 Found 2169 hits with Last Name = 'wood' and Initial = 'hb'
Found 2169 hits with Last Name = 'wood' and Initial = 'hb' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581548
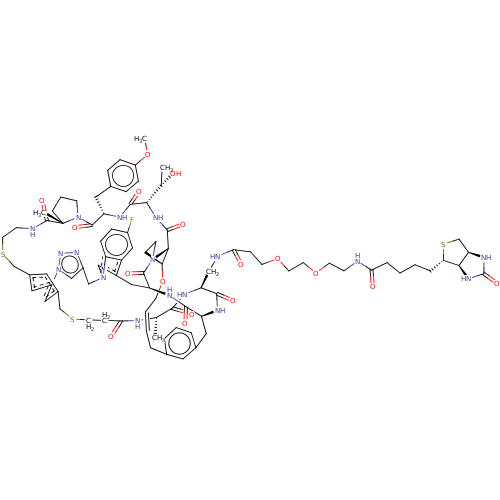
(CHEMBL5085124)Show SMILES [H][C@@]12CS[C@@H](CCCCC(=O)NCCOCCOCCC(=O)NC[C@@H]3NC(=O)[C@H](C)NC(=O)CCSCc4cc5CSCCNC(=O)[C@]6(C)CCCN6C(=O)[C@H](Cc6ccc(OC)cc6)NC(=O)[C@@H](NC(=O)[C@@H]6[C@@H]7CCN6C(=O)[C@H](Cc6cn(Cc8cn(nn8)-c(c5)c4)c4ccc(F)cc64)NC(=O)[C@H](Cc4cccc(C\C=C\CO7)c4)NC3=O)[C@H](C)O)[C@]1([H])NC(=O)N2 |r,t:119| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET ultra assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581547
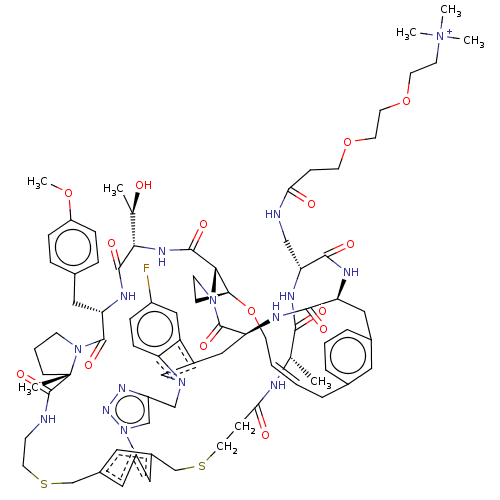
(CHEMBL5081349)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](C)C(=O)N[C@H](CNC(=O)CCOCCOCC[N+](C)(C)C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:97| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.000930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET ultra assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581546
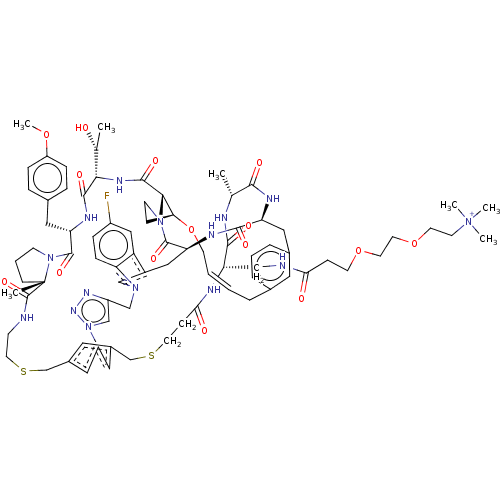
(CHEMBL5084416)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CNC(=O)CCOCCOCC[N+](C)(C)C)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:97| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00239 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET ultra assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581547

(CHEMBL5081349)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](C)C(=O)N[C@H](CNC(=O)CCOCCOCC[N+](C)(C)C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:97| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00736 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581545
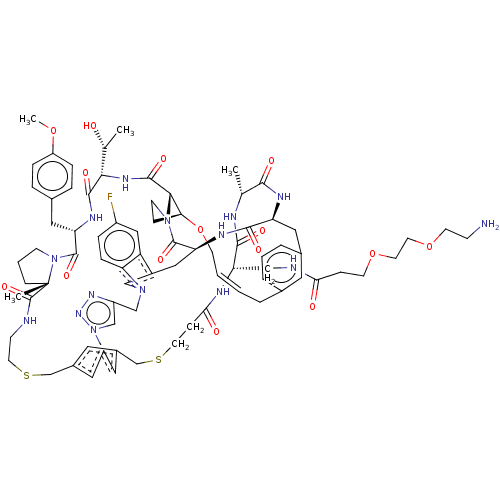
(CHEMBL5084902)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CNC(=O)CCOCCOCCN)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:94| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00813 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET ultra assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581544
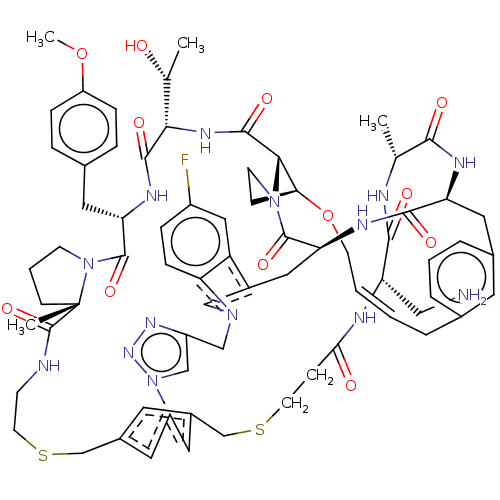
(CHEMBL5086475)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CN)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:83| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00826 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581544

(CHEMBL5086475)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CN)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:83| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET ultra assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581548

(CHEMBL5085124)Show SMILES [H][C@@]12CS[C@@H](CCCCC(=O)NCCOCCOCCC(=O)NC[C@@H]3NC(=O)[C@H](C)NC(=O)CCSCc4cc5CSCCNC(=O)[C@]6(C)CCCN6C(=O)[C@H](Cc6ccc(OC)cc6)NC(=O)[C@@H](NC(=O)[C@@H]6[C@@H]7CCN6C(=O)[C@H](Cc6cn(Cc8cn(nn8)-c(c5)c4)c4ccc(F)cc64)NC(=O)[C@H](Cc4cccc(C\C=C\CO7)c4)NC3=O)[C@H](C)O)[C@]1([H])NC(=O)N2 |r,t:119| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581546

(CHEMBL5084416)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CNC(=O)CCOCCOCC[N+](C)(C)C)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:97| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0114 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581549
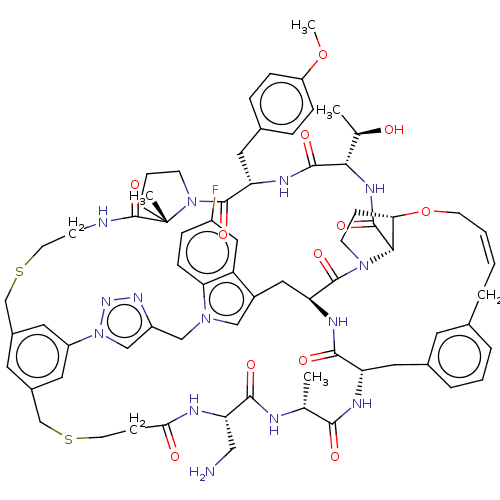
(CHEMBL5082483)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CN)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C/CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,c:83| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581546

(CHEMBL5084416)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CNC(=O)CCOCCOCC[N+](C)(C)C)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:97| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| >0.129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581548

(CHEMBL5085124)Show SMILES [H][C@@]12CS[C@@H](CCCCC(=O)NCCOCCOCCC(=O)NC[C@@H]3NC(=O)[C@H](C)NC(=O)CCSCc4cc5CSCCNC(=O)[C@]6(C)CCCN6C(=O)[C@H](Cc6ccc(OC)cc6)NC(=O)[C@@H](NC(=O)[C@@H]6[C@@H]7CCN6C(=O)[C@H](Cc6cn(Cc8cn(nn8)-c(c5)c4)c4ccc(F)cc64)NC(=O)[C@H](Cc4cccc(C\C=C\CO7)c4)NC3=O)[C@H](C)O)[C@]1([H])NC(=O)N2 |r,t:119| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >0.129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581547

(CHEMBL5081349)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](C)C(=O)N[C@H](CNC(=O)CCOCCOCC[N+](C)(C)C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:97| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >0.129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581545

(CHEMBL5084902)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CNC(=O)CCOCCOCCN)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:94| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >0.129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581544

(CHEMBL5086475)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@@H]3Cc5cn(Cc6cn(nn6)-c6cc(CSCCNC(=O)[C@]7(C)CCCN7C2=O)cc(CSCCC(=O)N[C@@H](CN)C(=O)N[C@H](C)C(=O)N[C@@H](Cc2cccc(C\C=C\CO4)c2)C(=O)N3)c6)c2ccc(F)cc52)[C@@H](C)O)cc1 |r,t:83| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >0.129 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581542
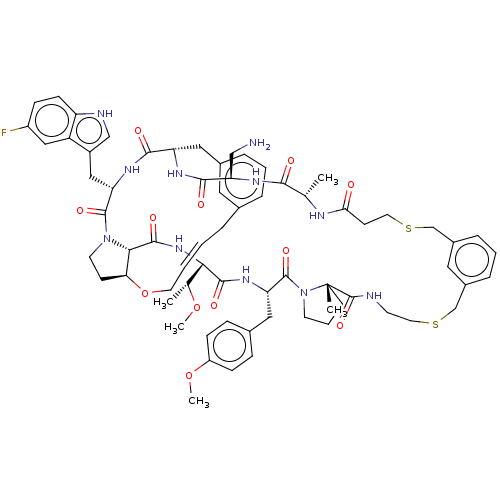
(CHEMBL5081587)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C\CO3)c2)NC(=O)[C@@H](CN)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,t:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581542

(CHEMBL5081587)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C\CO3)c2)NC(=O)[C@@H](CN)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,t:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581540
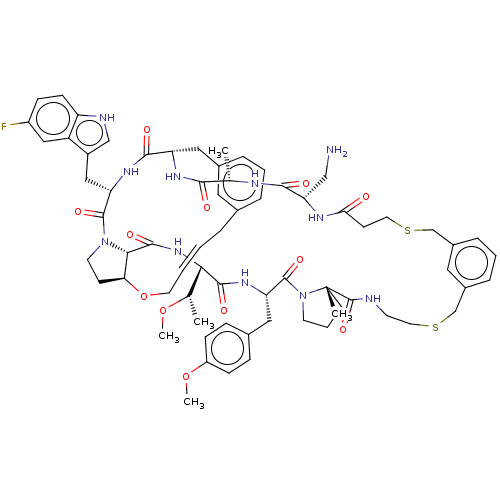
(CHEMBL5087487)Show SMILES CO[C@@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C\CO3)c2)NC(=O)[C@H](C)NC(=O)[C@H](CN)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,t:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581537
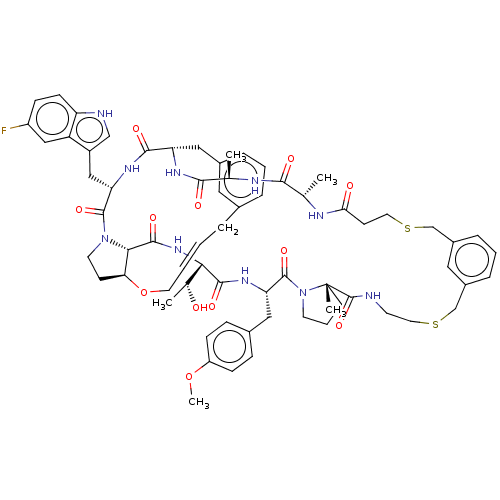
(CHEMBL5086286)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@H](Cc3c[nH]c5ccc(F)cc35)NC(=O)[C@H](Cc3cccc(C\C=C\CO4)c3)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc3cccc(CSCCNC(=O)[C@]4(C)CCCN4C2=O)c3)[C@@H](C)O)cc1 |r,t:48| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581535
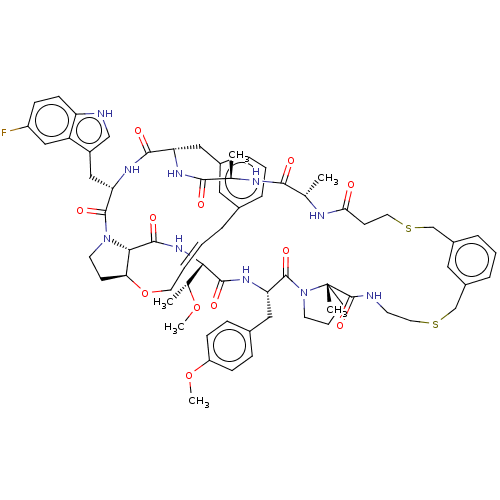
(CHEMBL5091040)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C\CO3)c2)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,t:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581537

(CHEMBL5086286)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@H](Cc3c[nH]c5ccc(F)cc35)NC(=O)[C@H](Cc3cccc(C\C=C\CO4)c3)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc3cccc(CSCCNC(=O)[C@]4(C)CCCN4C2=O)c3)[C@@H](C)O)cc1 |r,t:48| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581540

(CHEMBL5087487)Show SMILES CO[C@@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C\CO3)c2)NC(=O)[C@H](C)NC(=O)[C@H](CN)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,t:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581535

(CHEMBL5091040)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C\CO3)c2)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,t:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126919
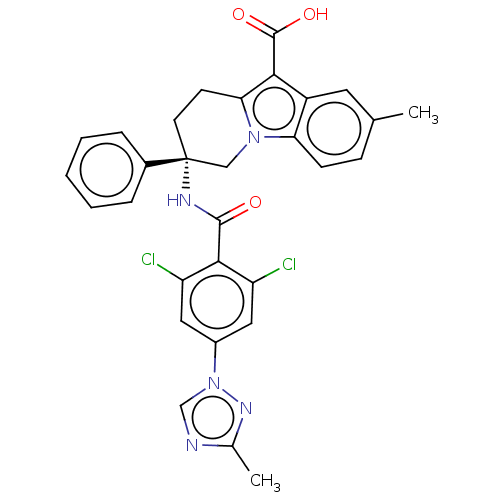
(CHEMBL3629114)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)N[C@@]2(CCc3c(C(O)=O)c4cc(C)ccc4n3C2)c2ccccc2)c(Cl)c1 |r| Show InChI InChI=1S/C30H25Cl2N5O3/c1-17-8-9-24-21(12-17)26(29(39)40)25-10-11-30(15-36(24)25,19-6-4-3-5-7-19)34-28(38)27-22(31)13-20(14-23(27)32)37-16-33-18(2)35-37/h3-9,12-14,16H,10-11,15H2,1-2H3,(H,34,38)(H,39,40)/t30-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126916
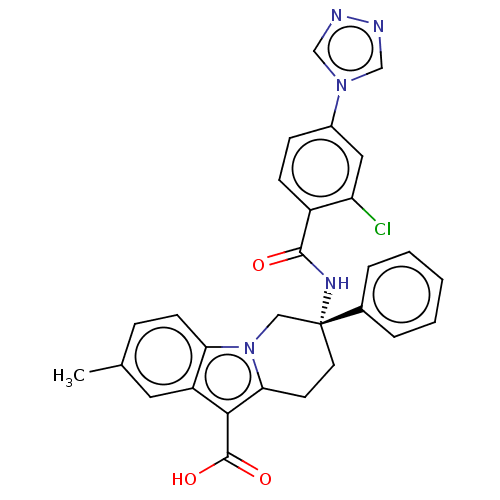
(CHEMBL3629111 | US10351558, Example 139)Show SMILES Cc1ccc2n3C[C@@](CCc3c(C(O)=O)c2c1)(NC(=O)c1ccc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C29H24ClN5O3/c1-18-7-10-24-22(13-18)26(28(37)38)25-11-12-29(15-35(24)25,19-5-3-2-4-6-19)33-27(36)21-9-8-20(14-23(21)30)34-16-31-32-17-34/h2-10,13-14,16-17H,11-12,15H2,1H3,(H,33,36)(H,37,38)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126872
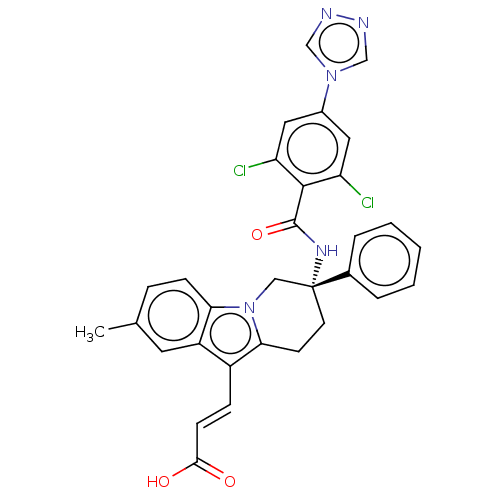
(CHEMBL3628964)Show SMILES Cc1ccc2n3C[C@@](CCc3c(\C=C\C(O)=O)c2c1)(NC(=O)c1c(Cl)cc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C31H25Cl2N5O3/c1-19-7-9-26-23(13-19)22(8-10-28(39)40)27-11-12-31(16-38(26)27,20-5-3-2-4-6-20)36-30(41)29-24(32)14-21(15-25(29)33)37-17-34-35-18-37/h2-10,13-15,17-18H,11-12,16H2,1H3,(H,36,41)(H,39,40)/b10-8+/t31-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50125979
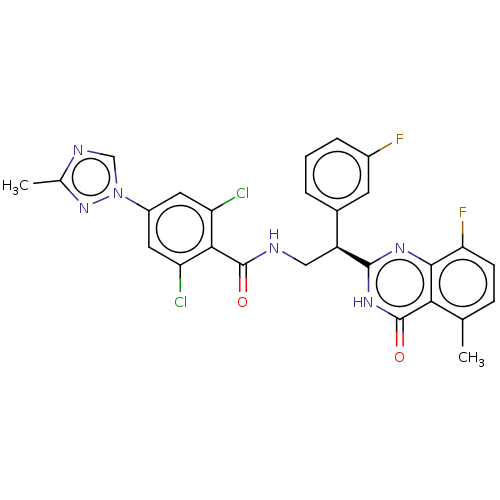
(CHEMBL3627899)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)NC[C@@H](c2cccc(F)c2)c2nc3c(F)ccc(C)c3c(=O)[nH]2)c(Cl)c1 |r| Show InChI InChI=1S/C27H20Cl2F2N6O2/c1-13-6-7-21(31)24-22(13)27(39)35-25(34-24)18(15-4-3-5-16(30)8-15)11-32-26(38)23-19(28)9-17(10-20(23)29)37-12-33-14(2)36-37/h3-10,12,18H,11H2,1-2H3,(H,32,38)(H,34,35,39)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 9a using fluorescent peptide CH3SO2-D-CHG-Gly-Arg-AFC-AcoH as substrate |
Bioorg Med Chem Lett 25: 4945-9 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.057
BindingDB Entry DOI: 10.7270/Q20Z753B |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126956
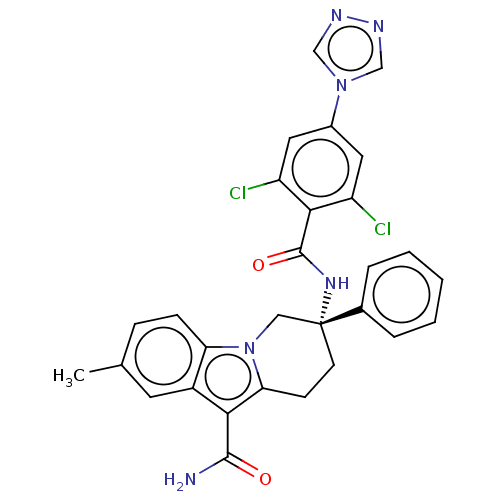
(CHEMBL3628961 | US10351558, Example 174)Show SMILES Cc1ccc2n3C[C@@](CCc3c(C(N)=O)c2c1)(NC(=O)c1c(Cl)cc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C29H24Cl2N6O2/c1-17-7-8-23-20(11-17)25(27(32)38)24-9-10-29(14-37(23)24,18-5-3-2-4-6-18)35-28(39)26-21(30)12-19(13-22(26)31)36-15-33-34-16-36/h2-8,11-13,15-16H,9-10,14H2,1H3,(H2,32,38)(H,35,39)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581543
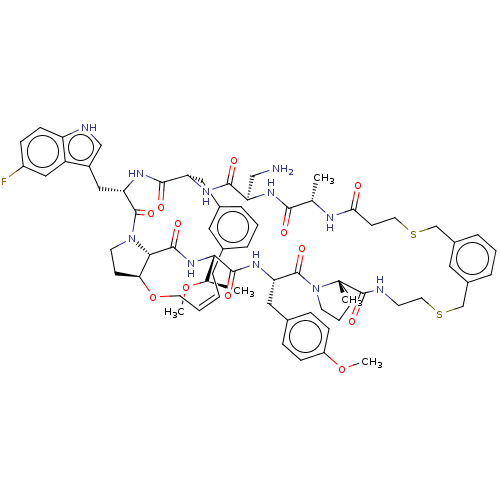
(CHEMBL5094648)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C/CO3)c2)NC(=O)[C@@H](CN)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,c:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50554754
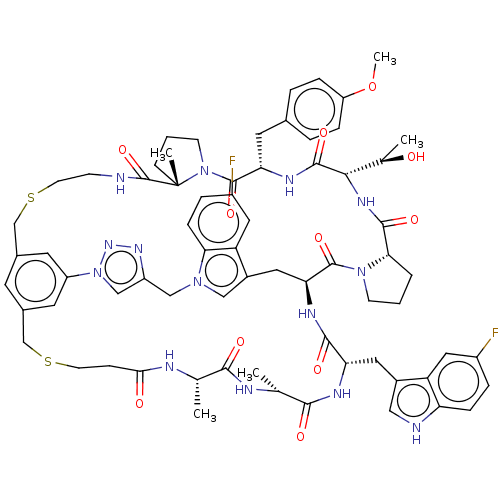
(CHEMBL4790764)Show SMILES [H][C@]1(NC(=O)[C@@H]2CCCN2C(=O)[C@@H]2Cc3cn(Cc4cn(nn4)-c4cc(CSCCNC(=O)[C@]5(C)CCCN5C(=O)[C@H](Cc5ccc(OC)cc5)NC1=O)cc(CSCCC(=O)N[C@@H](C)C(=O)N[C@H](C)C(=O)N[C@@H](Cc1c[nH]c5ccc(F)cc15)C(=O)N2)c4)c1ccc(F)cc31)[C@@H](C)O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126913
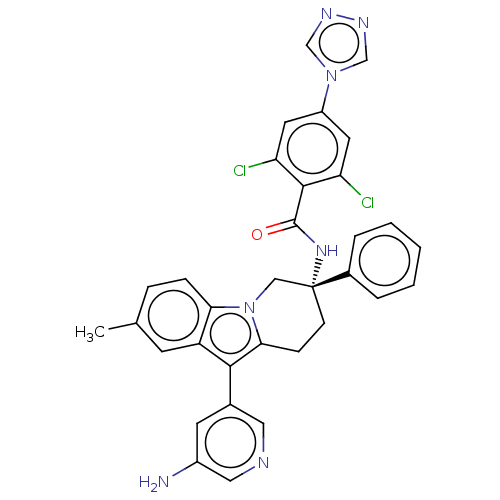
(CHEMBL3628966)Show SMILES Cc1ccc2n3C[C@@](CCc3c(-c3cncc(N)c3)c2c1)(NC(=O)c1c(Cl)cc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C33H27Cl2N7O/c1-20-7-8-28-25(11-20)30(21-12-23(36)16-37-15-21)29-9-10-33(17-42(28)29,22-5-3-2-4-6-22)40-32(43)31-26(34)13-24(14-27(31)35)41-18-38-39-19-41/h2-8,11-16,18-19H,9-10,17,36H2,1H3,(H,40,43)/t33-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126949
(CHEMBL3628954)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)N[C@@]2(CCc3nn4cc(C)ccc4c3C2)c2cccc(C)c2)c(Cl)c1 |r| Show InChI InChI=1S/C29H26Cl2N6O/c1-17-5-4-6-20(11-17)29(10-9-25-22(14-29)26-8-7-18(2)15-36(26)35-25)33-28(38)27-23(30)12-21(13-24(27)31)37-16-32-19(3)34-37/h4-8,11-13,15-16H,9-10,14H2,1-3H3,(H,33,38)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
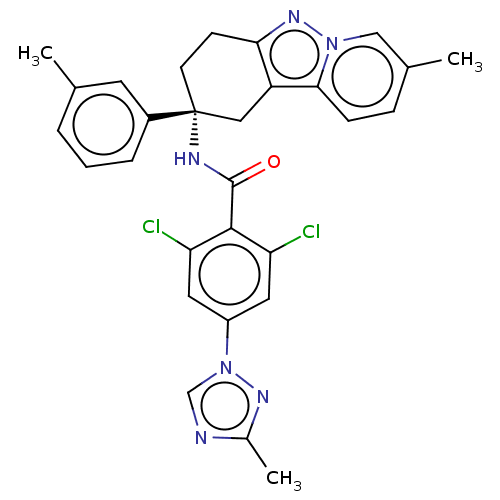
(Homo sapiens (Human)) | BDBM50126941
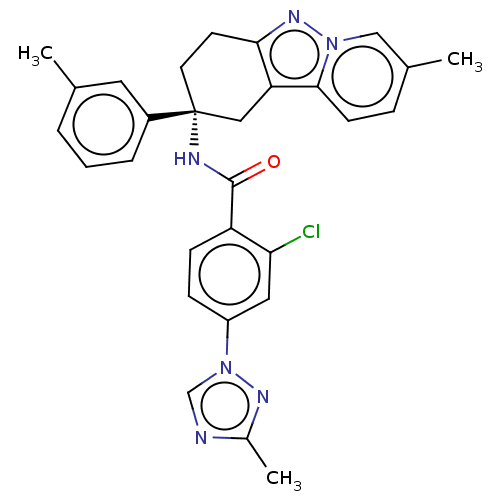
(CHEMBL3628838)Show SMILES Cc1ncn(n1)-c1ccc(C(=O)N[C@@]2(CCc3nn4cc(C)ccc4c3C2)c2cccc(C)c2)c(Cl)c1 |r| Show InChI InChI=1S/C29H27ClN6O/c1-18-5-4-6-21(13-18)29(12-11-26-24(15-29)27-10-7-19(2)16-35(27)34-26)32-28(37)23-9-8-22(14-25(23)30)36-17-31-20(3)33-36/h4-10,13-14,16-17H,11-12,15H2,1-3H3,(H,32,37)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581539
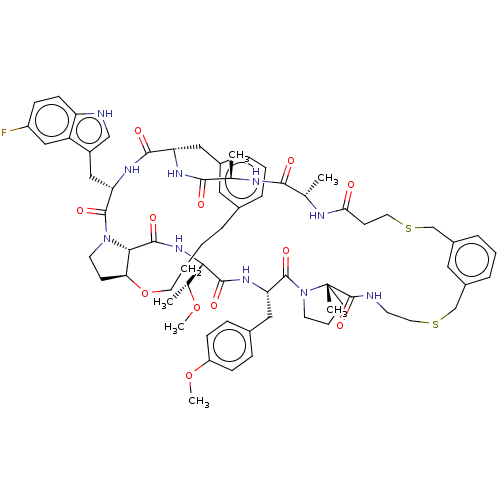
(CHEMBL5084586)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(CCCCO3)c2)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581543

(CHEMBL5094648)Show SMILES CO[C@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C/CO3)c2)NC(=O)[C@@H](CN)NC(=O)[C@H](C)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,c:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET plus assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126912
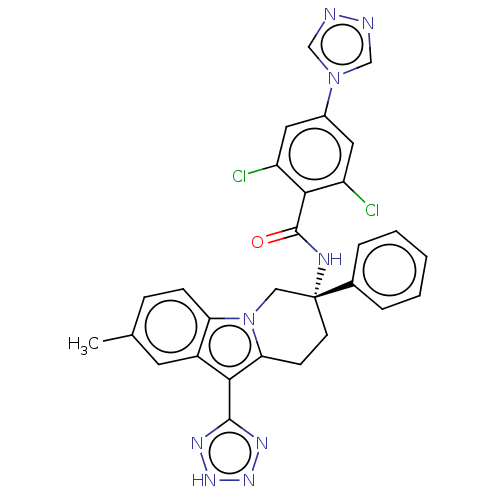
(CHEMBL3628965)Show SMILES Cc1ccc2n3C[C@@](CCc3c(-c3nn[nH]n3)c2c1)(NC(=O)c1c(Cl)cc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C29H23Cl2N9O/c1-17-7-8-23-20(11-17)25(27-35-37-38-36-27)24-9-10-29(14-40(23)24,18-5-3-2-4-6-18)34-28(41)26-21(30)12-19(13-22(26)31)39-15-32-33-16-39/h2-8,11-13,15-16H,9-10,14H2,1H3,(H,34,41)(H,35,36,37,38)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126855
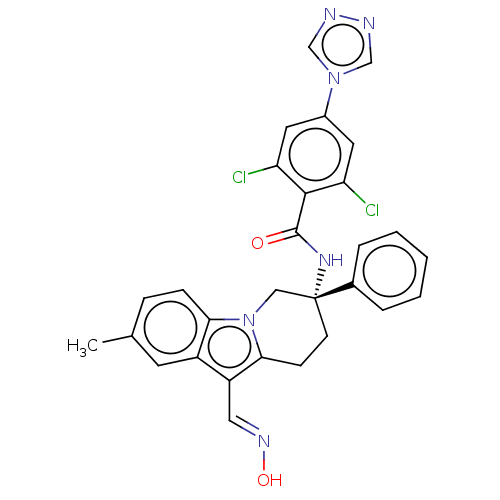
(CHEMBL3628962 | US10351558, Example 173)Show SMILES Cc1ccc2n3C[C@@](CCc3c(\C=N\O)c2c1)(NC(=O)c1c(Cl)cc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C29H24Cl2N6O2/c1-18-7-8-25-21(11-18)22(14-34-39)26-9-10-29(15-37(25)26,19-5-3-2-4-6-19)35-28(38)27-23(30)12-20(13-24(27)31)36-16-32-33-17-36/h2-8,11-14,16-17,39H,9-10,15H2,1H3,(H,35,38)/b34-14+/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
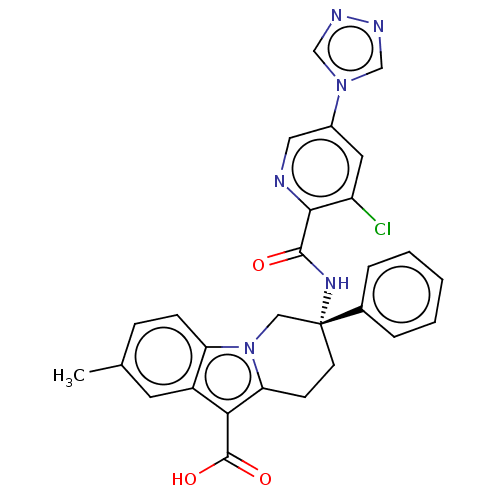
(Homo sapiens (Human)) | BDBM50126917
(CHEMBL3629112)Show SMILES Cc1ccc2n3C[C@@](CCc3c(C(O)=O)c2c1)(NC(=O)c1ncc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C28H23ClN6O3/c1-17-7-8-22-20(11-17)24(27(37)38)23-9-10-28(14-35(22)23,18-5-3-2-4-6-18)33-26(36)25-21(29)12-19(13-30-25)34-15-31-32-16-34/h2-8,11-13,15-16H,9-10,14H2,1H3,(H,33,36)(H,37,38)/t28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126915
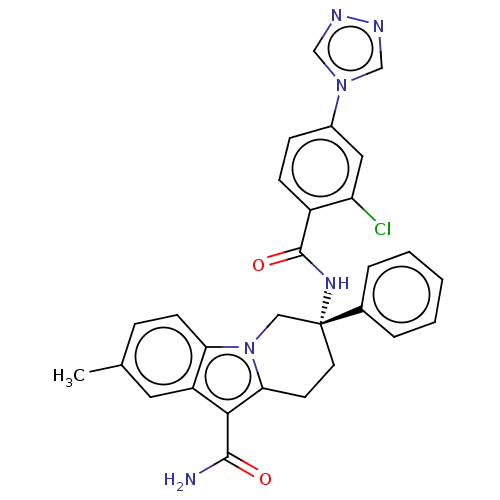
(CHEMBL3629110 | US10351558, Example 138)Show SMILES Cc1ccc2n3C[C@@](CCc3c(C(N)=O)c2c1)(NC(=O)c1ccc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C29H25ClN6O2/c1-18-7-10-24-22(13-18)26(27(31)37)25-11-12-29(15-36(24)25,19-5-3-2-4-6-19)34-28(38)21-9-8-20(14-23(21)30)35-16-32-33-17-35/h2-10,13-14,16-17H,11-12,15H2,1H3,(H2,31,37)(H,34,38)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126926
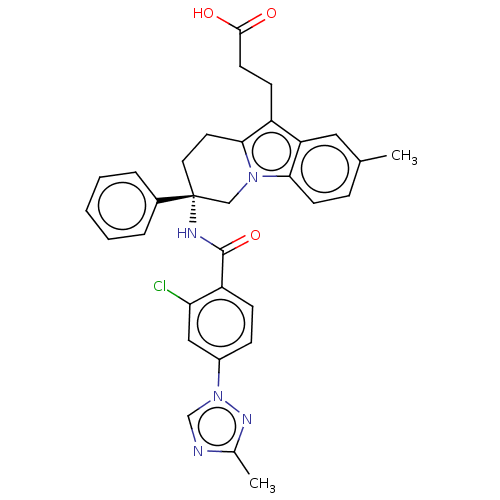
(CHEMBL3628835 | US10351558, Example 183)Show SMILES Cc1ncn(n1)-c1ccc(C(=O)N[C@@]2(CCc3c(CCC(O)=O)c4cc(C)ccc4n3C2)c2ccccc2)c(Cl)c1 |r| Show InChI InChI=1S/C32H30ClN5O3/c1-20-8-12-28-26(16-20)24(11-13-30(39)40)29-14-15-32(18-37(28)29,22-6-4-3-5-7-22)35-31(41)25-10-9-23(17-27(25)33)38-19-34-21(2)36-38/h3-10,12,16-17,19H,11,13-15,18H2,1-2H3,(H,35,41)(H,39,40)/t32-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126856
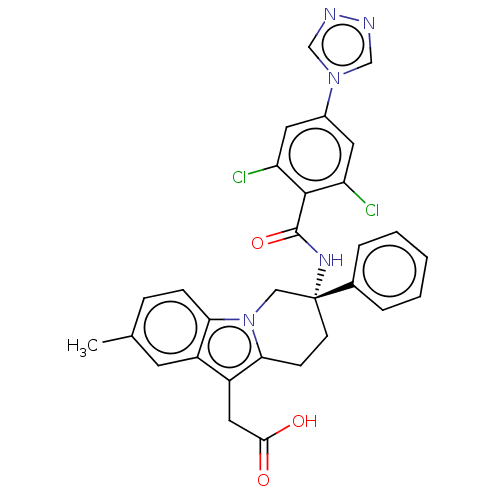
(CHEMBL3628963)Show SMILES Cc1ccc2n3C[C@@](CCc3c(CC(O)=O)c2c1)(NC(=O)c1c(Cl)cc(cc1Cl)-n1cnnc1)c1ccccc1 |r| Show InChI InChI=1S/C30H25Cl2N5O3/c1-18-7-8-25-21(11-18)22(14-27(38)39)26-9-10-30(15-37(25)26,19-5-3-2-4-6-19)35-29(40)28-23(31)12-20(13-24(28)32)36-16-33-34-17-36/h2-8,11-13,16-17H,9-10,14-15H2,1H3,(H,35,40)(H,38,39)/t30-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50125972
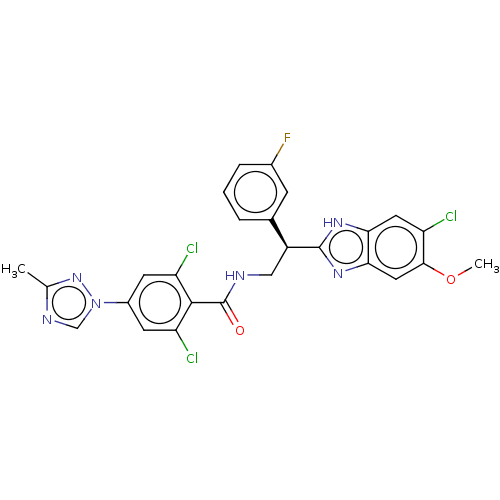
(CHEMBL3627894)Show SMILES COc1cc2nc([nH]c2cc1Cl)[C@@H](CNC(=O)c1c(Cl)cc(cc1Cl)-n1cnc(C)n1)c1cccc(F)c1 |r| Show InChI InChI=1S/C26H20Cl3FN6O2/c1-13-32-12-36(35-13)16-7-19(28)24(20(29)8-16)26(37)31-11-17(14-4-3-5-15(30)6-14)25-33-21-9-18(27)23(38-2)10-22(21)34-25/h3-10,12,17H,11H2,1-2H3,(H,31,37)(H,33,34)/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 9a using fluorescent peptide CH3SO2-D-CHG-Gly-Arg-AFC-AcoH as substrate |
Bioorg Med Chem Lett 25: 4945-9 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.057
BindingDB Entry DOI: 10.7270/Q20Z753B |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126942
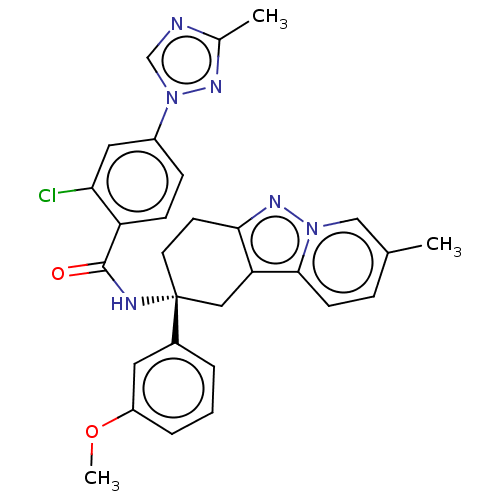
(CHEMBL3628947)Show SMILES COc1cccc(c1)[C@]1(CCc2nn3cc(C)ccc3c2C1)NC(=O)c1ccc(cc1Cl)-n1cnc(C)n1 |r| Show InChI InChI=1S/C29H27ClN6O2/c1-18-7-10-27-24-15-29(12-11-26(24)34-35(27)16-18,20-5-4-6-22(13-20)38-3)32-28(37)23-9-8-21(14-25(23)30)36-17-31-19(2)33-36/h4-10,13-14,16-17H,11-12,15H2,1-3H3,(H,32,37)/t29-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126944
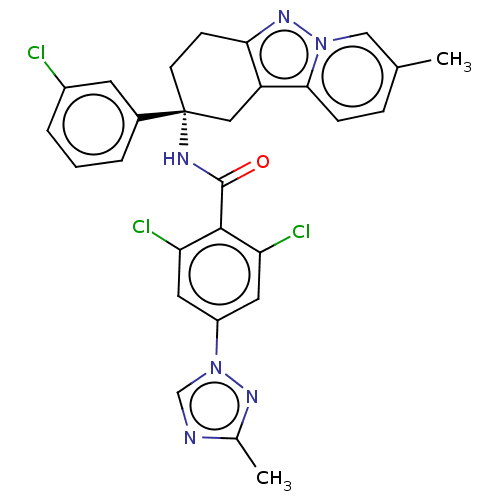
(CHEMBL3628949)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)N[C@@]2(CCc3nn4cc(C)ccc4c3C2)c2cccc(Cl)c2)c(Cl)c1 |r| Show InChI InChI=1S/C28H23Cl3N6O/c1-16-6-7-25-21-13-28(18-4-3-5-19(29)10-18,9-8-24(21)35-36(25)14-16)33-27(38)26-22(30)11-20(12-23(26)31)37-15-32-17(2)34-37/h3-7,10-12,14-15H,8-9,13H2,1-2H3,(H,33,38)/t28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50125980
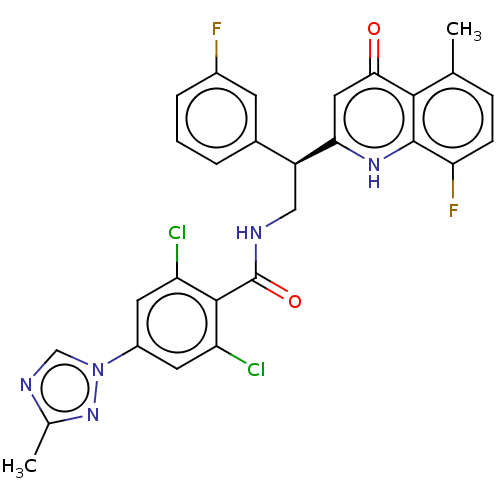
(CHEMBL3627900)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)NC[C@@H](c2cccc(F)c2)c2cc(=O)c3c(C)ccc(F)c3[nH]2)c(Cl)c1 |r| Show InChI InChI=1S/C28H21Cl2F2N5O2/c1-14-6-7-22(32)27-25(14)24(38)11-23(35-27)19(16-4-3-5-17(31)8-16)12-33-28(39)26-20(29)9-18(10-21(26)30)37-13-34-15(2)36-37/h3-11,13,19H,12H2,1-2H3,(H,33,39)(H,35,38)/t19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 9a using fluorescent peptide CH3SO2-D-CHG-Gly-Arg-AFC-AcoH as substrate |
Bioorg Med Chem Lett 25: 4945-9 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.057
BindingDB Entry DOI: 10.7270/Q20Z753B |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50125978
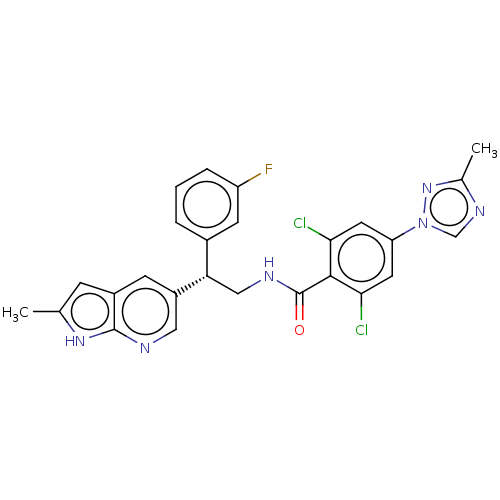
(CHEMBL3627898 | US10189819, Example 77)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)NC[C@@H](c2cccc(F)c2)c2cnc3[nH]c(C)cc3c2)c(Cl)c1 |r| Show InChI InChI=1S/C26H21Cl2FN6O/c1-14-6-17-7-18(11-30-25(17)33-14)21(16-4-3-5-19(29)8-16)12-31-26(36)24-22(27)9-20(10-23(24)28)35-13-32-15(2)34-35/h3-11,13,21H,12H2,1-2H3,(H,30,33)(H,31,36)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 9a using fluorescent peptide CH3SO2-D-CHG-Gly-Arg-AFC-AcoH as substrate |
Bioorg Med Chem Lett 25: 4945-9 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.057
BindingDB Entry DOI: 10.7270/Q20Z753B |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126946
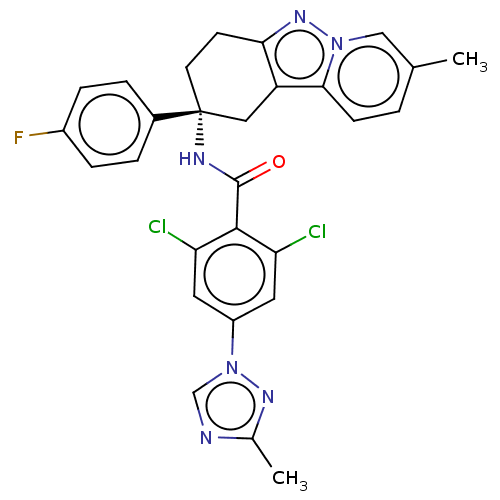
(CHEMBL3628951)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)N[C@@]2(CCc3nn4cc(C)ccc4c3C2)c2ccc(F)cc2)c(Cl)c1 |r| Show InChI InChI=1S/C28H23Cl2FN6O/c1-16-3-8-25-21-13-28(18-4-6-19(31)7-5-18,10-9-24(21)35-36(25)14-16)33-27(38)26-22(29)11-20(12-23(26)30)37-15-32-17(2)34-37/h3-8,11-12,14-15H,9-10,13H2,1-2H3,(H,33,38)/t28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581541
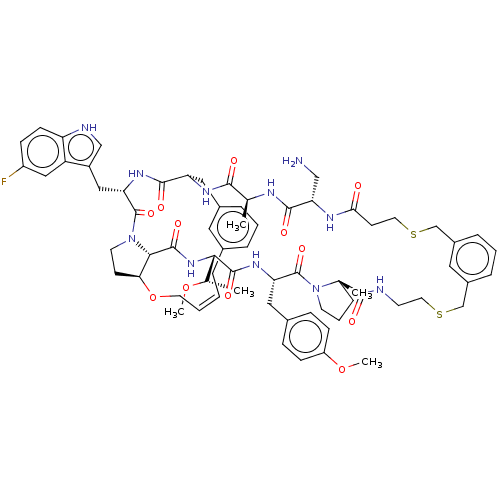
(CHEMBL5078356)Show SMILES CO[C@@H](C)[C@@H]1NC(=O)[C@@H]2[C@@H]3CCN2C(=O)[C@H](Cc2c[nH]c4ccc(F)cc24)NC(=O)[C@H](Cc2cccc(C\C=C/CO3)c2)NC(=O)[C@H](C)NC(=O)[C@H](CN)NC(=O)CCSCc2cccc(CSCCNC(=O)[C@]3(C)CCCN3C(=O)[C@H](Cc3ccc(OC)cc3)NC1=O)c2 |r,c:41| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |
Coagulation factor IX
(Homo sapiens (Human)) | BDBM50126945
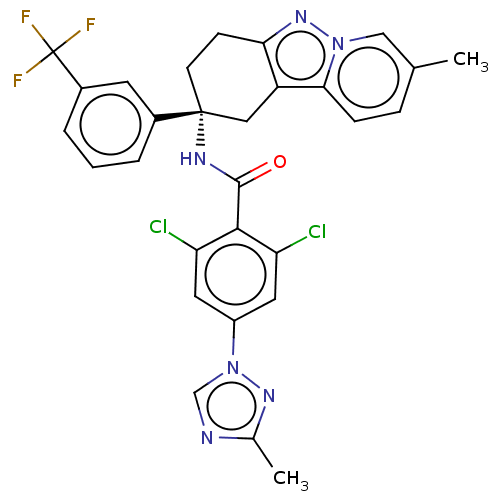
(CHEMBL3628950)Show SMILES Cc1ncn(n1)-c1cc(Cl)c(C(=O)N[C@@]2(CCc3nn4cc(C)ccc4c3C2)c2cccc(c2)C(F)(F)F)c(Cl)c1 |r| Show InChI InChI=1S/C29H23Cl2F3N6O/c1-16-6-7-25-21-13-28(9-8-24(21)38-39(25)14-16,18-4-3-5-19(10-18)29(32,33)34)36-27(41)26-22(30)11-20(12-23(26)31)40-15-35-17(2)37-40/h3-7,10-12,14-15H,8-9,13H2,1-2H3,(H,36,41)/t28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human F9a using CH3SO2-D-CHG-Gly-Arg-AFC.AcOH as substrate by fluorescence assay |
Bioorg Med Chem Lett 25: 5437-43 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.078
BindingDB Entry DOI: 10.7270/Q2B859X6 |
More data for this
Ligand-Target Pair | |
Proprotein convertase subtilisin/kexin type 9
(Homo sapiens (Human)) | BDBM50581538

(CHEMBL5081995)Show SMILES COc1ccc(C[C@@H]2NC(=O)[C@@H](NC(=O)[C@@H]3[C@@H]4CCN3C(=O)[C@H](Cc3c[nH]c5ccc(F)cc35)NC(=O)[C@H](Cc3cccc(C\C=C/CO4)c3)NC(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)CCSCc3cccc(CSCCNC(=O)[C@]4(C)CCCN4C2=O)c3)[C@@H](C)O)cc1 |r,c:48| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PCSK9 using Alexa Fluor 674 as substrate incubated for 2 hrs by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01599
BindingDB Entry DOI: 10.7270/Q2TM7G0Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data