 Found 174 hits with Last Name = 'wyss' and Initial = 'df'
Found 174 hits with Last Name = 'wyss' and Initial = 'df' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cathepsin D
(Homo sapiens (Human)) | BDBM50300228
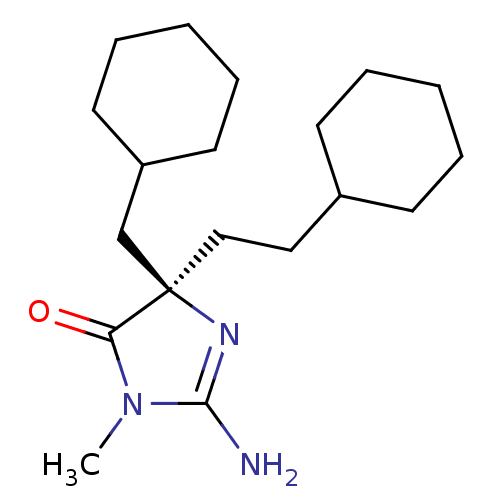
((2E,5R)-5-(2-cyclohexylethyl)-5-(cyclohexylmethyl)...)Show SMILES CN1C(N)=N[C@](CCC2CCCCC2)(CC2CCCCC2)C1=O |r,c:3| Show InChI InChI=1S/C19H33N3O/c1-22-17(23)19(21-18(22)20,14-16-10-6-3-7-11-16)13-12-15-8-4-2-5-9-15/h15-16H,2-14H2,1H3,(H2,20,21)/t19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D preincubated for 30 mins |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145828
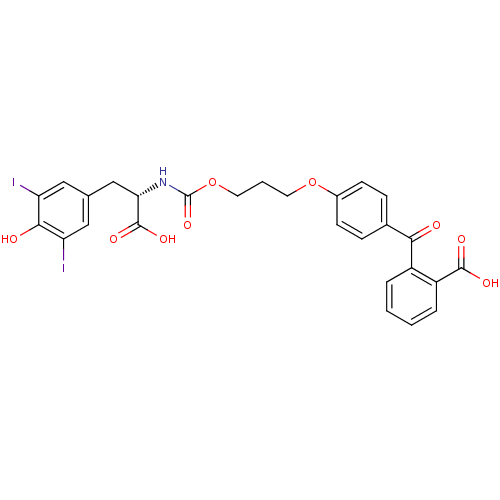
(2-(4-{3-[(S)-1-Carboxy-2-(4-hydroxy-3,5-diiodo-phe...)Show SMILES OC(=O)[C@H](Cc1cc(I)c(O)c(I)c1)NC(=O)OCCCOc1ccc(cc1)C(=O)c1ccccc1C(O)=O Show InChI InChI=1S/C27H23I2NO9/c28-20-12-15(13-21(29)24(20)32)14-22(26(35)36)30-27(37)39-11-3-10-38-17-8-6-16(7-9-17)23(31)18-4-1-2-5-19(18)25(33)34/h1-2,4-9,12-13,22,32H,3,10-11,14H2,(H,30,37)(H,33,34)(H,35,36)/t22-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145844
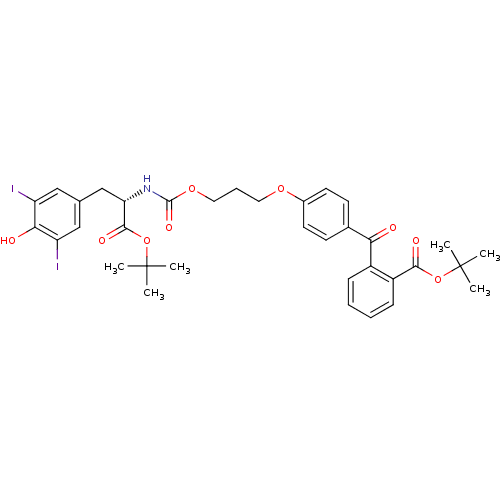
(2-(4-{3-[(S)-1-tert-Butoxycarbonyl-2-(4-hydroxy-3,...)Show SMILES CC(C)(C)OC(=O)[C@H](Cc1cc(I)c(O)c(I)c1)NC(=O)OCCCOc1ccc(cc1)C(=O)c1ccccc1C(=O)OC(C)(C)C Show InChI InChI=1S/C35H39I2NO9/c1-34(2,3)46-31(41)25-11-8-7-10-24(25)29(39)22-12-14-23(15-13-22)44-16-9-17-45-33(43)38-28(32(42)47-35(4,5)6)20-21-18-26(36)30(40)27(37)19-21/h7-8,10-15,18-19,28,40H,9,16-17,20H2,1-6H3,(H,38,43)/t28-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145820
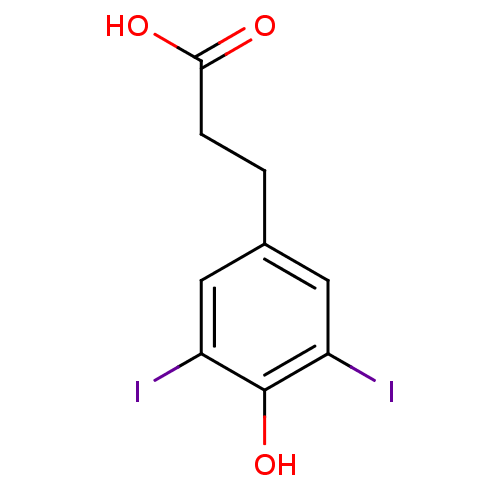
(3-(4-Hydroxy-3,5-diiodo-phenyl)-propionic acid | C...)Show InChI InChI=1S/C9H8I2O3/c10-6-3-5(1-2-8(12)13)4-7(11)9(6)14/h3-4,14H,1-2H2,(H,12,13) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145823
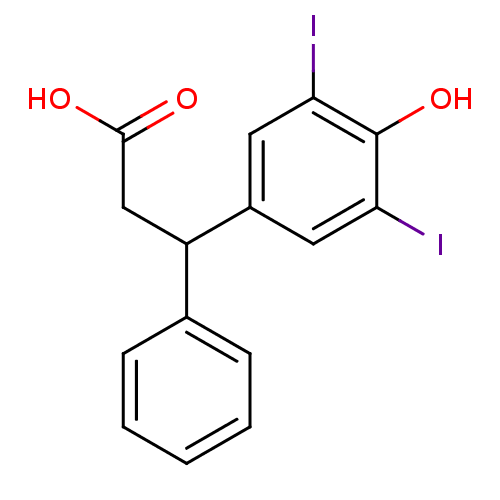
(3-(4-Hydroxy-3,5-diiodo-phenyl)-3-phenyl-propionic...)Show InChI InChI=1S/C15H12I2O3/c16-12-6-10(7-13(17)15(12)20)11(8-14(18)19)9-4-2-1-3-5-9/h1-7,11,20H,8H2,(H,18,19) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145804
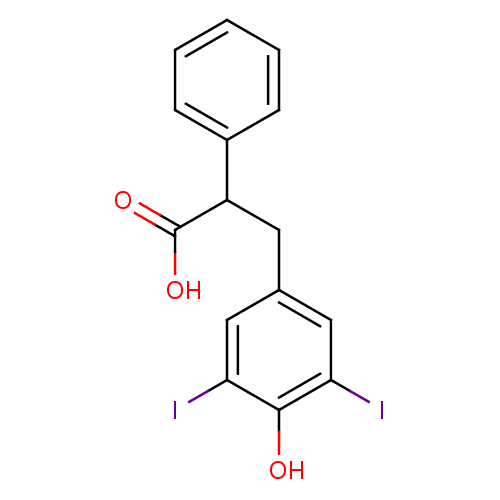
(3-(4-Hydroxy-3,5-diiodo-phenyl)-2-phenyl-propionic...)Show InChI InChI=1S/C15H12I2O3/c16-12-7-9(8-13(17)14(12)18)6-11(15(19)20)10-4-2-1-3-5-10/h1-5,7-8,11,18H,6H2,(H,19,20) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145815
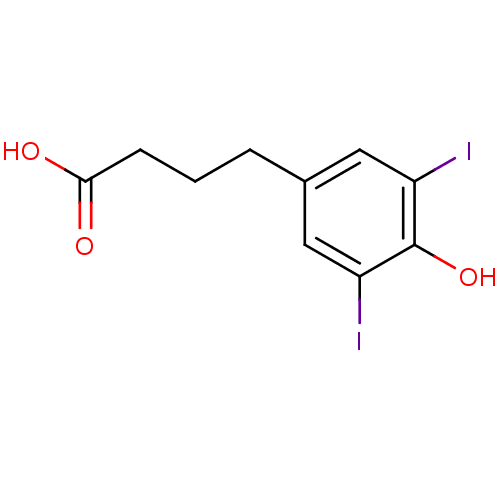
(4-(4-Hydroxy-3,5-diiodo-phenyl)-butyric acid | CHE...)Show InChI InChI=1S/C10H10I2O3/c11-7-4-6(2-1-3-9(13)14)5-8(12)10(7)15/h4-5,15H,1-3H2,(H,13,14) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145808
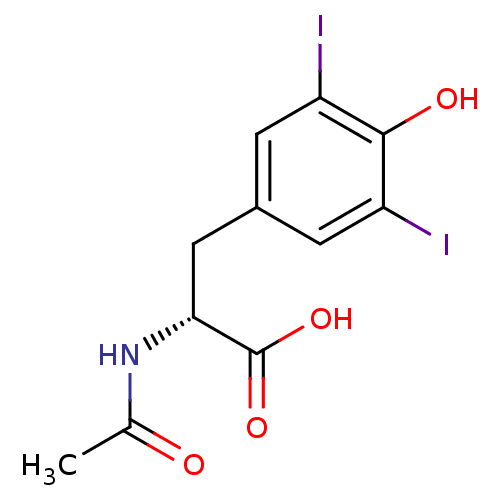
((R)-2-Acetylamino-3-(4-hydroxy-3,5-diiodo-phenyl)-...)Show InChI InChI=1S/C11H11I2NO4/c1-5(15)14-9(11(17)18)4-6-2-7(12)10(16)8(13)3-6/h2-3,9,16H,4H2,1H3,(H,14,15)(H,17,18)/t9-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145825
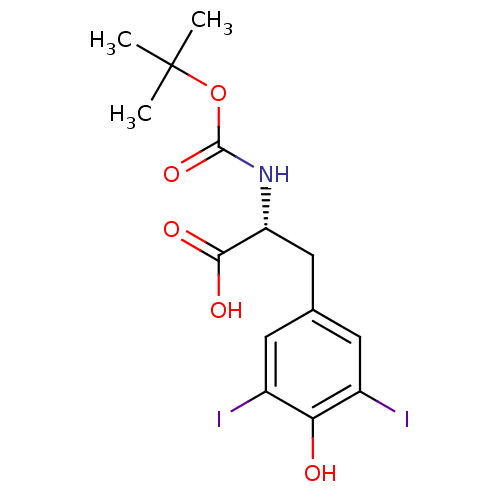
((R)-2-tert-Butoxycarbonylamino-3-(4-hydroxy-3,5-di...)Show SMILES CC(C)(C)OC(=O)N[C@H](Cc1cc(I)c(O)c(I)c1)C(O)=O Show InChI InChI=1S/C14H17I2NO5/c1-14(2,3)22-13(21)17-10(12(19)20)6-7-4-8(15)11(18)9(16)5-7/h4-5,10,18H,6H2,1-3H3,(H,17,21)(H,19,20)/t10-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145833

((S)-2-tert-Butoxycarbonylamino-3-(4-hydroxy-3,5-di...)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1cc(I)c(O)c(I)c1)C(O)=O Show InChI InChI=1S/C14H17I2NO5/c1-14(2,3)22-13(21)17-10(12(19)20)6-7-4-8(15)11(18)9(16)5-7/h4-5,10,18H,6H2,1-3H3,(H,17,21)(H,19,20)/t10-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145835

(4-Hydroxy-3,5-diiodo-benzoic acid | CHEMBL83650)Show InChI InChI=1S/C7H4I2O3/c8-4-1-3(7(11)12)2-5(9)6(4)10/h1-2,10H,(H,11,12) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 6.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50145827
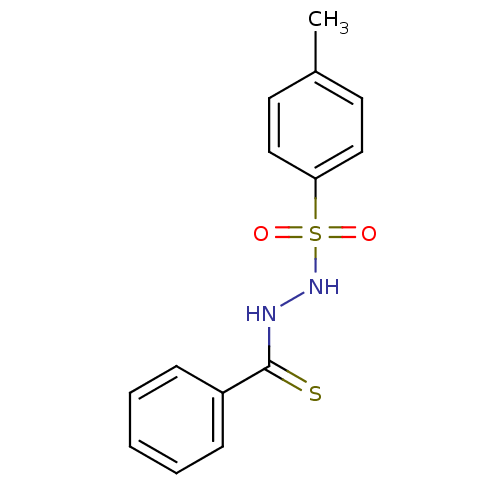
(CHEMBL311209 | N-[(4-methylbenzene)sulfonamido]ben...)Show InChI InChI=1S/C14H14N2O2S2/c1-11-7-9-13(10-8-11)20(17,18)16-15-14(19)12-5-3-2-4-6-12/h2-10,16H,1H3,(H,15,19) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition constant for HCV NS3 protease substrate binding site |
J Med Chem 47: 2486-98 (2004)
Article DOI: 10.1021/jm0305117
BindingDB Entry DOI: 10.7270/Q2FF3RSP |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604556
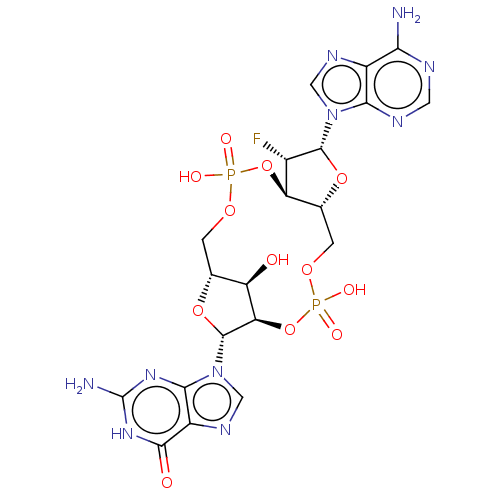
(CHEMBL5175703)Show SMILES N.N.[H][C@@]12COP(O)(=O)O[C@@]3([H])[C@H](F)[C@@H](O[C@]3([H])COP(O)(=O)O[C@]([H])([C@@H]1O)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O)n1cnc2c(N)ncnc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509399
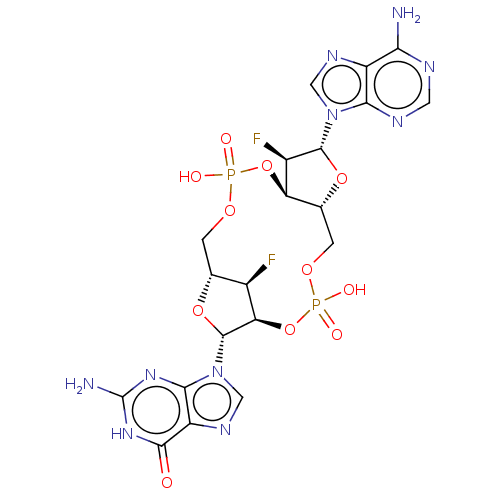
(CHEMBL4468010)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2COP(O)(=O)O[C@@H]3[C@@H](COP(O)(=O)O[C@@H]1[C@@H]2F)O[C@H]([C@@H]3F)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H22F2N10O11P2/c21-8-6-1-38-44(34,35)42-12-7(41-18(9(12)22)31-4-27-10-14(23)25-3-26-15(10)31)2-39-45(36,37)43-13(8)19(40-6)32-5-28-11-16(32)29-20(24)30-17(11)33/h3-9,12-13,18-19H,1-2H2,(H,34,35)(H,36,37)(H2,23,25,26)(H3,24,29,30,33)/t6-,7-,8-,9-,12-,13-,18-,19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509421
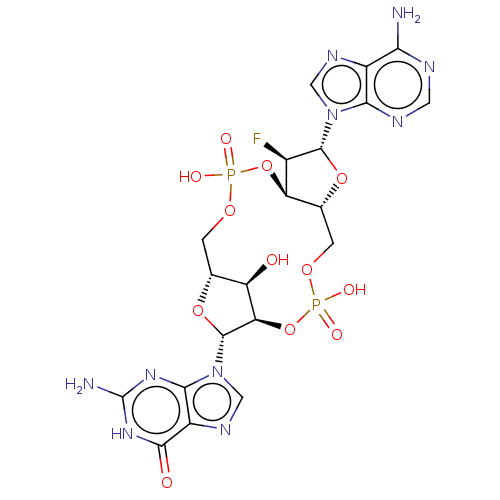
(CHEMBL4577528)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2COP(O)(=O)O[C@@H]3[C@@H](COP(O)(=O)O[C@@H]1[C@@H]2O)O[C@H]([C@@H]3F)n1cnc2c(N)ncnc12 Show InChI InChI=1S/C20H23FN10O12P2/c21-8-12-7(41-18(8)30-4-26-9-14(22)24-3-25-15(9)30)2-39-45(36,37)43-13-11(32)6(1-38-44(34,35)42-12)40-19(13)31-5-27-10-16(31)28-20(23)29-17(10)33/h3-8,11-13,18-19,32H,1-2H2,(H,34,35)(H,36,37)(H2,22,24,25)(H3,23,28,29,33)/t6-,7-,8-,11-,12-,13-,18-,19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Stimulator of interferon genes protein
(Human) | BDBM573971
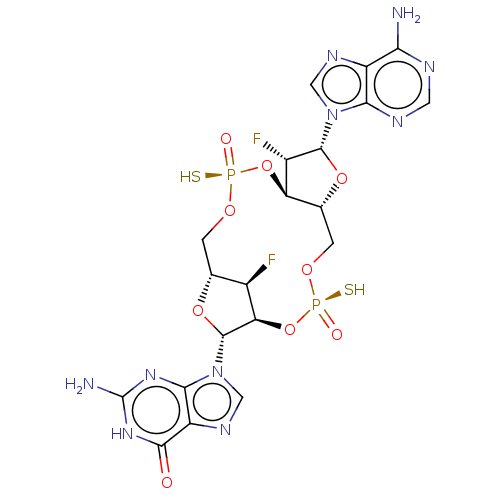
(US11453697, Example 247)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2CO[P@](S)(=O)O[C@@H]3[C@@H](CO[P@](S)(=O)O[C@@H]1[C@@H]2F)O[C@H]([C@H]3F)n1cnc2c(N)ncnc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509408
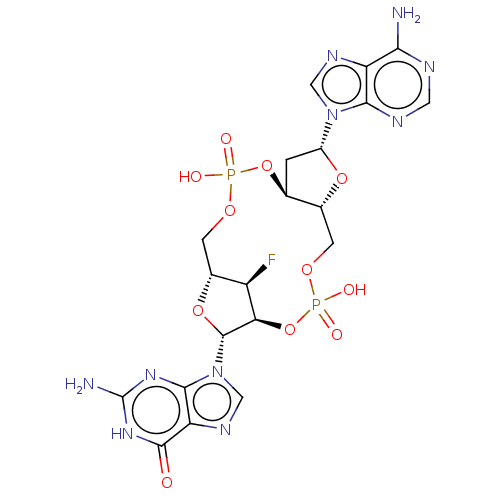
(CHEMBL4459563)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2COP(O)(=O)O[C@H]3C[C@@H](O[C@@H]3COP(O)(=O)O[C@@H]1[C@@H]2F)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H23FN10O11P2/c21-11-9-3-38-43(33,34)41-7-1-10(30-5-26-12-15(22)24-4-25-16(12)30)39-8(7)2-37-44(35,36)42-14(11)19(40-9)31-6-27-13-17(31)28-20(23)29-18(13)32/h4-11,14,19H,1-3H2,(H,33,34)(H,35,36)(H2,22,24,25)(H3,23,28,29,32)/t7-,8+,9+,10+,11+,14+,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509422
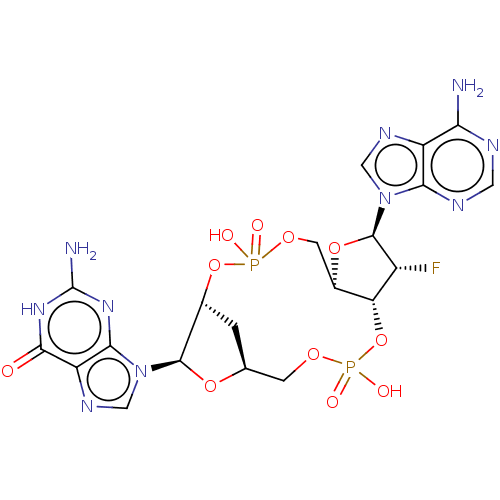
(CHEMBL4476079)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@H]2C[C@H]1OP(O)(=O)OC[C@H]1O[C@H]([C@H](F)[C@@H]1OP(O)(=O)OC2)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H23FN10O11P2/c21-10-13-9(40-19(10)30-5-26-11-14(22)24-4-25-15(11)30)3-38-43(33,34)41-8-1-7(2-37-44(35,36)42-13)39-18(8)31-6-27-12-16(31)28-20(23)29-17(12)32/h4-10,13,18-19H,1-3H2,(H,33,34)(H,35,36)(H2,22,24,25)(H3,23,28,29,32)/t7-,8+,9+,10+,13+,18+,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM573856
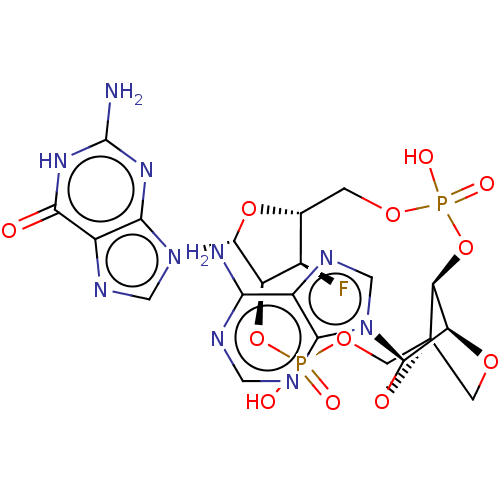
(US11453697, Example 20)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2COP(O)(=O)O[C@H]3[C@H]4OC[C@]3(COP(O)(=O)O[C@@H]1[C@@H]2F)O[C@H]4n1cnc2c(N)ncnc12 |r,TLB:19:20:35.34:23.22| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509431
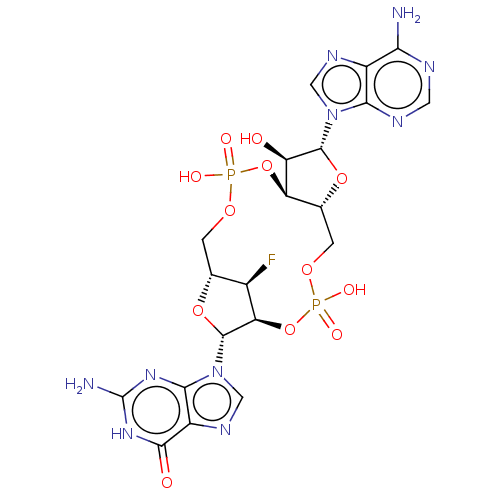
(CHEMBL4589856)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2COP(O)(=O)O[C@@H]3[C@@H](COP(O)(=O)O[C@@H]1[C@@H]2F)O[C@H]([C@@H]3O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H23FN10O12P2/c21-8-6-1-38-44(34,35)42-12-7(41-18(11(12)32)30-4-26-9-14(22)24-3-25-15(9)30)2-39-45(36,37)43-13(8)19(40-6)31-5-27-10-16(31)28-20(23)29-17(10)33/h3-8,11-13,18-19,32H,1-2H2,(H,34,35)(H,36,37)(H2,22,24,25)(H3,23,28,29,33)/t6-,7-,8-,11-,12-,13-,18-,19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604561
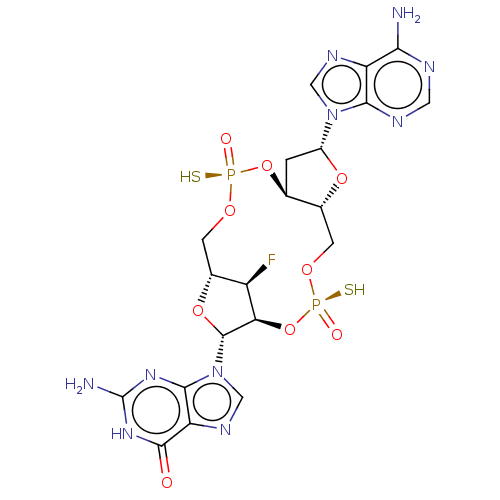
(CHEMBL5197904)Show SMILES N.N.[H][C@@]12CO[P@](S)(=O)O[C@@]3([H])C[C@@H](O[C@]3([H])CO[P@](S)(=O)O[C@]([H])([C@@H]1F)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O)n1cnc2c(N)ncnc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50506263
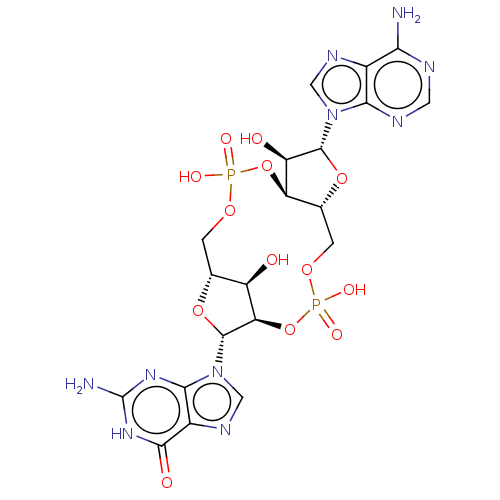
(CHEMBL4465054 | US11401295, Compound 2',3'-cGAMP)Show SMILES [H][C@]12COP(O)(=O)O[C@@]3([H])[C@@H](O)[C@@H](O[C@]3([H])COP(O)(=O)O[C@@]([H])([C@@H](O1)n1cnc3c1nc(N)[nH]c3=O)[C@@H]2O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H24N10O13P2/c21-14-8-15(24-3-23-14)29(4-25-8)18-11(32)12-7(41-18)2-39-45(36,37)43-13-10(31)6(1-38-44(34,35)42-12)40-19(13)30-5-26-9-16(30)27-20(22)28-17(9)33/h3-7,10-13,18-19,31-32H,1-2H2,(H,34,35)(H,36,37)(H2,21,23,24)(H3,22,27,28,33)/t6-,7-,10-,11-,12-,13-,18-,19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604563
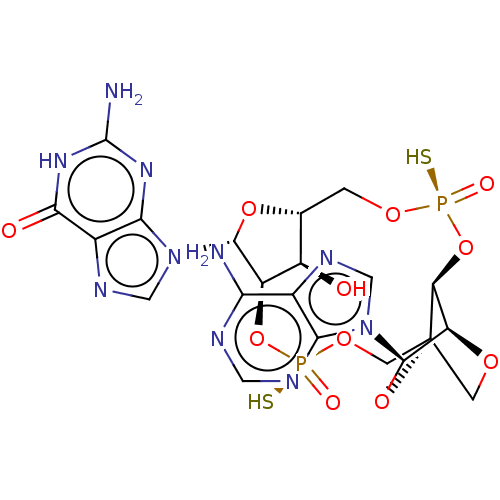
(CHEMBL5201164)Show SMILES N.N.[H][C@@]12CO[P@](S)(=O)O[C@@]3([H])[C@@]4([H])OC[C@]3(CO[P@](S)(=O)O[C@]([H])([C@@H]1O)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O)O[C@H]4n1cnc2c(N)ncnc12 |r,TLB:9:10:40.41:14.15| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604560
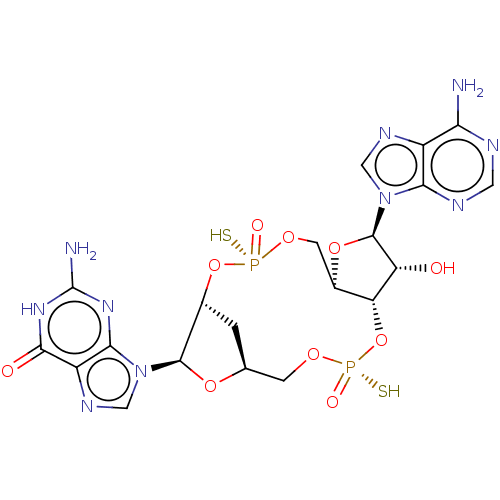
(CHEMBL5180005)Show SMILES N.N.[H][C@]12C[C@@]([H])(O[P@@](S)(=O)OC[C@@]3([H])O[C@H]([C@H](O)[C@]3([H])O[P@@](S)(=O)OC1)n1cnc3c(N)ncnc13)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604555
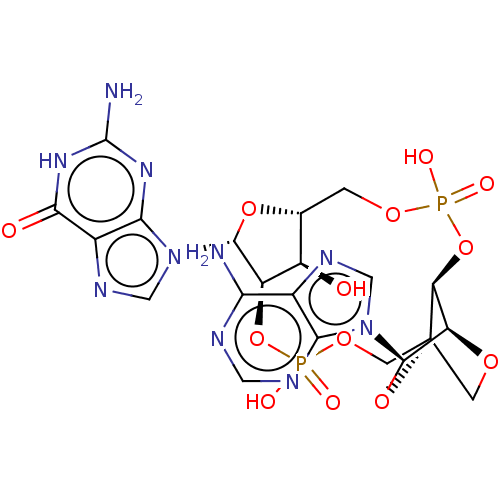
(CHEMBL5181746)Show SMILES N.N.[H][C@@]12COP(O)(=O)O[C@@]3([H])[C@@]4([H])OC[C@]3(COP(O)(=O)O[C@]([H])([C@@H]1O)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O)O[C@H]4n1cnc2c(N)ncnc12 |r,TLB:9:10:40.41:14.15| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604562
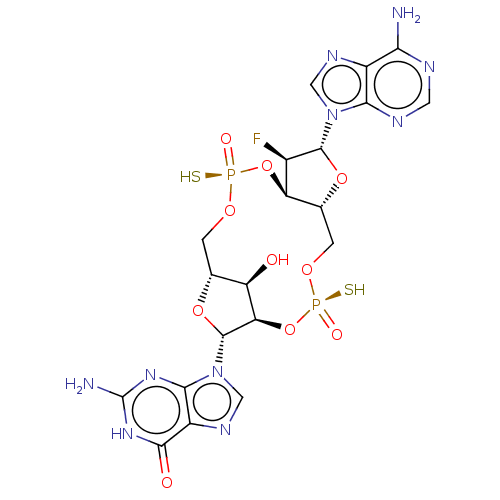
(CHEMBL5185127)Show SMILES N.N.[H][C@@]12CO[P@](S)(=O)O[C@@]3([H])[C@@H](F)[C@@H](O[C@]3([H])CO[P@](S)(=O)O[C@]([H])([C@@H]1O)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O)n1cnc2c(N)ncnc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509412
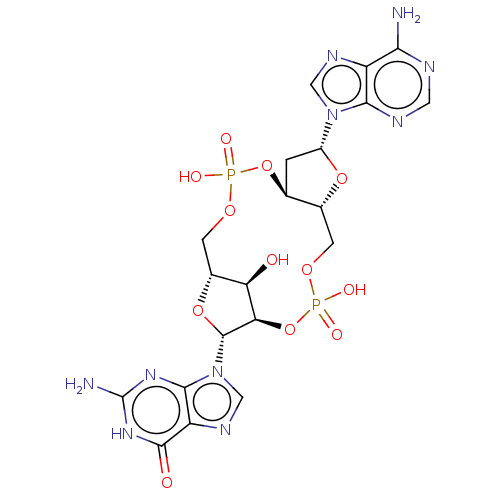
(CHEMBL4474321 | US11453697, Example 1)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@@H]2COP(O)(=O)O[C@H]3C[C@@H](O[C@@H]3COP(O)(=O)O[C@@H]1[C@@H]2O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H24N10O12P2/c21-15-11-16(24-4-23-15)29(5-25-11)10-1-7-8(39-10)2-37-44(35,36)42-14-13(31)9(3-38-43(33,34)41-7)40-19(14)30-6-26-12-17(30)27-20(22)28-18(12)32/h4-10,13-14,19,31H,1-3H2,(H,33,34)(H,35,36)(H2,21,23,24)(H3,22,27,28,32)/t7-,8+,9+,10+,13+,14+,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604559
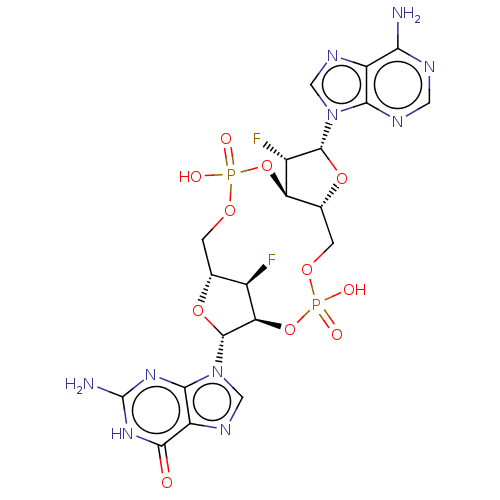
(CHEMBL5174023)Show SMILES N.N.[H][C@@]12COP(O)(=O)O[C@@]3([H])[C@H](F)[C@@H](O[C@]3([H])COP(O)(=O)O[C@]([H])([C@@H]1F)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O)n1cnc2c(N)ncnc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604557
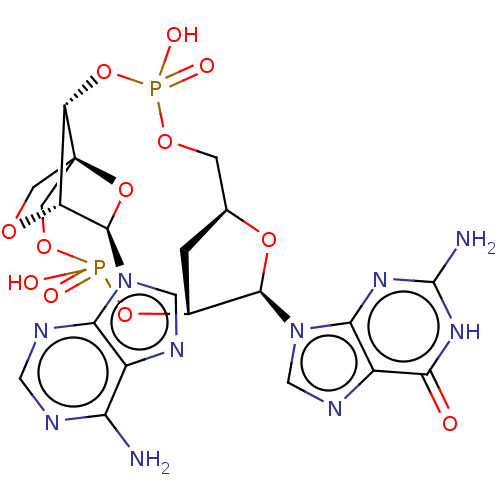
(CHEMBL5191008)Show SMILES N.N.[H][C@]12C[C@@]([H])(OP(O)(=O)OC[C@@]34CO[C@@]([H])([C@@H](O3)n3cnc5c(N)ncnc35)[C@]4([H])OP(O)(=O)OC1)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O |r,TLB:32:30:14.15:18.19| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509430
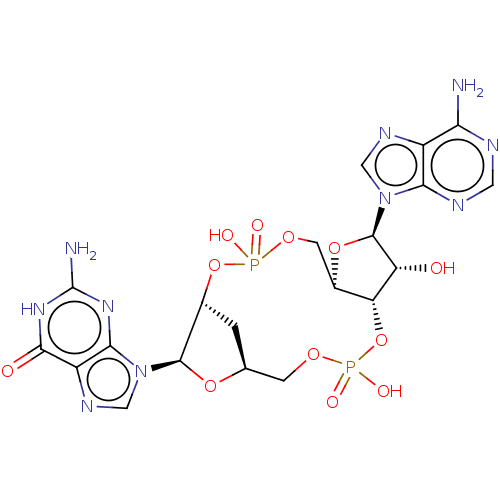
(CHEMBL4570468)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@H]2C[C@H]1OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(=O)OC2)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H24N10O12P2/c21-14-10-15(24-4-23-14)29(5-25-10)19-12(31)13-9(40-19)3-38-43(33,34)41-8-1-7(2-37-44(35,36)42-13)39-18(8)30-6-26-11-16(30)27-20(22)28-17(11)32/h4-9,12-13,18-19,31H,1-3H2,(H,33,34)(H,35,36)(H2,21,23,24)(H3,22,27,28,32)/t7-,8+,9+,12+,13+,18+,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50509425
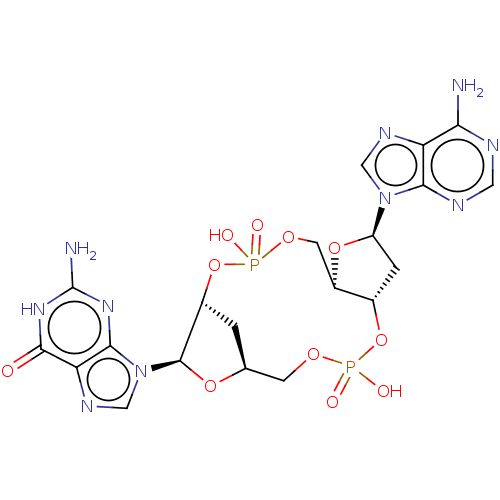
(CHEMBL4566174 | US11453697, Example 27)Show SMILES Nc1nc2n(cnc2c(=O)[nH]1)[C@@H]1O[C@H]2C[C@H]1OP(O)(=O)OC[C@H]1O[C@H](C[C@@H]1OP(O)(=O)OC2)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C20H24N10O11P2/c21-15-13-16(24-5-23-15)29(6-25-13)12-2-9-11(39-12)4-37-43(34,35)41-10-1-8(3-36-42(32,33)40-9)38-19(10)30-7-26-14-17(30)27-20(22)28-18(14)31/h5-12,19H,1-4H2,(H,32,33)(H,34,35)(H2,21,23,24)(H3,22,27,28,31)/t8-,9-,10+,11+,12+,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50604558

(CHEMBL5205731)Show SMILES N.N.[H][C@]12C[C@@]([H])(OP(O)(=O)OC[C@@]3([H])O[C@H]([C@@H](F)[C@]3([H])OP(O)(=O)OC1)n1cnc3c(N)ncnc13)[C@@H](O2)n1cnc2c1nc(N)[nH]c2=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300237

((R)-5-(2-Cyclohexylethyl)-5-(cyclohexylmethyl)-3-[...)Show SMILES CCC[C@H]1CN(Cc2ccc(CN3C(N)=NC(CCC4CCCCC4)(CC4CCCCC4)C3=O)cc2)C(=O)N1 |r,c:14| Show InChI InChI=1S/C32H49N5O2/c1-2-9-28-23-36(31(39)34-28)21-26-14-16-27(17-15-26)22-37-29(38)32(35-30(37)33,20-25-12-7-4-8-13-25)19-18-24-10-5-3-6-11-24/h14-17,24-25,28H,2-13,18-23H2,1H3,(H2,33,35)(H,34,39)/t28-,32?/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated human Swedish amyloid precursor protein peptide hydrolysis by HTRF assay |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
Stimulator of interferon genes protein
(Human) | BDBM50582513
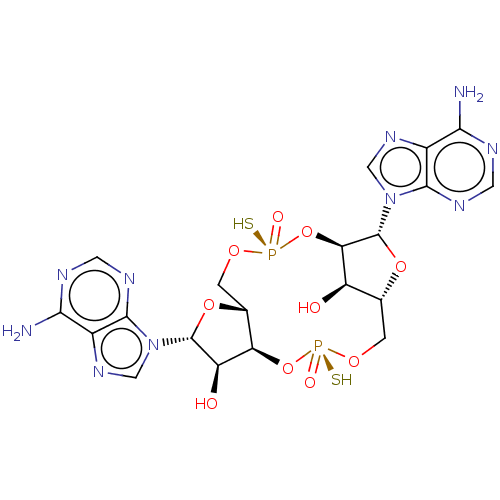
(CHEMBL5093161)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@@H]2CO[P@](S)(=O)O[C@@H]3[C@H](O)[C@@H](CO[P@](S)(=O)O[C@H]2[C@H]1O)O[C@H]3n1cnc2c(N)ncnc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c02197
BindingDB Entry DOI: 10.7270/Q2028WM5 |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380310
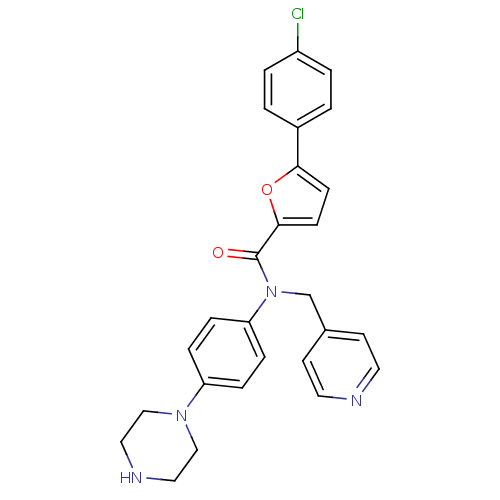
(CHEMBL2017619)Show SMILES Clc1ccc(cc1)-c1ccc(o1)C(=O)N(Cc1ccncc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C27H25ClN4O2/c28-22-3-1-21(2-4-22)25-9-10-26(34-25)27(33)32(19-20-11-13-29-14-12-20)24-7-5-23(6-8-24)31-17-15-30-16-18-31/h1-14,30H,15-19H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380309
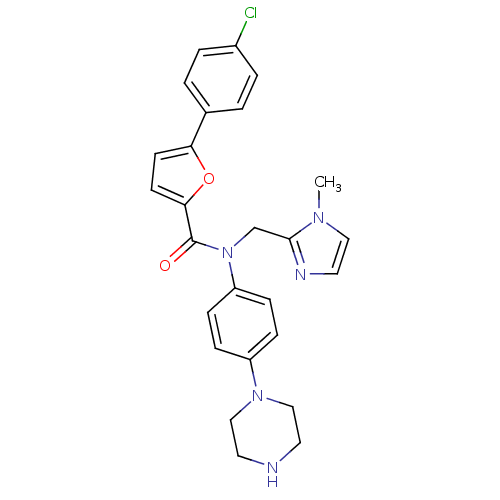
(CHEMBL2017463)Show SMILES Cn1ccnc1CN(C(=O)c1ccc(o1)-c1ccc(Cl)cc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C26H26ClN5O2/c1-30-15-14-29-25(30)18-32(22-8-6-21(7-9-22)31-16-12-28-13-17-31)26(33)24-11-10-23(34-24)19-2-4-20(27)5-3-19/h2-11,14-15,28H,12-13,16-18H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50362106
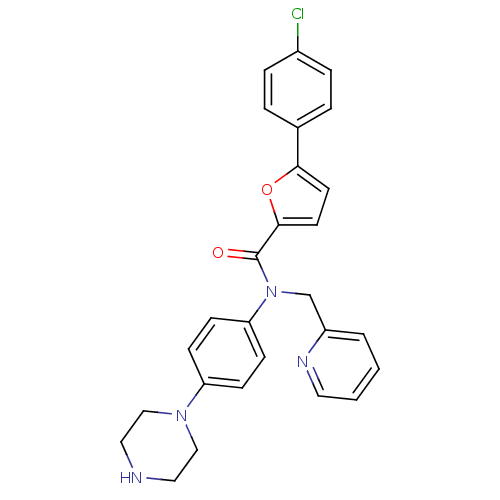
(CHEMBL1938681)Show SMILES Clc1ccc(cc1)-c1ccc(o1)C(=O)N(Cc1ccccn1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C27H25ClN4O2/c28-21-6-4-20(5-7-21)25-12-13-26(34-25)27(33)32(19-22-3-1-2-14-30-22)24-10-8-23(9-11-24)31-17-15-29-16-18-31/h1-14,29H,15-19H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380305
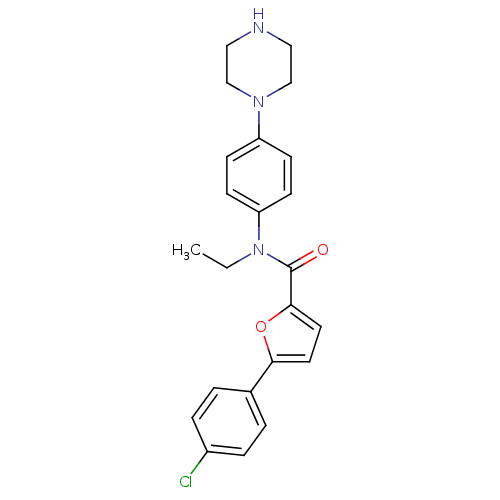
(CHEMBL2017459)Show SMILES CCN(C(=O)c1ccc(o1)-c1ccc(Cl)cc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C23H24ClN3O2/c1-2-27(20-9-7-19(8-10-20)26-15-13-25-14-16-26)23(28)22-12-11-21(29-22)17-3-5-18(24)6-4-17/h3-12,25H,2,13-16H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300236
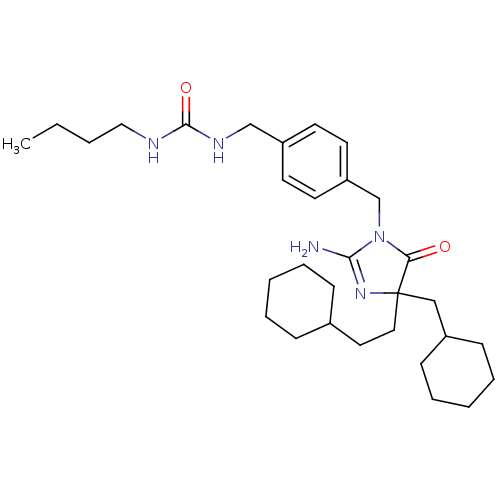
(CHEMBL568942 | rac-3-[(3-Butylureido)methyl]benzyl...)Show SMILES CCCCNC(=O)NCc1ccc(CN2C(N)=NC(CCC3CCCCC3)(CC3CCCCC3)C2=O)cc1 |c:16| Show InChI InChI=1S/C31H49N5O2/c1-2-3-20-33-30(38)34-22-26-14-16-27(17-15-26)23-36-28(37)31(35-29(36)32,21-25-12-8-5-9-13-25)19-18-24-10-6-4-7-11-24/h14-17,24-25H,2-13,18-23H2,1H3,(H2,32,35)(H2,33,34,38) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated human Swedish amyloid precursor protein peptide hydrolysis by HTRF assay |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380308
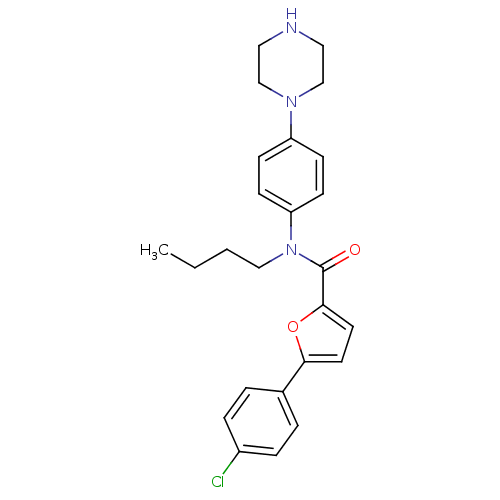
(CHEMBL2017462)Show SMILES CCCCN(C(=O)c1ccc(o1)-c1ccc(Cl)cc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C25H28ClN3O2/c1-2-3-16-29(22-10-8-21(9-11-22)28-17-14-27-15-18-28)25(30)24-13-12-23(31-24)19-4-6-20(26)7-5-19/h4-13,27H,2-3,14-18H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380304
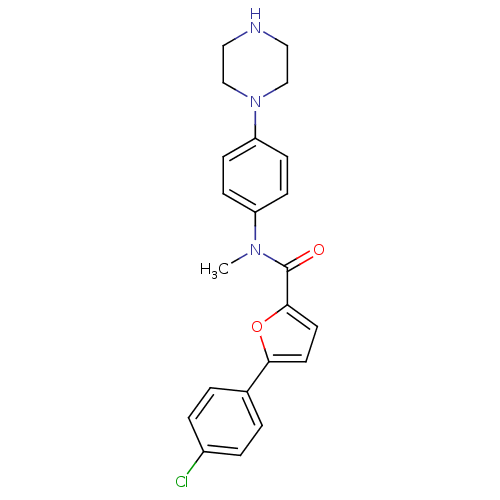
(CHEMBL2017458)Show SMILES CN(C(=O)c1ccc(o1)-c1ccc(Cl)cc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C22H22ClN3O2/c1-25(18-6-8-19(9-7-18)26-14-12-24-13-15-26)22(27)21-11-10-20(28-21)16-2-4-17(23)5-3-16/h2-11,24H,12-15H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300228

((2E,5R)-5-(2-cyclohexylethyl)-5-(cyclohexylmethyl)...)Show SMILES CN1C(N)=N[C@](CCC2CCCCC2)(CC2CCCCC2)C1=O |r,c:3| Show InChI InChI=1S/C19H33N3O/c1-22-17(23)19(21-18(22)20,14-16-10-6-3-7-11-16)13-12-15-8-4-2-5-9-15/h15-16H,2-14H2,1H3,(H2,20,21)/t19-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 605 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated human Swedish amyloid precursor protein peptide hydrolysis by HTRF assay |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380306
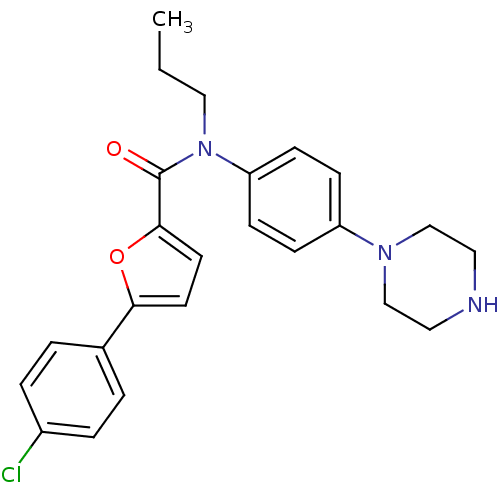
(CHEMBL2017460)Show SMILES CCCN(C(=O)c1ccc(o1)-c1ccc(Cl)cc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C24H26ClN3O2/c1-2-15-28(21-9-7-20(8-10-21)27-16-13-26-14-17-27)24(29)23-12-11-22(30-23)18-3-5-19(25)6-4-18/h3-12,26H,2,13-17H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380307
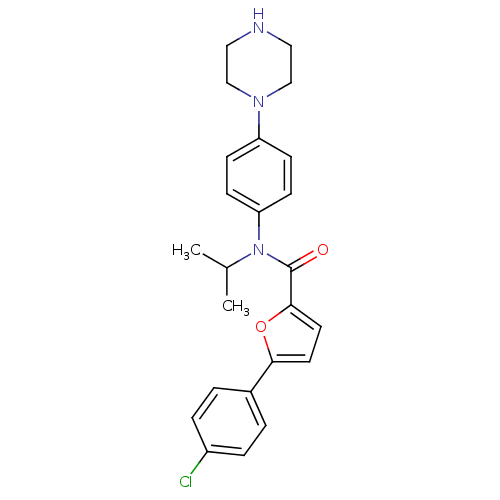
(CHEMBL2017461)Show SMILES CC(C)N(C(=O)c1ccc(o1)-c1ccc(Cl)cc1)c1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C24H26ClN3O2/c1-17(2)28(21-9-7-20(8-10-21)27-15-13-26-14-16-27)24(29)23-12-11-22(30-23)18-3-5-19(25)6-4-18/h3-12,17,26H,13-16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300233
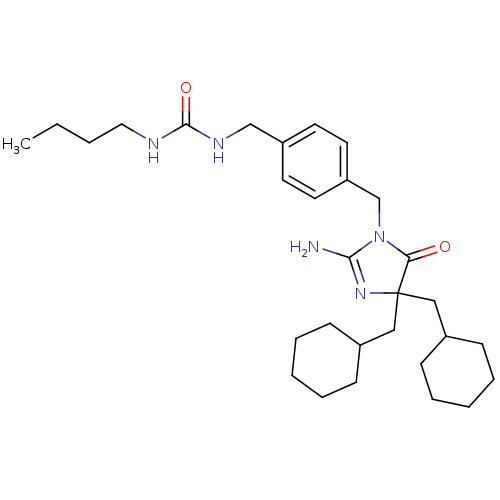
(CHEMBL571249 | rac-3-[(3-Butylureido)methyl]benzyl...)Show SMILES CCCCNC(=O)NCc1ccc(CN2C(N)=NC(CC3CCCCC3)(CC3CCCCC3)C2=O)cc1 |c:16| Show InChI InChI=1S/C30H47N5O2/c1-2-3-18-32-29(37)33-21-25-14-16-26(17-15-25)22-35-27(36)30(34-28(35)31,19-23-10-6-4-7-11-23)20-24-12-8-5-9-13-24/h14-17,23-24H,2-13,18-22H2,1H3,(H2,31,34)(H2,32,33,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated human Swedish amyloid precursor protein peptide hydrolysis by HTRF assay |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50380298
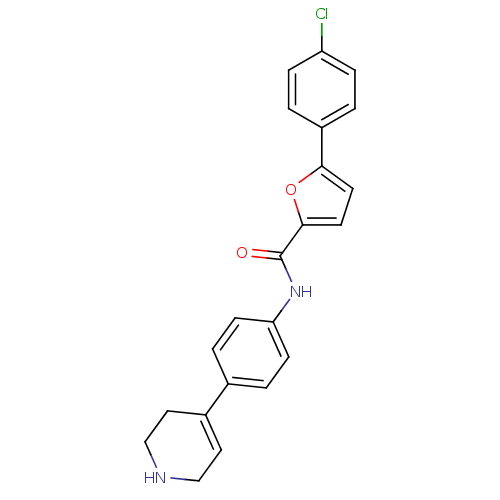
(CHEMBL2017452)Show SMILES Clc1ccc(cc1)-c1ccc(o1)C(=O)Nc1ccc(cc1)C1=CCNCC1 |t:24| Show InChI InChI=1S/C22H19ClN2O2/c23-18-5-1-17(2-6-18)20-9-10-21(27-20)22(26)25-19-7-3-15(4-8-19)16-11-13-24-14-12-16/h1-11,24H,12-14H2,(H,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 100 uM ATP by DELFIA... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300228

((2E,5R)-5-(2-cyclohexylethyl)-5-(cyclohexylmethyl)...)Show SMILES CN1C(N)=N[C@](CCC2CCCCC2)(CC2CCCCC2)C1=O |r,c:3| Show InChI InChI=1S/C19H33N3O/c1-22-17(23)19(21-18(22)20,14-16-10-6-3-7-11-16)13-12-15-8-4-2-5-9-15/h15-16H,2-14H2,1H3,(H2,20,21)/t19-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated hydrolysis of human amyloid precursor protein with Swedish and London mutation in HEK293 cells by ELISA |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300235
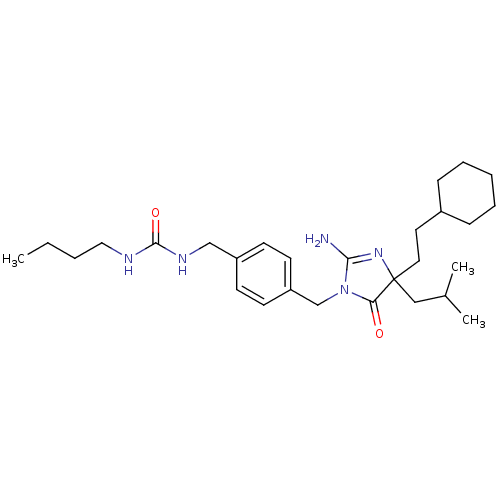
(CHEMBL571728 | rac-3-[(3-Butyl-ureidomethyl)benzyl...)Show SMILES CCCCNC(=O)NCc1ccc(CN2C(N)=NC(CCC3CCCCC3)(CC(C)C)C2=O)cc1 |c:16| Show InChI InChI=1S/C28H45N5O2/c1-4-5-17-30-27(35)31-19-23-11-13-24(14-12-23)20-33-25(34)28(18-21(2)3,32-26(33)29)16-15-22-9-7-6-8-10-22/h11-14,21-22H,4-10,15-20H2,1-3H3,(H2,29,32)(H2,30,31,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated human Swedish amyloid precursor protein peptide hydrolysis by HTRF assay |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50300232

(CHEMBL568968 | rac-3-[(3-Butylureidomethyl)benzyl]...)Show SMILES CCCCNC(=O)NCc1ccc(CN2C(N)=NC(C)(CC3CCCCC3)C2=O)cc1 |c:16| Show InChI InChI=1S/C24H37N5O2/c1-3-4-14-26-23(31)27-16-19-10-12-20(13-11-19)17-29-21(30)24(2,28-22(29)25)15-18-8-6-5-7-9-18/h10-13,18H,3-9,14-17H2,1-2H3,(H2,25,28)(H2,26,27,31) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 mediated human Swedish amyloid precursor protein peptide hydrolysis by HTRF assay |
J Med Chem 53: 951-65 (2010)
Article DOI: 10.1021/jm901408p
BindingDB Entry DOI: 10.7270/Q26W9B4S |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50362169
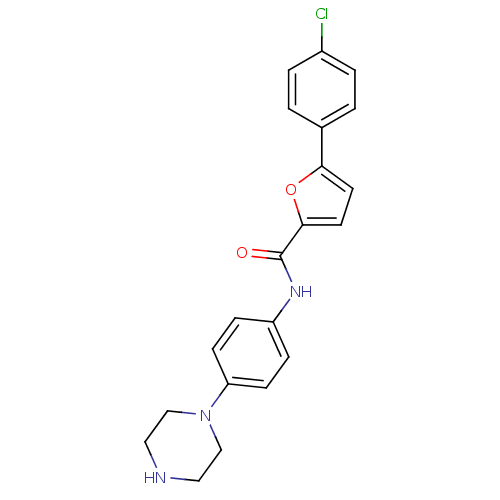
(CHEMBL1938680)Show SMILES Clc1ccc(cc1)-c1ccc(o1)C(=O)Nc1ccc(cc1)N1CCNCC1 Show InChI InChI=1S/C21H20ClN3O2/c22-16-3-1-15(2-4-16)19-9-10-20(27-19)21(26)24-17-5-7-18(8-6-17)25-13-11-23-12-14-25/h1-10,23H,11-14H2,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MK2 using Acam peptide as substrate incubated for 30 mins prior to substrate addition measured after 10 mins using 2 uM ATP by DELFIA a... |
ACS Med Chem Lett 2: 632-637 (2011)
Article DOI: 10.1021/ml200113y
BindingDB Entry DOI: 10.7270/Q2J967CS |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data