+,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219591
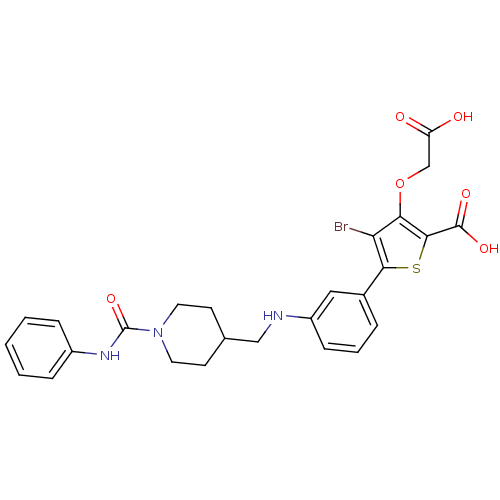
(5-[3-({[1-(anilinocarbonyl)piperidin-4-yl]methyl}a...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NCC2CCN(CC2)C(=O)Nc2ccccc2)c1 Show InChI InChI=1S/C26H26BrN3O6S/c27-21-22(36-15-20(31)32)24(25(33)34)37-23(21)17-5-4-8-19(13-17)28-14-16-9-11-30(12-10-16)26(35)29-18-6-2-1-3-7-18/h1-8,13,16,28H,9-12,14-15H2,(H,29,35)(H,31,32)(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315681
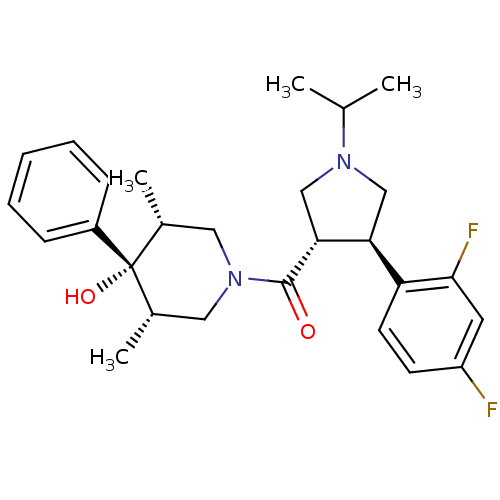
(((3S,4R)-4-(2,4-difluorophenyl)-1-isopropylpyrroli...)Show SMILES CC(C)N1C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1C[C@H](C)[C@](O)([C@H](C)C1)c1ccccc1 |r| Show InChI InChI=1S/C27H34F2N2O2/c1-17(2)30-15-23(22-11-10-21(28)12-25(22)29)24(16-30)26(32)31-13-18(3)27(33,19(4)14-31)20-8-6-5-7-9-20/h5-12,17-19,23-24,33H,13-16H2,1-4H3/t18-,19+,23-,24+,27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50378743
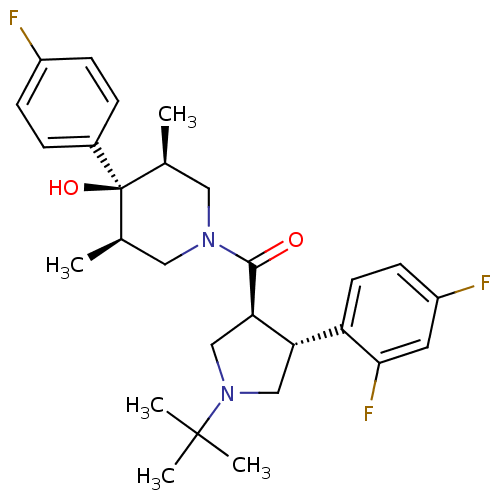
(CHEMBL1204061)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccc(F)cc1)C(=O)[C@@H]1CN(C[C@H]1c1ccc(F)cc1F)C(C)(C)C |r| Show InChI InChI=1S/C28H35F3N2O2/c1-17-13-32(14-18(2)28(17,35)19-6-8-20(29)9-7-19)26(34)24-16-33(27(3,4)5)15-23(24)22-11-10-21(30)12-25(22)31/h6-12,17-18,23-24,35H,13-16H2,1-5H3/t17-,18+,23-,24+,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219589
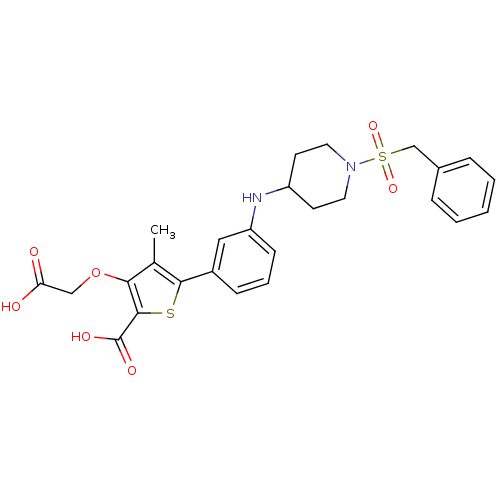
(5-(3-{[1-(benzylsulfonyl)piperidin-4-yl]amino}phen...)Show SMILES Cc1c(OCC(O)=O)c(sc1-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2)c1)C(O)=O Show InChI InChI=1S/C26H28N2O7S2/c1-17-23(35-15-22(29)30)25(26(31)32)36-24(17)19-8-5-9-21(14-19)27-20-10-12-28(13-11-20)37(33,34)16-18-6-3-2-4-7-18/h2-9,14,20,27H,10-13,15-16H2,1H3,(H,29,30)(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219565

(4-bromo-3-(carboxymethoxy)-5-[3-({1-[(2,6-dimethyl...)Show SMILES Cc1cccc(C)c1CS(=O)(=O)N1CCC(CC1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C27H29BrN2O7S2/c1-16-5-3-6-17(2)21(16)15-39(35,36)30-11-9-19(10-12-30)29-20-8-4-7-18(13-20)25-23(28)24(37-14-22(31)32)26(38-25)27(33)34/h3-8,13,19,29H,9-12,14-15H2,1-2H3,(H,31,32)(H,33,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Caspase-1
(Homo sapiens (Human)) | BDBM50290014
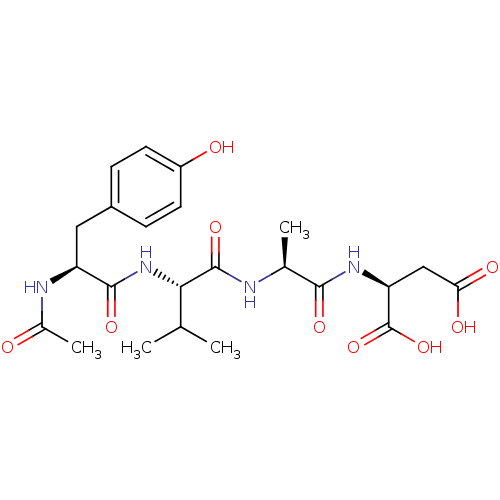
((S)-2-((S)-2-{(S)-2-[(S)-2-Acetylamino-3-(4-hydrox...)Show SMILES CC(C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(O)=O)C(O)=O Show InChI InChI=1S/C23H32N4O9/c1-11(2)19(22(34)24-12(3)20(32)26-17(23(35)36)10-18(30)31)27-21(33)16(25-13(4)28)9-14-5-7-15(29)8-6-14/h5-8,11-12,16-17,19,29H,9-10H2,1-4H3,(H,24,34)(H,25,28)(H,26,32)(H,27,33)(H,30,31)(H,35,36)/t12-,16-,17-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of IL-1 beta converting enzyme |
Bioorg Med Chem Lett 7: 2181-2186 (1997)
Article DOI: 10.1016/S0960-894X(97)00394-6
BindingDB Entry DOI: 10.7270/Q2VH5NT2 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219569

(4-bromo-3-carboxymethoxy-5-(3-{[1-(2,6-dimethylphe...)Show SMILES Cc1cccc(C)c1NC(=O)N1CCC(CNc2cccc(c2)-c2sc(C(O)=O)c(OCC(O)=O)c2Br)CC1 Show InChI InChI=1S/C28H30BrN3O6S/c1-16-5-3-6-17(2)23(16)31-28(37)32-11-9-18(10-12-32)14-30-20-8-4-7-19(13-20)25-22(29)24(38-15-21(33)34)26(39-25)27(35)36/h3-8,13,18,30H,9-12,14-15H2,1-2H3,(H,31,37)(H,33,34)(H,35,36) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219576
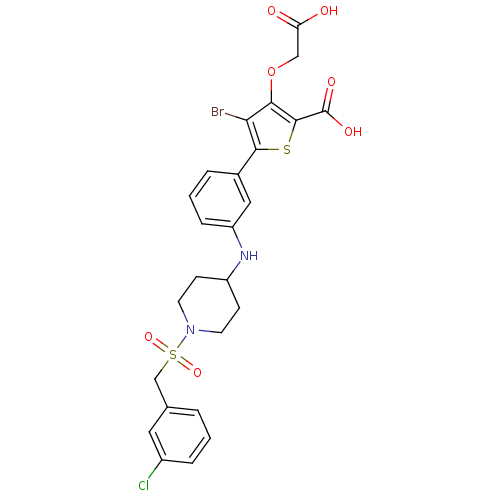
(4-bromo-3-(carboxymethoxy)-5-(3-(1-(3-chlorobenzyl...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2cccc(Cl)c2)c1 Show InChI InChI=1S/C25H24BrClN2O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)16-4-2-6-19(12-16)28-18-7-9-29(10-8-18)38(34,35)14-15-3-1-5-17(27)11-15/h1-6,11-12,18,28H,7-10,13-14H2,(H,30,31)(H,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315679

(((3R,4S)-1-cyclopropyl-4-(2,4-difluorophenyl)pyrro...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)C1CC1 |r| Show InChI InChI=1S/C27H32F2N2O2/c1-17-13-31(14-18(2)27(17,33)19-6-4-3-5-7-19)26(32)24-16-30(21-9-10-21)15-23(24)22-11-8-20(28)12-25(22)29/h3-8,11-12,17-18,21,23-24,33H,9-10,13-16H2,1-2H3/t17-,18+,23-,24+,27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >9.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50219576

(4-bromo-3-(carboxymethoxy)-5-(3-(1-(3-chlorobenzyl...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2cccc(Cl)c2)c1 Show InChI InChI=1S/C25H24BrClN2O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)16-4-2-6-19(12-16)28-18-7-9-29(10-8-18)38(34,35)14-15-3-1-5-17(27)11-15/h1-6,11-12,18,28H,7-10,13-14H2,(H,30,31)(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM50219580
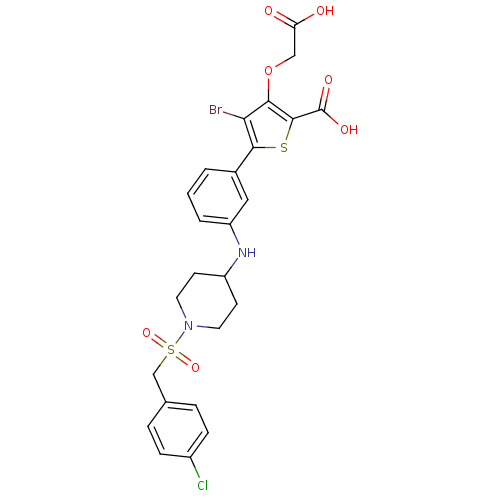
(4-bromo-3-(carboxymethoxy)-5-[3-({1-[(4-chlorobenz...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccc(Cl)cc2)c1 Show InChI InChI=1S/C25H24BrClN2O7S2/c26-21-22(36-13-20(30)31)24(25(32)33)37-23(21)16-2-1-3-19(12-16)28-18-8-10-29(11-9-18)38(34,35)14-15-4-6-17(27)7-5-15/h1-7,12,18,28H,8-11,13-14H2,(H,30,31)(H,32,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant TCPTP |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315673
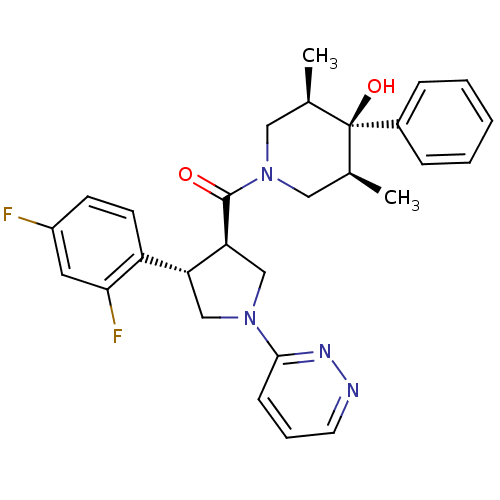
(((3R,4S)-4-(2,4-difluorophenyl)-1-(pyridazin-3-yl)...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)c1cccnn1 |r| Show InChI InChI=1S/C28H30F2N4O2/c1-18-14-34(15-19(2)28(18,36)20-7-4-3-5-8-20)27(35)24-17-33(26-9-6-12-31-32-26)16-23(24)22-11-10-21(29)13-25(22)30/h3-13,18-19,23-24,36H,14-17H2,1-2H3/t18-,19+,23-,24+,28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50378748
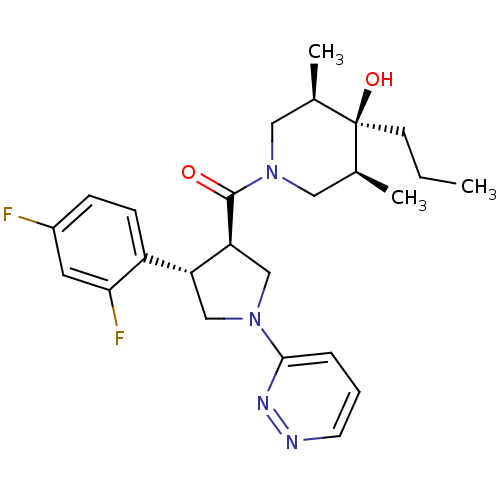
(CHEMBL1204054)Show SMILES CCC[C@]1(O)[C@@H](C)CN(C[C@H]1C)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)c1cccnn1 |r| Show InChI InChI=1S/C25H32F2N4O2/c1-4-9-25(33)16(2)12-31(13-17(25)3)24(32)21-15-30(23-6-5-10-28-29-23)14-20(21)19-8-7-18(26)11-22(19)27/h5-8,10-11,16-17,20-21,33H,4,9,12-15H2,1-3H3/t16-,17+,20-,21+,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50315673

(((3R,4S)-4-(2,4-difluorophenyl)-1-(pyridazin-3-yl)...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)c1cccnn1 |r| Show InChI InChI=1S/C28H30F2N4O2/c1-18-14-34(15-19(2)28(18,36)20-7-4-3-5-8-20)27(35)24-17-33(26-9-6-12-31-32-26)16-23(24)22-11-10-21(29)13-25(22)30/h3-13,18-19,23-24,36H,14-17H2,1-2H3/t18-,19+,23-,24+,28-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H] melanocortin-2 from human recombinant MC4 receptor expressed in CHO cells by scintillation counting |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50315669
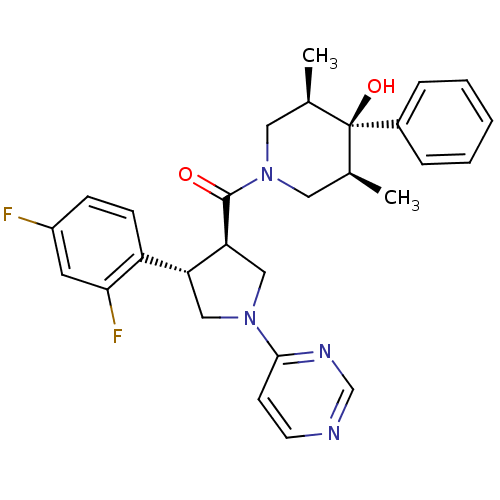
(((3R,4S)-4-(2,4-difluorophenyl)-1-(pyrimidin-4-yl)...)Show SMILES C[C@H]1CN(C[C@@H](C)[C@]1(O)c1ccccc1)C(=O)[C@H]1CN(C[C@@H]1c1ccc(F)cc1F)c1ccncn1 |r| Show InChI InChI=1S/C28H30F2N4O2/c1-18-13-34(14-19(2)28(18,36)20-6-4-3-5-7-20)27(35)24-16-33(26-10-11-31-17-32-26)15-23(24)22-9-8-21(29)12-25(22)30/h3-12,17-19,23-24,36H,13-16H2,1-2H3/t18-,19+,23-,24+,28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in CHO cells by patch clamp method |
J Med Chem 53: 3183-97 (2010)
Article DOI: 10.1021/jm9017866
BindingDB Entry DOI: 10.7270/Q20G3M4T |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data