

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50085044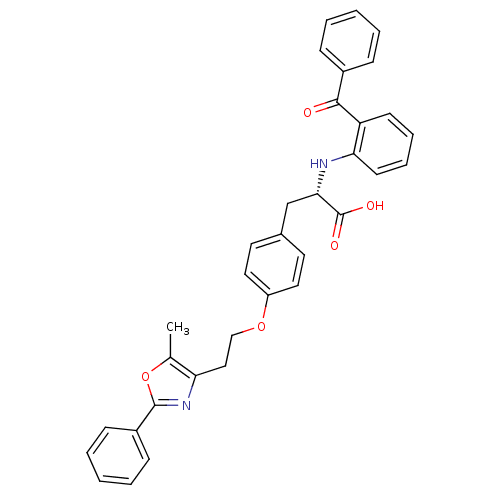 ((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB PubMed | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50085046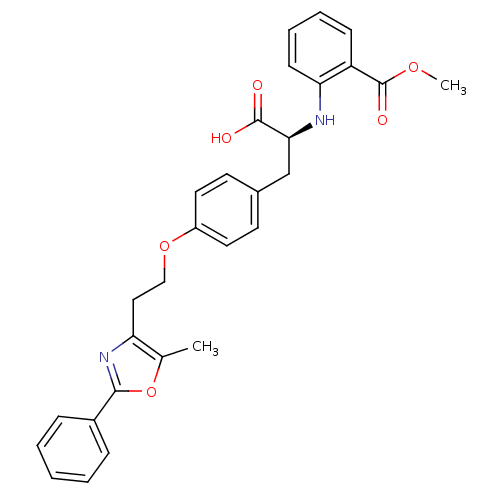 (2-((S)-1-carboxy-2-{4-[2-(5-methyl-2-phenyl-oxazol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826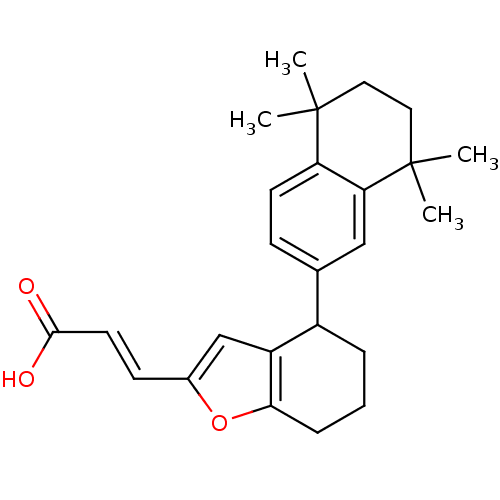 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143825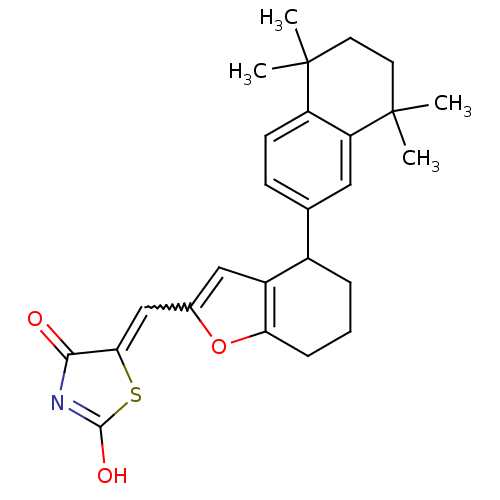 (5-[1-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143827 ((E)-3-[4-(3-Methoxy-5,5,8,8-tetramethyl-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143832 ((E)-3-[4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143833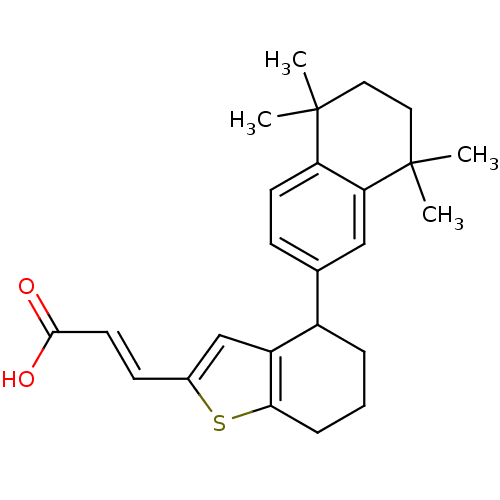 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM34017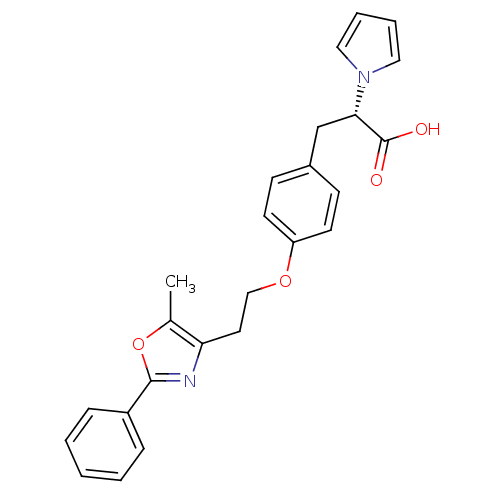 (CHEMBL104850 | phenylpropanoic acid derivative, 7) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM34017 (CHEMBL104850 | phenylpropanoic acid derivative, 7) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Maximal reporter activity against human Peroxisome proliferator activated receptor gamma Gal4 chimeric in transiently transfected CV-1 cells by funct... | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106960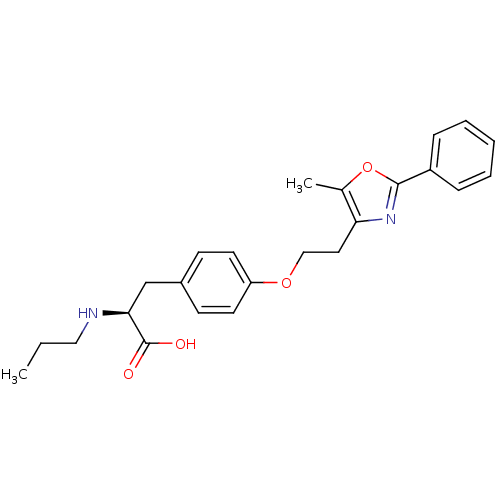 ((S)-3-{4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50534427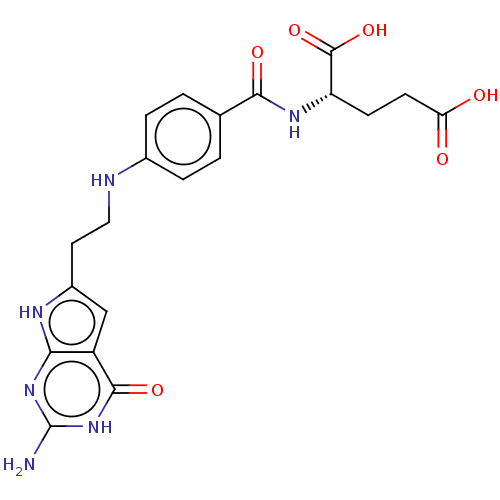 (CHEMBL4437824) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 59 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50534431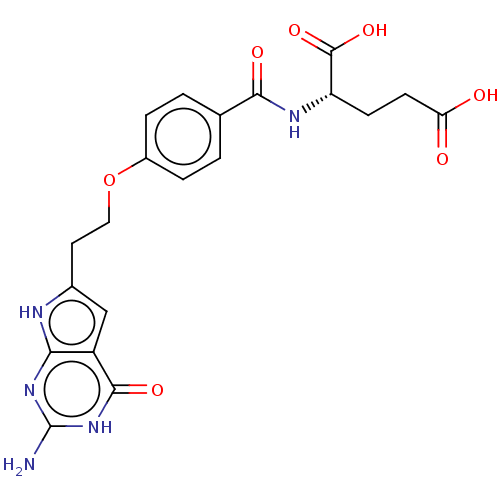 (CHEMBL4447805) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 61 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50534429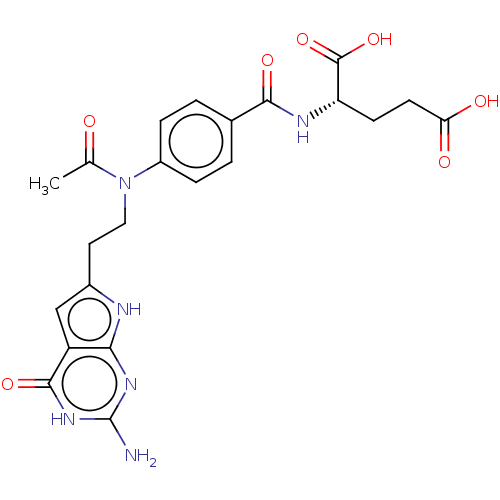 (CHEMBL4444011) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50409928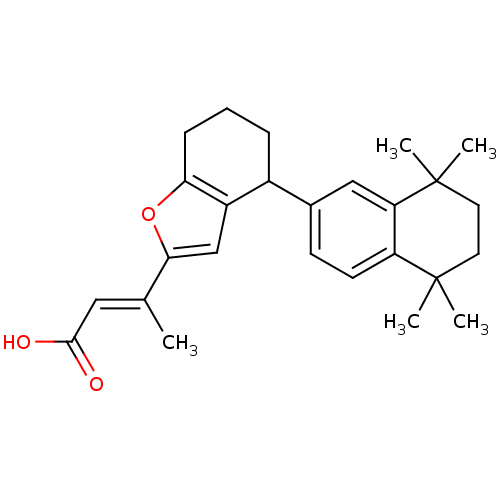 (CHEMBL2113737) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50354833 (AGF94 | CHEMBL1834488) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50106960 ((S)-3-{4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 72 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50409929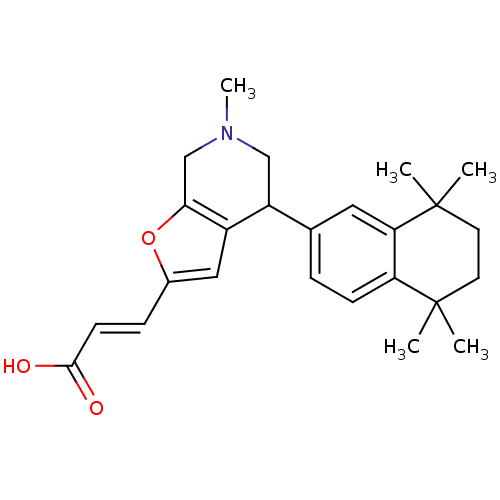 (CHEMBL2113736) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143826 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143824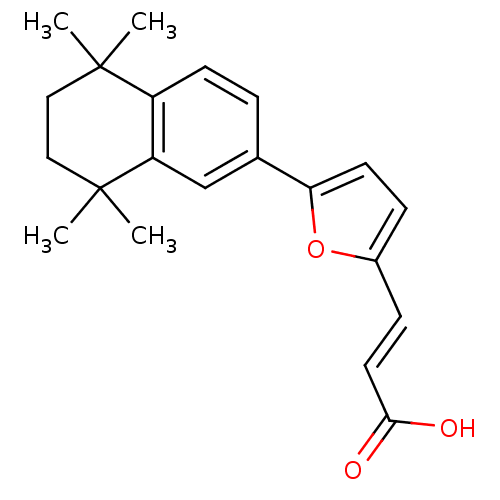 ((E)-3-[5-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proton-coupled folate transporter (Homo sapiens (Human)) | BDBM50027656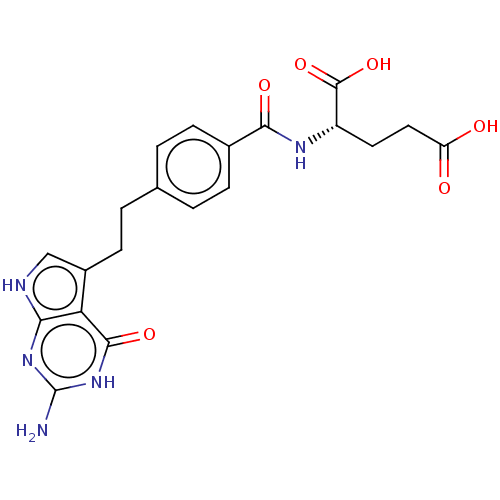 (CHEBI:63616 | LY-2315 | LY-231514 | PEMETREXED | U...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of [3H]MTX uptake at human PCFT expressed in Chinese hamster R2/PCFT4 cells at pH 5.5 measured after 5 mins by Dixon plot analysis | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50534430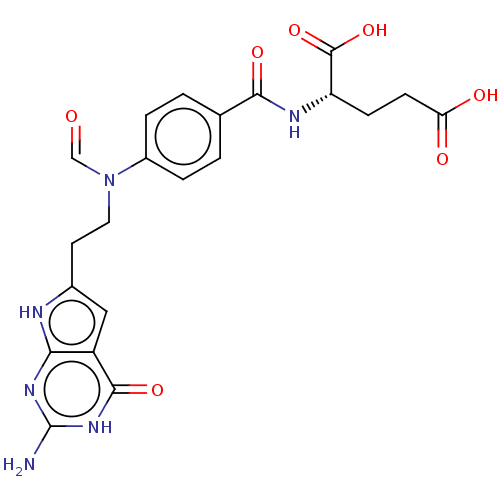 (CHEMBL4471269) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 99 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143821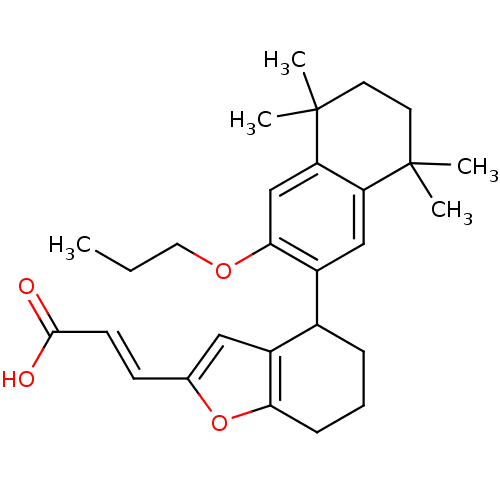 ((E)-3-[4-(5,5,8,8-Tetramethyl-3-propoxy-5,6,7,8-te...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM28681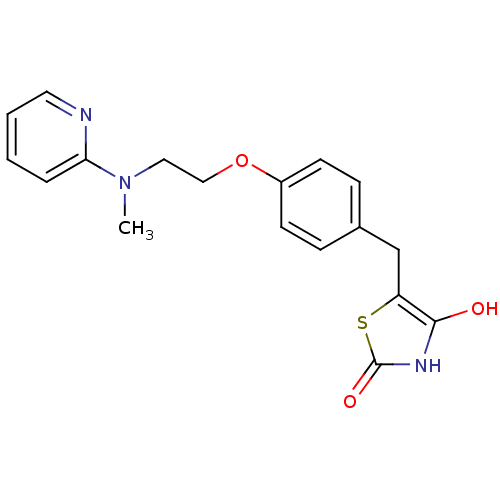 (5-[(4-{2-[methyl(pyridin-2-yl)amino]ethoxy}phenyl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50534426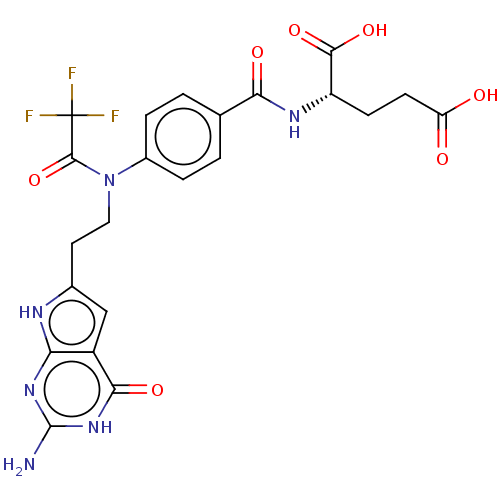 (CHEMBL4553188) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 122 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143835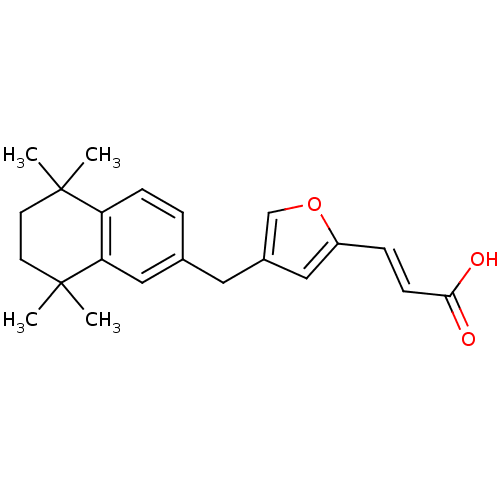 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 138 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106967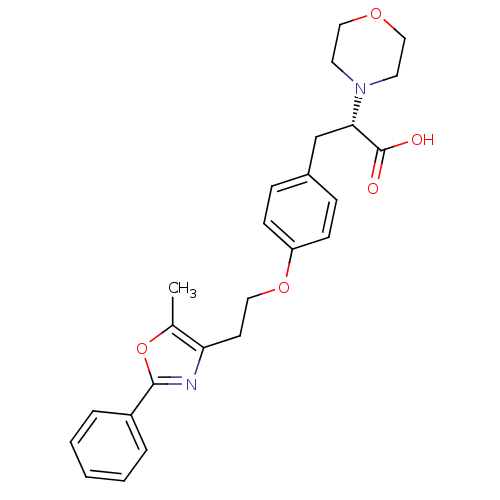 ((S)-3-{4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143823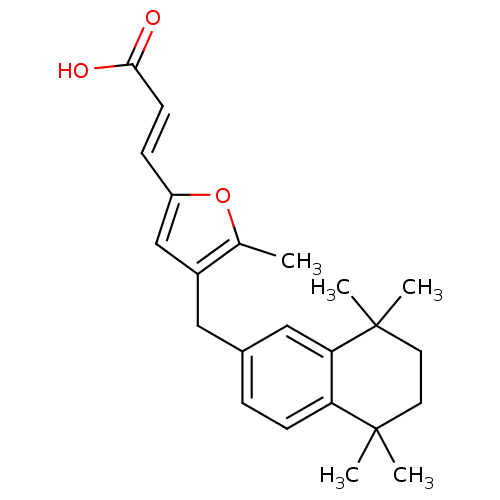 ((E)-3-[5-Methyl-4-(5,5,8,8-tetramethyl-5,6,7,8-tet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143831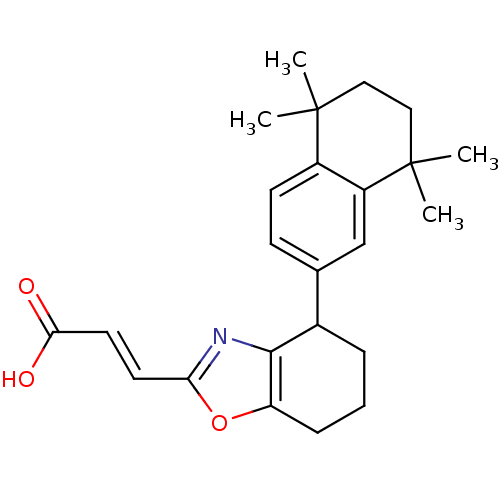 ((E)-3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-n...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50171513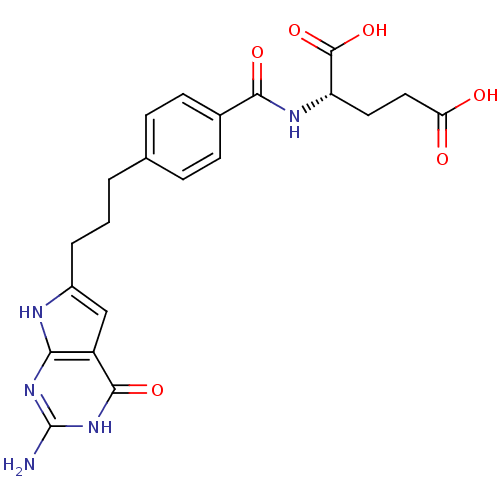 ((S)-2-(4-(3-(2-amino-4-oxo-4,7-dihydro-3H-pyrrolo[...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50143828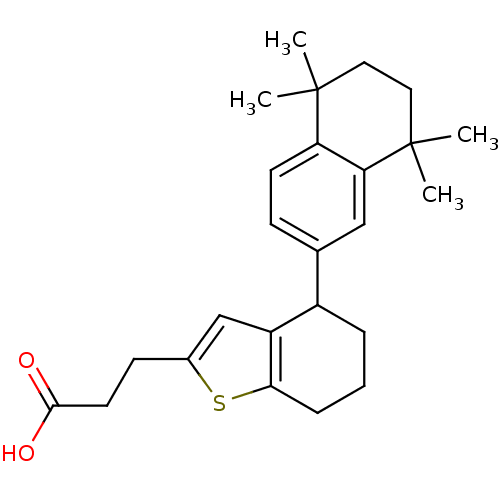 (3-[4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-napht...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoid X receptor alpha (RXRalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50534428 (CHEMBL4467936) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 201 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proton-coupled folate transporter (Homo sapiens (Human)) | BDBM50171513 ((S)-2-(4-(3-(2-amino-4-oxo-4,7-dihydro-3H-pyrrolo[...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of [3H]MTX uptake at human PCFT expressed in Chinese hamster R2/PCFT4 cells at pH 5.5 measured after 5 mins by Dixon plot analysis | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106965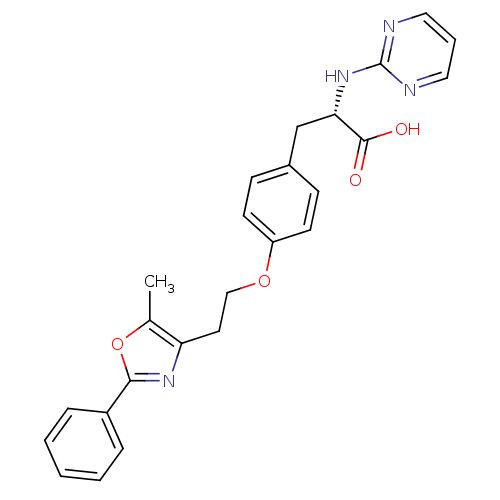 ((S)-3-{4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106964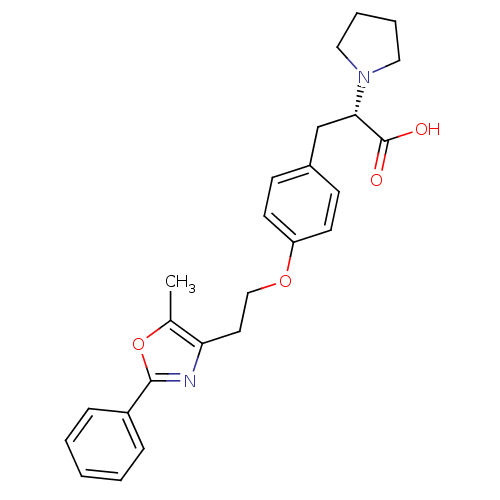 ((S)-3-{4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106966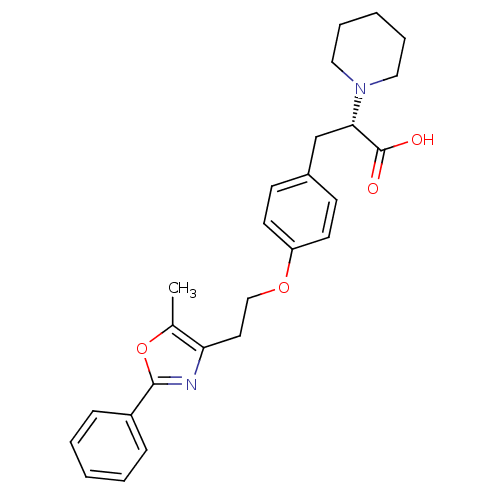 ((S)-3-{4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor alpha (Homo sapiens (Human)) | BDBM50085044 ((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Maximal reporter activity against human Peroxisome proliferator activated receptor alpha Gal4 chimeric in transiently transfected CV-1 cells by funct... | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106963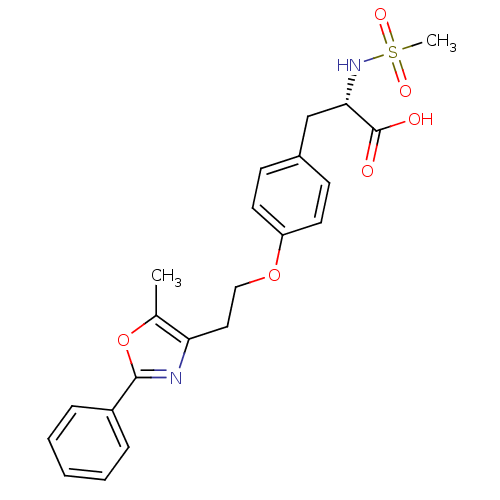 ((S)-2-Methanesulfonylamino-3-{4-[2-(5-methyl-2-phe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Maximal reporter activity against human Peroxisome proliferator activated receptor gamma Gal4 chimeric in transiently transfected CV-1 cells by funct... | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50049240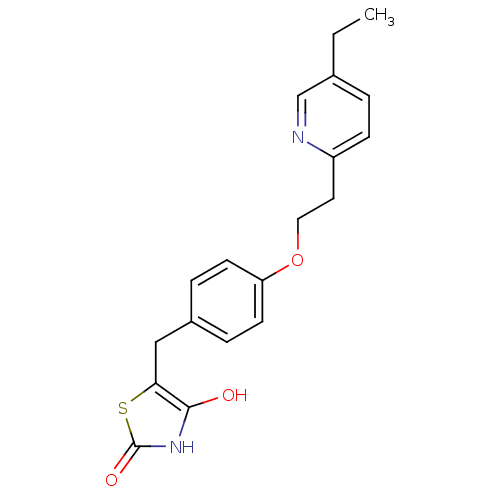 ((+-)-5-((4-(2-(5-ethyl-2-pyridinyl)ethoxy)phenyl)m...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Maximal reporter activity against human Peroxisome proliferator activated receptor alpha Gal4 chimeric in transiently transfected CV-1 cells by funct... | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proton-coupled folate transporter (Homo sapiens (Human)) | BDBM50534431 (CHEMBL4447805) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of [3H]MTX uptake at human PCFT expressed in Chinese hamster R2/PCFT4 cells at pH 5.5 measured after 5 mins by Dixon plot analysis | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proton-coupled folate transporter (Homo sapiens (Human)) | BDBM50534430 (CHEMBL4471269) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of [3H]MTX uptake at human PCFT expressed in Chinese hamster R2/PCFT4 cells at pH 5.5 measured after 5 mins by Dixon plot analysis | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proton-coupled folate transporter (Homo sapiens (Human)) | BDBM50534426 (CHEMBL4553188) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of [3H]MTX uptake at human PCFT expressed in Chinese hamster R2/PCFT4 cells at pH 5.5 measured after 5 mins by Dixon plot analysis | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106959 ((S)-2-Dimethylamino-3-{4-[2-(5-methyl-2-phenyl-oxa...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proton-coupled folate transporter (Homo sapiens (Human)) | BDBM50534427 (CHEMBL4437824) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of [3H]MTX uptake at human PCFT expressed in Chinese hamster R2/PCFT4 cells at pH 5.5 measured after 5 mins by Dixon plot analysis | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50106968 ((S)-2-(2,5-Dimethyl-pyrrol-1-yl)-3-{4-[2-(5-methyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 980 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Binding affinity to human Peroxisome proliferator activated receptor gamma using scintillation proximity assay | Bioorg Med Chem Lett 11: 3111-3 (2001) BindingDB Entry DOI: 10.7270/Q2B27TKM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trifunctional purine biosynthetic protein adenosine-3 (Homo sapiens (Human)) | BDBM50027656 (CHEBI:63616 | LY-2315 | LY-231514 | PEMETREXED | U...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University Curated by ChEMBL | Assay Description Inhibition of recombinant N-terminal 6His-tagged human GARFTase assessed as formation of 5,8-dideazafolate from 10-formyl-5,8-dideazafolic acid measu... | J Med Chem 59: 7856-76 (2016) Article DOI: 10.1021/acs.jmedchem.6b00594 BindingDB Entry DOI: 10.7270/Q2NG4V5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50143823 ((E)-3-[5-Methyl-4-(5,5,8,8-tetramethyl-5,6,7,8-tet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50409928 (CHEMBL2113737) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha (Homo sapiens (Human)) | BDBM50409929 (CHEMBL2113736) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity for retinoic acid receptor alpha (RARalpha), using 9-cis-[3H]-retinoic acid | J Med Chem 47: 2010-29 (2004) Article DOI: 10.1021/jm030565g BindingDB Entry DOI: 10.7270/Q2KH0MRC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 356 total ) | Next | Last >> |