 Found 7559 hits with Last Name = 'yan' and Initial = 'r'
Found 7559 hits with Last Name = 'yan' and Initial = 'r' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50379086
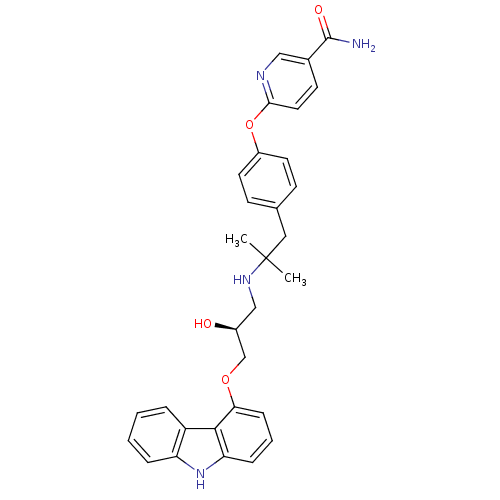
(CHEMBL2012521 | CHEMBL2012522 | LY-377604)Show SMILES CC(C)(Cc1ccc(Oc2ccc(cn2)C(N)=O)cc1)NC[C@H](O)COc1cccc2[nH]c3ccccc3c12 |r| Show InChI InChI=1S/C31H32N4O4/c1-31(2,16-20-10-13-23(14-11-20)39-28-15-12-21(17-33-28)30(32)37)34-18-22(36)19-38-27-9-5-8-26-29(27)24-6-3-4-7-25(24)35-26/h3-15,17,22,34-36H,16,18-19H2,1-2H3,(H2,32,37)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]Iodocyanopindolol from human adrenergic beta2 receptor expressed in insect sf9 cells by scintillation counting |
ACS Med Chem Lett 2: 583-586 (2011)
Article DOI: 10.1021/ml200071k
BindingDB Entry DOI: 10.7270/Q20R9QDP |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50379086

(CHEMBL2012521 | CHEMBL2012522 | LY-377604)Show SMILES CC(C)(Cc1ccc(Oc2ccc(cn2)C(N)=O)cc1)NC[C@H](O)COc1cccc2[nH]c3ccccc3c12 |r| Show InChI InChI=1S/C31H32N4O4/c1-31(2,16-20-10-13-23(14-11-20)39-28-15-12-21(17-33-28)30(32)37)34-18-22(36)19-38-27-9-5-8-26-29(27)24-6-3-4-7-25(24)35-26/h3-15,17,22,34-36H,16,18-19H2,1-2H3,(H2,32,37)/t22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]Iodocyanopindolol from human adrenergic beta1 receptor expressed in insect sf9 cells by scintillation counting |
ACS Med Chem Lett 2: 583-586 (2011)
Article DOI: 10.1021/ml200071k
BindingDB Entry DOI: 10.7270/Q20R9QDP |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50105879
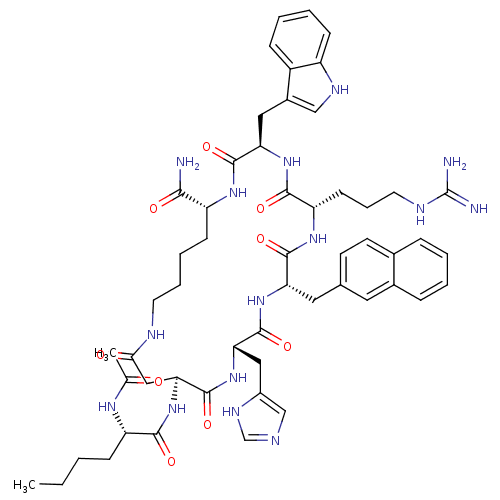
(15-(2-Acetylamino-hexanoylamino)-6-(3-guanidino-pr...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O Show InChI InChI=1S/C54H71N15O9/c1-3-4-15-40(63-31(2)70)48(73)69-45-27-46(71)59-21-10-9-17-39(47(55)72)64-51(76)43(25-35-28-61-38-16-8-7-14-37(35)38)67-49(74)41(18-11-22-60-54(56)57)65-50(75)42(24-32-19-20-33-12-5-6-13-34(33)23-32)66-52(77)44(68-53(45)78)26-36-29-58-30-62-36/h5-8,12-14,16,19-20,23,28-30,39-45,61H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,55,72)(H,58,62)(H,59,71)(H,63,70)(H,64,76)(H,65,75)(H,66,77)(H,67,74)(H,68,78)(H,69,73)(H4,56,57,60)/t39-,40+,41+,42+,43-,44+,45+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 4 (hMC4R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50491858
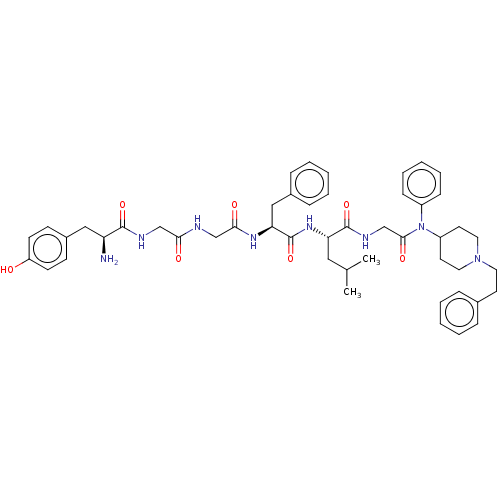
(CHEMBL2387338)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1 |r| Show InChI InChI=1S/C49H62N8O7/c1-34(2)28-42(48(63)53-33-46(61)57(38-16-10-5-11-17-38)39-23-26-56(27-24-39)25-22-35-12-6-3-7-13-35)55-49(64)43(30-36-14-8-4-9-15-36)54-45(60)32-51-44(59)31-52-47(62)41(50)29-37-18-20-40(58)21-19-37/h3-21,34,39,41-43,58H,22-33,50H2,1-2H3,(H,51,59)(H,52,62)(H,53,63)(H,54,60)(H,55,64)/t41-,42-,43-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50105879

(15-(2-Acetylamino-hexanoylamino)-6-(3-guanidino-pr...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O Show InChI InChI=1S/C54H71N15O9/c1-3-4-15-40(63-31(2)70)48(73)69-45-27-46(71)59-21-10-9-17-39(47(55)72)64-51(76)43(25-35-28-61-38-16-8-7-14-37(35)38)67-49(74)41(18-11-22-60-54(56)57)65-50(75)42(24-32-19-20-33-12-5-6-13-34(33)23-32)66-52(77)44(68-53(45)78)26-36-29-58-30-62-36/h5-8,12-14,16,19-20,23,28-30,39-45,61H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,55,72)(H,58,62)(H,59,71)(H,63,70)(H,64,76)(H,65,75)(H,66,77)(H,67,74)(H,68,78)(H,69,73)(H4,56,57,60)/t39-,40+,41+,42+,43-,44+,45+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 5 (hMC5R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50105880
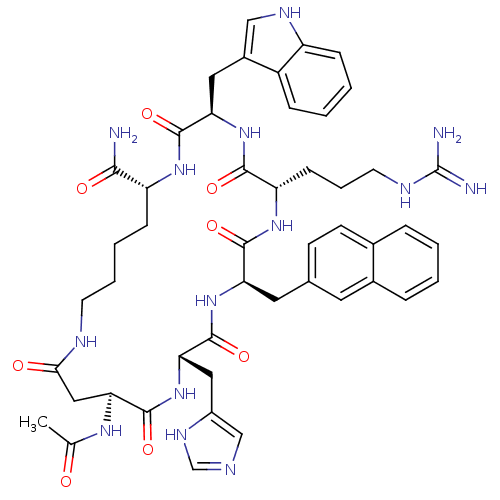
(15-Acetylamino-6-(3-guanidino-propyl)-12-(3H-imida...)Show SMILES CC(=O)N[C@H]1CC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O Show InChI InChI=1S/C48H60N14O8/c1-27(63)57-40-23-41(64)53-17-7-6-13-35(42(49)65)58-45(68)38(21-31-24-55-34-12-5-4-11-33(31)34)61-43(66)36(14-8-18-54-48(50)51)59-44(67)37(20-28-15-16-29-9-2-3-10-30(29)19-28)60-46(69)39(62-47(40)70)22-32-25-52-26-56-32/h2-5,9-12,15-16,19,24-26,35-40,55H,6-8,13-14,17-18,20-23H2,1H3,(H2,49,65)(H,52,56)(H,53,64)(H,57,63)(H,58,68)(H,59,67)(H,60,69)(H,61,66)(H,62,70)(H4,50,51,54)/t35-,36+,37-,38-,39+,40+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 4 (hMC4R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Gastrin-releasing peptide receptor
(MOUSE) | BDBM85488
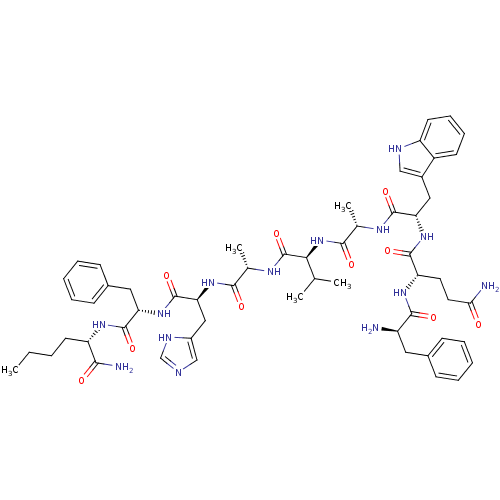
(DPhe6,BetaAla11,Phe13,Nle14-Bn(6-14))Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](N)Cc1ccccc1)C(C)C)C(N)=O Show InChI InChI=1S/C57H76N14O10/c1-6-7-21-42(49(60)73)66-55(79)44(26-36-18-12-9-13-19-36)69-56(80)46(28-38-30-61-31-63-38)68-50(74)33(4)65-57(81)48(32(2)3)71-51(75)34(5)64-54(78)45(27-37-29-62-41-22-15-14-20-39(37)41)70-53(77)43(23-24-47(59)72)67-52(76)40(58)25-35-16-10-8-11-17-35/h8-20,22,29-34,40,42-46,48,62H,6-7,21,23-28,58H2,1-5H3,(H2,59,72)(H2,60,73)(H,61,63)(H,64,78)(H,65,81)(H,66,79)(H,67,76)(H,68,74)(H,69,80)(H,70,77)(H,71,75)/t33-,34-,40+,42-,43-,44-,45-,46-,48-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by PDSP Ki Database
| |
Biochemistry 38: 7307-20 (1999)
Article DOI: 10.1021/bi990204w
BindingDB Entry DOI: 10.7270/Q29022BG |
More data for this
Ligand-Target Pair | |
Protein kinase C theta type
(Homo sapiens (Human)) | BDBM50391386
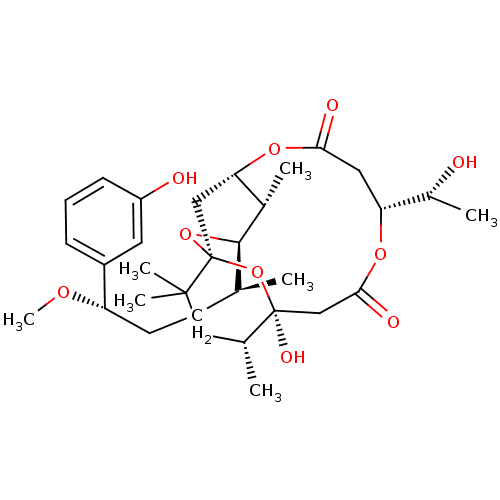
(CHEMBL2148106)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cccc(O)c1 |r| Show InChI InChI=1S/C32H48O10/c1-18(11-12-24(38-7)22-9-8-10-23(34)13-22)29-20(3)26-16-32(41-29)30(5,6)15-19(2)31(37,42-32)17-28(36)39-25(21(4)33)14-27(35)40-26/h8-10,13,18-21,24-26,29,33-34,37H,11-12,14-17H2,1-7H3/t18-,19+,20-,21+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCtheta C1B domain |
J Med Chem 55: 5614-26 (2012)
Article DOI: 10.1021/jm300566h
BindingDB Entry DOI: 10.7270/Q200036J |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50391386

(CHEMBL2148106)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cccc(O)c1 |r| Show InChI InChI=1S/C32H48O10/c1-18(11-12-24(38-7)22-9-8-10-23(34)13-22)29-20(3)26-16-32(41-29)30(5,6)15-19(2)31(37,42-32)17-28(36)39-25(21(4)33)14-27(35)40-26/h8-10,13,18-21,24-26,29,33-34,37H,11-12,14-17H2,1-7H3/t18-,19+,20-,21+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCeta C1B domain |
J Med Chem 55: 5614-26 (2012)
Article DOI: 10.1021/jm300566h
BindingDB Entry DOI: 10.7270/Q200036J |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50001157
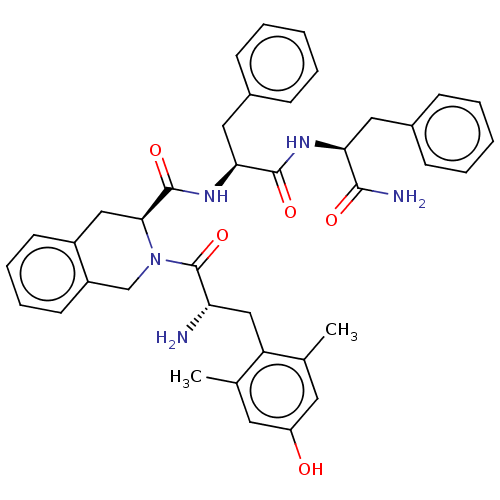
(CHEMBL538700)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H43N5O5/c1-24-17-30(45)18-25(2)31(24)22-32(40)39(49)44-23-29-16-10-9-15-28(29)21-35(44)38(48)43-34(20-27-13-7-4-8-14-27)37(47)42-33(36(41)46)19-26-11-5-3-6-12-26/h3-18,32-35,45H,19-23,40H2,1-2H3,(H2,41,46)(H,42,47)(H,43,48)/t32-,33-,34-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in human/mouse HN9.10 cell membranes after 3 hrs by liquid scintillation countin... |
ACS Med Chem Lett 4: 656-659 (2013)
Article DOI: 10.1021/ml400115n
BindingDB Entry DOI: 10.7270/Q2CN76VX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50491860

(CHEMBL2387215)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccc(Cl)cc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCNC(=O)NCCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C86H122ClN21O15/c1-52(2)45-69(105-81(119)70(49-58-28-30-59(87)31-29-58)100-73(111)51-98-75(113)55(5)99-76(114)64(88)50-63-53(3)46-62(109)47-54(63)4)80(118)102-66(27-18-40-95-85(92)93)77(115)101-65(26-17-39-94-84(90)91)78(116)103-67(32-33-72(89)110)79(117)106-71(48-57-21-11-7-12-22-57)82(120)104-68(83(121)122)25-15-16-38-96-86(123)97-41-34-74(112)108(60-23-13-8-14-24-60)61-36-43-107(44-37-61)42-35-56-19-9-6-10-20-56/h6-14,19-24,28-31,46-47,52,55,61,64-71,109H,15-18,25-27,32-45,48-51,88H2,1-5H3,(H2,89,110)(H,98,113)(H,99,114)(H,100,111)(H,101,115)(H,102,118)(H,103,116)(H,104,120)(H,105,119)(H,106,117)(H,121,122)(H4,90,91,94)(H4,92,93,95)(H2,96,97,123)/t55-,64+,65-,66+,67+,68+,69+,70+,71+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Protein kinase C theta type
(Homo sapiens (Human)) | BDBM50327943
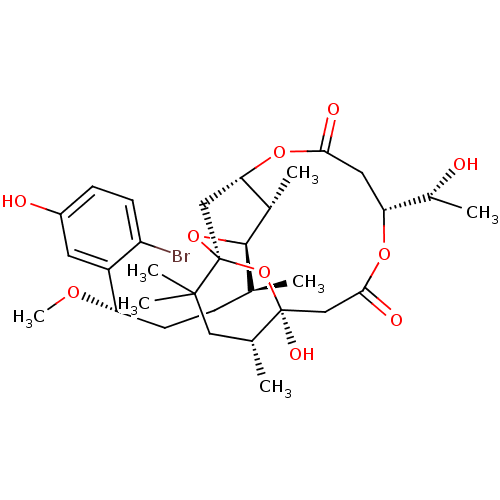
(Aplysiatoxin | CHEMBL1256416)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cc(O)ccc1Br |r| Show InChI InChI=1S/C32H47BrO10/c1-17(8-11-24(39-7)22-12-21(35)9-10-23(22)33)29-19(3)26-15-32(42-29)30(5,6)14-18(2)31(38,43-32)16-28(37)40-25(20(4)34)13-27(36)41-26/h9-10,12,17-20,24-26,29,34-35,38H,8,11,13-16H2,1-7H3/t17-,18+,19-,20+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCtheta C1B peptide |
Bioorg Med Chem Lett 20: 6064-6 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.051
BindingDB Entry DOI: 10.7270/Q2FJ2H04 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50491854
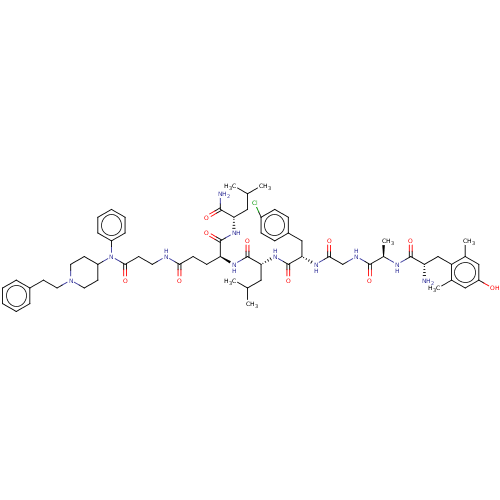
(CHEMBL2387327)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CCC(=O)NCCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](Cc1ccc(Cl)cc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(N)=O |r| Show InChI InChI=1S/C64H88ClN11O10/c1-39(2)32-53(59(67)81)73-62(84)52(22-23-56(78)68-28-24-58(80)76(47-16-12-9-13-17-47)48-26-30-75(31-27-48)29-25-44-14-10-8-11-15-44)72-63(85)54(33-40(3)4)74-64(86)55(36-45-18-20-46(65)21-19-45)71-57(79)38-69-60(82)43(7)70-61(83)51(66)37-50-41(5)34-49(77)35-42(50)6/h8-21,34-35,39-40,43,48,51-55,77H,22-33,36-38,66H2,1-7H3,(H2,67,81)(H,68,78)(H,69,82)(H,70,83)(H,71,79)(H,72,85)(H,73,84)(H,74,86)/t43-,51+,52+,53+,54-,55+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50063266
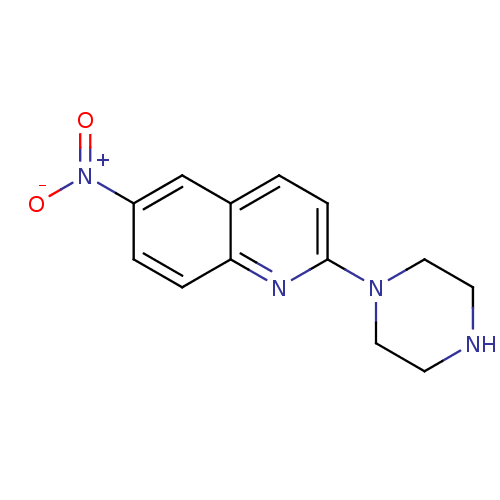
(6-Nitro-2-piperazin-1-yl-quinoline | 6-nitroquipaz...)Show InChI InChI=1S/C13H14N4O2/c18-17(19)11-2-3-12-10(9-11)1-4-13(15-12)16-7-5-14-6-8-16/h1-4,9,14H,5-8H2 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Medicines Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]-5HT from human SERT |
Eur J Med Chem 49: 200-10 (2012)
Article DOI: 10.1016/j.ejmech.2012.01.012
BindingDB Entry DOI: 10.7270/Q2K35V4Z |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50391386

(CHEMBL2148106)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cccc(O)c1 |r| Show InChI InChI=1S/C32H48O10/c1-18(11-12-24(38-7)22-9-8-10-23(34)13-22)29-20(3)26-16-32(41-29)30(5,6)15-19(2)31(37,42-32)17-28(36)39-25(21(4)33)14-27(35)40-26/h8-10,13,18-21,24-26,29,33-34,37H,11-12,14-17H2,1-7H3/t18-,19+,20-,21+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCbeta C1A domain |
J Med Chem 55: 5614-26 (2012)
Article DOI: 10.1021/jm300566h
BindingDB Entry DOI: 10.7270/Q200036J |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50493217
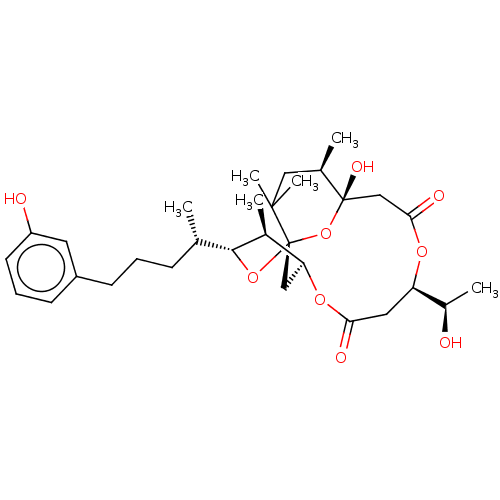
(DEMETHOXYDEBROMOAPLYSIATOXIN)Show SMILES [H][C@]12C[C@]3(O[C@]([H])([C@@H](C)CCCc4cccc(O)c4)[C@H]1C)O[C@@](O)(CC(=O)O[C@]([H])(CC(=O)O2)[C@@H](C)O)[C@H](C)CC3(C)C |r| Show InChI InChI=1S/C31H46O9/c1-18(9-7-10-22-11-8-12-23(33)13-22)28-20(3)25-16-31(39-28)29(5,6)15-19(2)30(36,40-31)17-27(35)37-24(21(4)32)14-26(34)38-25/h8,11-13,18-21,24-25,28,32-33,36H,7,9-10,14-17H2,1-6H3/t18-,19+,20-,21+,24+,25-,28+,30-,31-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu from PKCeta C1B domain (unknown origin) |
Bioorg Med Chem Lett 23: 4319-23 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.096
BindingDB Entry DOI: 10.7270/Q2VD72CX |
More data for this
Ligand-Target Pair | |
Gastrin-releasing peptide receptor
(MOUSE) | BDBM85488

(DPhe6,BetaAla11,Phe13,Nle14-Bn(6-14))Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](N)Cc1ccccc1)C(C)C)C(N)=O Show InChI InChI=1S/C57H76N14O10/c1-6-7-21-42(49(60)73)66-55(79)44(26-36-18-12-9-13-19-36)69-56(80)46(28-38-30-61-31-63-38)68-50(74)33(4)65-57(81)48(32(2)3)71-51(75)34(5)64-54(78)45(27-37-29-62-41-22-15-14-20-39(37)41)70-53(77)43(23-24-47(59)72)67-52(76)40(58)25-35-16-10-8-11-17-35/h8-20,22,29-34,40,42-46,48,62H,6-7,21,23-28,58H2,1-5H3,(H2,59,72)(H2,60,73)(H,61,63)(H,64,78)(H,65,81)(H,66,79)(H,67,76)(H,68,74)(H,69,80)(H,70,77)(H,71,75)/t33-,34-,40+,42-,43-,44-,45-,46-,48-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by PDSP Ki Database
| |
Biochemistry 38: 7307-20 (1999)
Article DOI: 10.1021/bi990204w
BindingDB Entry DOI: 10.7270/Q29022BG |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50492533

(CHEMBL2408011)Show SMILES CCC(=O)N(C1CCN(CC1)C(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1ccccc1 |r| Show InChI InChI=1S/C44H51N5O5/c1-4-41(51)49(34-17-9-6-10-18-34)35-19-21-47(22-20-35)44(54)39(25-31-13-7-5-8-14-31)46-42(52)40-26-32-15-11-12-16-33(32)28-48(40)43(53)38(45)27-37-29(2)23-36(50)24-30(37)3/h5-18,23-24,35,38-40,50H,4,19-22,25-28,45H2,1-3H3,(H,46,52)/t38-,39-,40-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in human/mouse HN9.10 cell membranes after 3 hrs by liquid scintillation countin... |
ACS Med Chem Lett 4: 656-659 (2013)
Article DOI: 10.1021/ml400115n
BindingDB Entry DOI: 10.7270/Q2CN76VX |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50391386

(CHEMBL2148106)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cccc(O)c1 |r| Show InChI InChI=1S/C32H48O10/c1-18(11-12-24(38-7)22-9-8-10-23(34)13-22)29-20(3)26-16-32(41-29)30(5,6)15-19(2)31(37,42-32)17-28(36)39-25(21(4)33)14-27(35)40-26/h8-10,13,18-21,24-26,29,33-34,37H,11-12,14-17H2,1-7H3/t18-,19+,20-,21+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCdelta C1B domain |
J Med Chem 55: 5614-26 (2012)
Article DOI: 10.1021/jm300566h
BindingDB Entry DOI: 10.7270/Q200036J |
More data for this
Ligand-Target Pair | |
Protein kinase C theta type
(Homo sapiens (Human)) | BDBM50493217

(DEMETHOXYDEBROMOAPLYSIATOXIN)Show SMILES [H][C@]12C[C@]3(O[C@]([H])([C@@H](C)CCCc4cccc(O)c4)[C@H]1C)O[C@@](O)(CC(=O)O[C@]([H])(CC(=O)O2)[C@@H](C)O)[C@H](C)CC3(C)C |r| Show InChI InChI=1S/C31H46O9/c1-18(9-7-10-22-11-8-12-23(33)13-22)28-20(3)25-16-31(39-28)29(5,6)15-19(2)30(36,40-31)17-27(35)37-24(21(4)32)14-26(34)38-25/h8,11-13,18-21,24-25,28,32-33,36H,7,9-10,14-17H2,1-6H3/t18-,19+,20-,21+,24+,25-,28+,30-,31-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu from PKCtheta C1B domain (unknown origin) |
Bioorg Med Chem Lett 23: 4319-23 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.096
BindingDB Entry DOI: 10.7270/Q2VD72CX |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 3
(Homo sapiens (Human)) | BDBM50105879

(15-(2-Acetylamino-hexanoylamino)-6-(3-guanidino-pr...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O Show InChI InChI=1S/C54H71N15O9/c1-3-4-15-40(63-31(2)70)48(73)69-45-27-46(71)59-21-10-9-17-39(47(55)72)64-51(76)43(25-35-28-61-38-16-8-7-14-37(35)38)67-49(74)41(18-11-22-60-54(56)57)65-50(75)42(24-32-19-20-33-12-5-6-13-34(33)23-32)66-52(77)44(68-53(45)78)26-36-29-58-30-62-36/h5-8,12-14,16,19-20,23,28-30,39-45,61H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,55,72)(H,58,62)(H,59,71)(H,63,70)(H,64,76)(H,65,75)(H,66,77)(H,67,74)(H,68,78)(H,69,73)(H4,56,57,60)/t39-,40+,41+,42+,43-,44+,45+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 3 (hMC3R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50205841
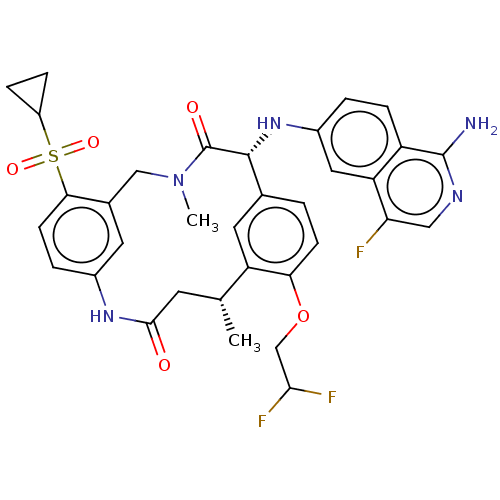
(CHEMBL3898956)Show SMILES C[C@@H]1CC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc(OCC(F)F)c1c3)c2)S(=O)(=O)C1CC1 |r| Show InChI InChI=1S/C34H34F3N5O5S/c1-18-11-31(43)40-21-5-10-29(48(45,46)23-6-7-23)20(12-21)16-42(2)34(44)32(19-3-9-28(25(18)13-19)47-17-30(36)37)41-22-4-8-24-26(14-22)27(35)15-39-33(24)38/h3-5,8-10,12-15,18,23,30,32,41H,6-7,11,16-17H2,1-2H3,(H2,38,39)(H,40,43)/t18-,32-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human factor-7a/TF using S2288 as substrate measured after 60 mins at 37 degC |
ACS Med Chem Lett 8: 67-72 (2017)
Article DOI: 10.1021/acsmedchemlett.6b00375
BindingDB Entry DOI: 10.7270/Q20P120Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gastrin-releasing peptide receptor
(MOUSE) | BDBM85488

(DPhe6,BetaAla11,Phe13,Nle14-Bn(6-14))Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](N)Cc1ccccc1)C(C)C)C(N)=O Show InChI InChI=1S/C57H76N14O10/c1-6-7-21-42(49(60)73)66-55(79)44(26-36-18-12-9-13-19-36)69-56(80)46(28-38-30-61-31-63-38)68-50(74)33(4)65-57(81)48(32(2)3)71-51(75)34(5)64-54(78)45(27-37-29-62-41-22-15-14-20-39(37)41)70-53(77)43(23-24-47(59)72)67-52(76)40(58)25-35-16-10-8-11-17-35/h8-20,22,29-34,40,42-46,48,62H,6-7,21,23-28,58H2,1-5H3,(H2,59,72)(H2,60,73)(H,61,63)(H,64,78)(H,65,81)(H,66,79)(H,67,76)(H,68,74)(H,69,80)(H,70,77)(H,71,75)/t33-,34-,40+,42-,43-,44-,45-,46-,48-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by PDSP Ki Database
| |
Biochemistry 38: 7307-20 (1999)
Article DOI: 10.1021/bi990204w
BindingDB Entry DOI: 10.7270/Q29022BG |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50391386

(CHEMBL2148106)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cccc(O)c1 |r| Show InChI InChI=1S/C32H48O10/c1-18(11-12-24(38-7)22-9-8-10-23(34)13-22)29-20(3)26-16-32(41-29)30(5,6)15-19(2)31(37,42-32)17-28(36)39-25(21(4)33)14-27(35)40-26/h8-10,13,18-21,24-26,29,33-34,37H,11-12,14-17H2,1-7H3/t18-,19+,20-,21+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCalpha C1A domain |
J Med Chem 55: 5614-26 (2012)
Article DOI: 10.1021/jm300566h
BindingDB Entry DOI: 10.7270/Q200036J |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50491856
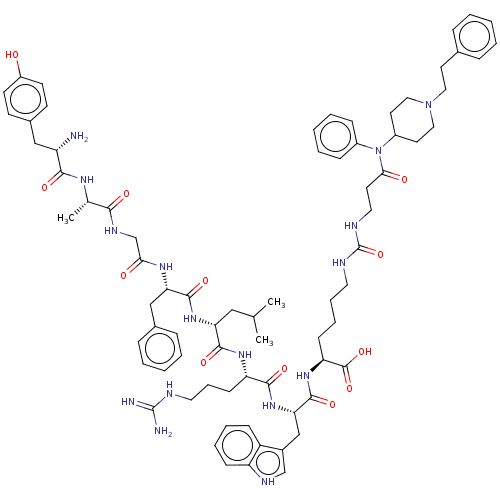
(CHEMBL2387214)Show SMILES CC(C)C[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCCNC(=O)NCCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C75H100N16O12/c1-48(2)42-62(88-71(99)63(44-51-20-9-5-10-21-51)85-65(93)47-83-67(95)49(3)84-68(96)58(76)43-52-28-30-56(92)31-29-52)70(98)86-60(27-17-37-79-74(77)78)69(97)89-64(45-53-46-82-59-25-14-13-24-57(53)59)72(100)87-61(73(101)102)26-15-16-36-80-75(103)81-38-32-66(94)91(54-22-11-6-12-23-54)55-34-40-90(41-35-55)39-33-50-18-7-4-8-19-50/h4-14,18-25,28-31,46,48-49,55,58,60-64,82,92H,15-17,26-27,32-45,47,76H2,1-3H3,(H,83,95)(H,84,96)(H,85,93)(H,86,98)(H,87,100)(H,88,99)(H,89,97)(H,101,102)(H4,77,78,79)(H2,80,81,103)/t49-,58-,60-,61-,62+,63-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50105893
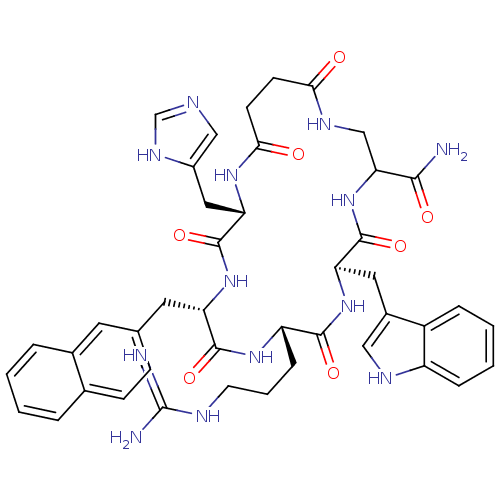
(9-(3-Guanidino-propyl)-15-(3H-imidazol-4-ylmethyl)...)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)CCC(=O)NCC(NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC1=O)C(N)=O Show InChI InChI=1S/C43H51N13O7/c44-38(59)35-22-50-36(57)13-14-37(58)52-34(19-28-21-47-23-51-28)42(63)54-32(17-24-11-12-25-6-1-2-7-26(25)16-24)40(61)53-31(10-5-15-48-43(45)46)39(60)55-33(41(62)56-35)18-27-20-49-30-9-4-3-8-29(27)30/h1-4,6-9,11-12,16,20-21,23,31-35,49H,5,10,13-15,17-19,22H2,(H2,44,59)(H,47,51)(H,50,57)(H,52,58)(H,53,61)(H,54,63)(H,55,60)(H,56,62)(H4,45,46,48)/t31-,32-,33+,34-,35?/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 4 (hMC4R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Gastrin-releasing peptide receptor
(MOUSE) | BDBM85488

(DPhe6,BetaAla11,Phe13,Nle14-Bn(6-14))Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](N)Cc1ccccc1)C(C)C)C(N)=O Show InChI InChI=1S/C57H76N14O10/c1-6-7-21-42(49(60)73)66-55(79)44(26-36-18-12-9-13-19-36)69-56(80)46(28-38-30-61-31-63-38)68-50(74)33(4)65-57(81)48(32(2)3)71-51(75)34(5)64-54(78)45(27-37-29-62-41-22-15-14-20-39(37)41)70-53(77)43(23-24-47(59)72)67-52(76)40(58)25-35-16-10-8-11-17-35/h8-20,22,29-34,40,42-46,48,62H,6-7,21,23-28,58H2,1-5H3,(H2,59,72)(H2,60,73)(H,61,63)(H,64,78)(H,65,81)(H,66,79)(H,67,76)(H,68,74)(H,69,80)(H,70,77)(H,71,75)/t33-,34-,40+,42-,43-,44-,45-,46-,48-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by PDSP Ki Database
| |
Biochemistry 38: 7307-20 (1999)
Article DOI: 10.1021/bi990204w
BindingDB Entry DOI: 10.7270/Q29022BG |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50491865

(CHEMBL2387211)Show SMILES CC(C)[C@H](NC(=O)[C@@H](NC(=O)C(C)(C)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(C)C)C(=O)N[C@@H](CCC(=O)NCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)C(N)=O |r| Show InChI InChI=1S/C61H83N11O10/c1-38(2)52(58(80)66-48(54(63)76)27-28-50(74)64-37-51(75)72(44-21-15-10-16-22-44)45-30-33-71(34-31-45)32-29-41-17-11-8-12-18-41)68-59(81)53(39(3)4)69-60(82)61(6,7)70-57(79)49(36-42-19-13-9-14-20-42)67-55(77)40(5)65-56(78)47(62)35-43-23-25-46(73)26-24-43/h8-26,38-40,45,47-49,52-53,73H,27-37,62H2,1-7H3,(H2,63,76)(H,64,74)(H,65,78)(H,66,80)(H,67,77)(H,68,81)(H,69,82)(H,70,79)/t40-,47+,48+,49+,52+,53+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50105892
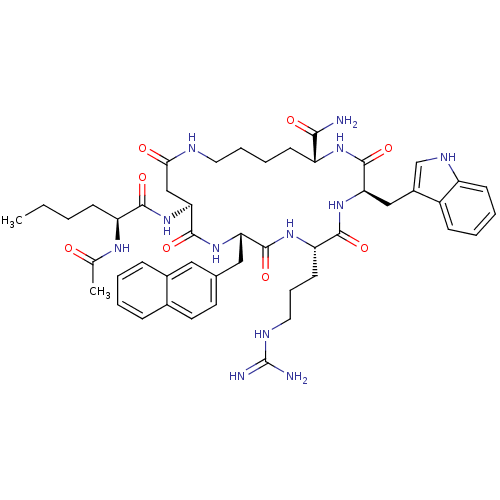
(12-(2-Acetylamino-hexanoylamino)-6-(3-guanidino-pr...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC1=O)C(N)=O Show InChI InChI=1S/C48H64N12O8/c1-3-4-15-36(55-28(2)61)43(64)60-40-26-41(62)52-21-10-9-17-35(42(49)63)56-46(67)39(25-32-27-54-34-16-8-7-14-33(32)34)59-44(65)37(18-11-22-53-48(50)51)57-45(66)38(58-47(40)68)24-29-19-20-30-12-5-6-13-31(30)23-29/h5-8,12-14,16,19-20,23,27,35-40,54H,3-4,9-11,15,17-18,21-22,24-26H2,1-2H3,(H2,49,63)(H,52,62)(H,55,61)(H,56,67)(H,57,66)(H,58,68)(H,59,65)(H,60,64)(H4,50,51,53)/t35-,36+,37+,38+,39-,40+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 4 (hMC4R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50491869
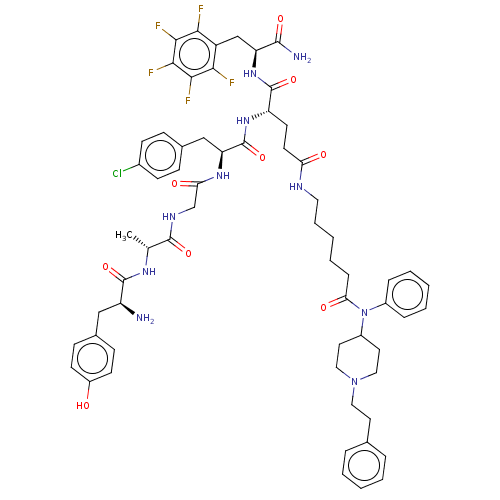
(CHEMBL2387328)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N[C@@H](Cc1ccc(Cl)cc1)C(=O)N[C@@H](CCC(=O)NCCCCCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)C(=O)N[C@@H](Cc1c(F)c(F)c(F)c(F)c1F)C(N)=O |r| Show InChI InChI=1S/C62H72ClF5N10O9/c1-37(73-60(85)46(69)33-39-18-22-44(79)23-19-39)59(84)72-36-51(81)74-49(34-40-16-20-41(63)21-17-40)62(87)75-47(61(86)76-48(58(70)83)35-45-53(64)55(66)57(68)56(67)54(45)65)24-25-50(80)71-29-10-4-9-15-52(82)78(42-13-7-3-8-14-42)43-27-31-77(32-28-43)30-26-38-11-5-2-6-12-38/h2-3,5-8,11-14,16-23,37,43,46-49,79H,4,9-10,15,24-36,69H2,1H3,(H2,70,83)(H,71,80)(H,72,84)(H,73,85)(H,74,81)(H,75,87)(H,76,86)/t37-,46+,47+,48+,49+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18699
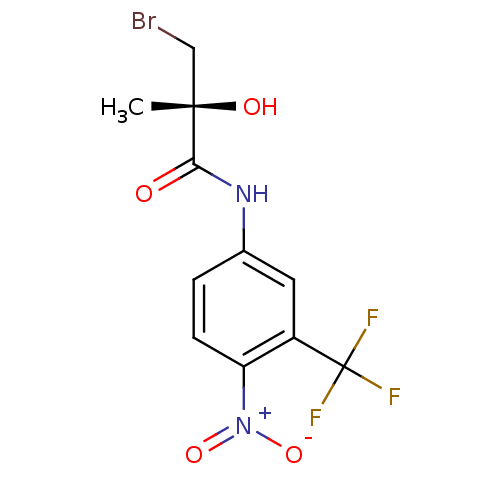
((2R)-3-bromo-2-hydroxy-2-methyl-N-[4-nitro-3-(trif...)Show SMILES C[C@](O)(CBr)C(=O)Nc1ccc(c(c1)C(F)(F)F)[N+]([O-])=O |r| Show InChI InChI=1S/C11H10BrF3N2O4/c1-10(19,5-12)9(18)16-6-2-3-8(17(20)21)7(4-6)11(13,14)15/h2-4,19H,5H2,1H3,(H,16,18)/t10-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GTx, Inc.
Curated by ChEMBL
| Assay Description
Agonist activity at androgen receptor (unknown origin) |
J Med Chem 52: 3597-617 (2009)
Article DOI: 10.1021/jm900280m
BindingDB Entry DOI: 10.7270/Q2GH9HWD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50063581
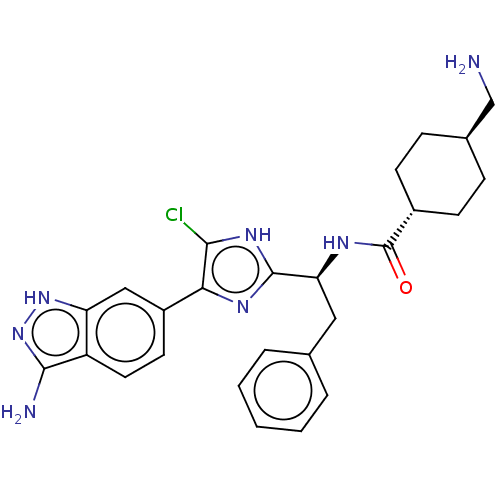
(CHEMBL3398612)Show SMILES NC[C@H]1CC[C@@H](CC1)C(=O)N[C@@H](Cc1ccccc1)c1nc(c(Cl)[nH]1)-c1ccc2c(N)n[nH]c2c1 |r,wU:5.8,wD:2.1,11.11,(25.62,-40.59,;26.39,-39.26,;27.93,-39.26,;28.7,-40.59,;30.24,-40.59,;31.01,-39.26,;30.24,-37.93,;28.7,-37.93,;32.55,-39.26,;33.32,-40.59,;33.32,-37.93,;34.86,-37.93,;35.63,-39.26,;37.17,-39.26,;37.94,-37.93,;39.48,-37.93,;40.25,-39.26,;39.48,-40.59,;37.94,-40.59,;35.63,-36.59,;35.01,-35.19,;36.14,-34.15,;37.48,-34.93,;38.89,-34.3,;37.16,-36.43,;35.98,-32.62,;37.23,-31.72,;37.07,-30.19,;35.66,-29.56,;35.19,-28.09,;36.09,-26.85,;33.65,-28.09,;33.17,-29.56,;34.42,-30.47,;34.58,-32,)| Show InChI InChI=1S/C26H28ClN7O/c27-23-22(18-10-11-19-20(13-18)33-34-24(19)29)31-25(32-23)21(12-15-4-2-1-3-5-15)30-26(35)17-8-6-16(14-28)7-9-17/h1-5,10-11,13,16-17,21H,6-9,12,14,28-29H2,(H,30,35)/b22-18-/t16-,17-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Competitive inhibition of human coagulation factor 11a assessed as reduction in release of p-nitroaniline after 10 to 120 mins by Michaelis-Menten eq... |
Bioorg Med Chem 24: 2257-72 (2016)
Article DOI: 10.1016/j.bmc.2016.03.062
BindingDB Entry DOI: 10.7270/Q2DN46X0 |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50493217

(DEMETHOXYDEBROMOAPLYSIATOXIN)Show SMILES [H][C@]12C[C@]3(O[C@]([H])([C@@H](C)CCCc4cccc(O)c4)[C@H]1C)O[C@@](O)(CC(=O)O[C@]([H])(CC(=O)O2)[C@@H](C)O)[C@H](C)CC3(C)C |r| Show InChI InChI=1S/C31H46O9/c1-18(9-7-10-22-11-8-12-23(33)13-22)28-20(3)25-16-31(39-28)29(5,6)15-19(2)30(36,40-31)17-27(35)37-24(21(4)32)14-26(34)38-25/h8,11-13,18-21,24-25,28,32-33,36H,7,9-10,14-17H2,1-6H3/t18-,19+,20-,21+,24+,25-,28+,30-,31-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu from PKCdelta C1B domain (unknown origin) |
Bioorg Med Chem Lett 23: 4319-23 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.096
BindingDB Entry DOI: 10.7270/Q2VD72CX |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50229772
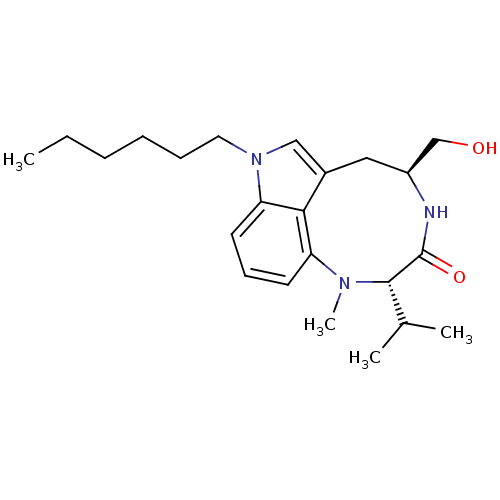
(1-hexylindolactam-V | CHEMBL399981)Show SMILES CCCCCCn1cc2C[C@@H](CO)NC(=O)[C@H](C(C)C)N(C)c3cccc1c23 Show InChI InChI=1S/C23H35N3O2/c1-5-6-7-8-12-26-14-17-13-18(15-27)24-23(28)22(16(2)3)25(4)19-10-9-11-20(26)21(17)19/h9-11,14,16,18,22,27H,5-8,12-13,15H2,1-4H3,(H,24,28)/t18-,22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu from PKCeta C1B domain |
J Med Chem 51: 46-56 (2008)
Article DOI: 10.1021/jm0706719
BindingDB Entry DOI: 10.7270/Q2445M7Q |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50491857
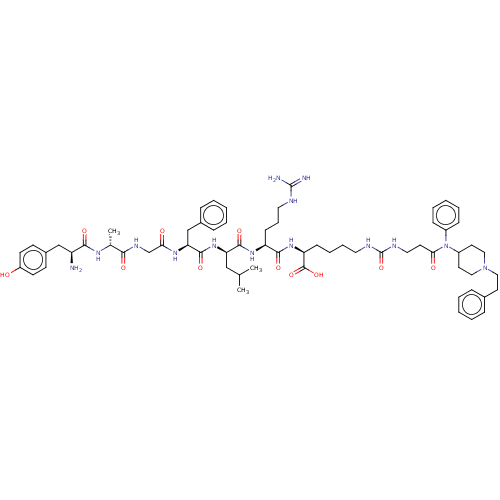
(CHEMBL2387213)Show SMILES CC(C)C[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCNC(=O)NCCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C64H90N14O11/c1-42(2)38-53(76-61(86)54(40-45-18-9-5-10-19-45)73-55(80)41-71-57(82)43(3)72-58(83)50(65)39-46-24-26-49(79)27-25-46)60(85)74-51(23-15-33-68-63(66)67)59(84)75-52(62(87)88)22-13-14-32-69-64(89)70-34-28-56(81)78(47-20-11-6-12-21-47)48-30-36-77(37-31-48)35-29-44-16-7-4-8-17-44/h4-12,16-21,24-27,42-43,48,50-54,79H,13-15,22-23,28-41,65H2,1-3H3,(H,71,82)(H,72,83)(H,73,80)(H,74,85)(H,75,84)(H,76,86)(H,87,88)(H4,66,67,68)(H2,69,70,89)/t43-,50+,51+,52+,53-,54+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50491854

(CHEMBL2387327)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CCC(=O)NCCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](Cc1ccc(Cl)cc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(N)=O |r| Show InChI InChI=1S/C64H88ClN11O10/c1-39(2)32-53(59(67)81)73-62(84)52(22-23-56(78)68-28-24-58(80)76(47-16-12-9-13-17-47)48-26-30-75(31-27-48)29-25-44-14-10-8-11-15-44)72-63(85)54(33-40(3)4)74-64(86)55(36-45-18-20-46(65)21-19-45)71-57(79)38-69-60(82)43(7)70-61(83)51(66)37-50-41(5)34-49(77)35-42(50)6/h8-21,34-35,39-40,43,48,51-55,77H,22-33,36-38,66H2,1-7H3,(H2,67,81)(H,68,78)(H,69,82)(H,70,83)(H,71,79)(H,72,85)(H,73,84)(H,74,86)/t43-,51+,52+,53+,54-,55+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50136575
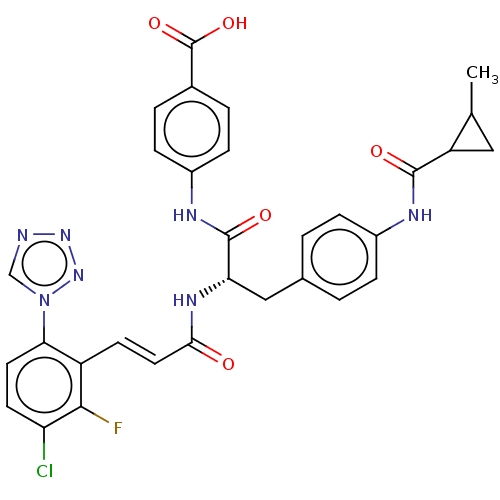
(CHEMBL3752610)Show SMILES CC1CC1C(=O)Nc1ccc(C[C@H](NC(=O)\C=C\c2c(F)c(Cl)ccc2-n2cnnn2)C(=O)Nc2ccc(cc2)C(O)=O)cc1 |r| Show InChI InChI=1S/C31H27ClFN7O5/c1-17-14-23(17)29(42)35-20-6-2-18(3-7-20)15-25(30(43)36-21-8-4-19(5-9-21)31(44)45)37-27(41)13-10-22-26(40-16-34-38-39-40)12-11-24(32)28(22)33/h2-13,16-17,23,25H,14-15H2,1H3,(H,35,42)(H,36,43)(H,37,41)(H,44,45)/b13-10+/t17?,23?,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 11a assessed as substrate hydrolysis to p-nitroaniline incubated for 10 to 120 mins by spectrophotometry analy... |
Bioorg Med Chem Lett 26: 472-8 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.089
BindingDB Entry DOI: 10.7270/Q27H1MFM |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM21121
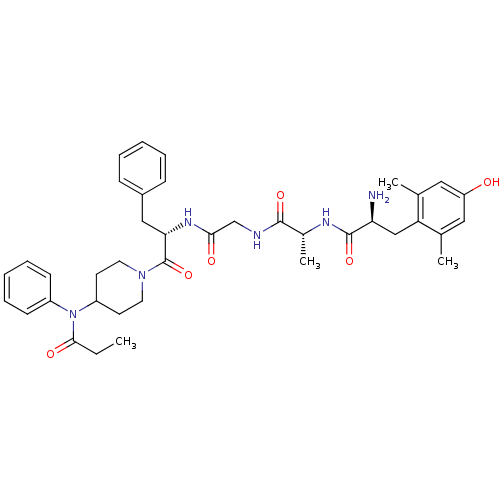
((2S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-N-[(...)Show SMILES CCC(=O)N(C1CCN(CC1)C(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1ccccc1 Show InChI InChI=1S/C39H50N6O6/c1-5-36(48)45(29-14-10-7-11-15-29)30-16-18-44(19-17-30)39(51)34(22-28-12-8-6-9-13-28)43-35(47)24-41-37(49)27(4)42-38(50)33(40)23-32-25(2)20-31(46)21-26(32)3/h6-15,20-21,27,30,33-34,46H,5,16-19,22-24,40H2,1-4H3,(H,41,49)(H,42,50)(H,43,47)/t27-,33+,34+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.360 | -53.9 | n/a | n/a | 0.770 | n/a | n/a | 7.4 | 25 |
University of Arizona at Tucson
| Assay Description
Log IC50 values for each test compound were determined from nonlinear regression analysis of data collected from two independent experiments performe... |
J Med Chem 50: 5528-32 (2007)
Article DOI: 10.1021/jm061465o
BindingDB Entry DOI: 10.7270/Q2D50K8P |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50327943

(Aplysiatoxin | CHEMBL1256416)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cc(O)ccc1Br |r| Show InChI InChI=1S/C32H47BrO10/c1-17(8-11-24(39-7)22-12-21(35)9-10-23(22)33)29-19(3)26-15-32(42-29)30(5,6)14-18(2)31(38,43-32)16-28(37)40-25(20(4)34)13-27(36)41-26/h9-10,12,17-20,24-26,29,34-35,38H,8,11,13-16H2,1-7H3/t17-,18+,19-,20+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCeta C1B peptide |
Bioorg Med Chem Lett 20: 6064-6 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.051
BindingDB Entry DOI: 10.7270/Q2FJ2H04 |
More data for this
Ligand-Target Pair | |
Neuromedin-B receptor
(RAT) | BDBM85488

(DPhe6,BetaAla11,Phe13,Nle14-Bn(6-14))Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](N)Cc1ccccc1)C(C)C)C(N)=O Show InChI InChI=1S/C57H76N14O10/c1-6-7-21-42(49(60)73)66-55(79)44(26-36-18-12-9-13-19-36)69-56(80)46(28-38-30-61-31-63-38)68-50(74)33(4)65-57(81)48(32(2)3)71-51(75)34(5)64-54(78)45(27-37-29-62-41-22-15-14-20-39(37)41)70-53(77)43(23-24-47(59)72)67-52(76)40(58)25-35-16-10-8-11-17-35/h8-20,22,29-34,40,42-46,48,62H,6-7,21,23-28,58H2,1-5H3,(H2,59,72)(H2,60,73)(H,61,63)(H,64,78)(H,65,81)(H,66,79)(H,67,76)(H,68,74)(H,69,80)(H,70,77)(H,71,75)/t33-,34-,40+,42-,43-,44-,45-,46-,48-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by PDSP Ki Database
| |
Biochemistry 38: 7307-20 (1999)
Article DOI: 10.1021/bi990204w
BindingDB Entry DOI: 10.7270/Q29022BG |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50105889
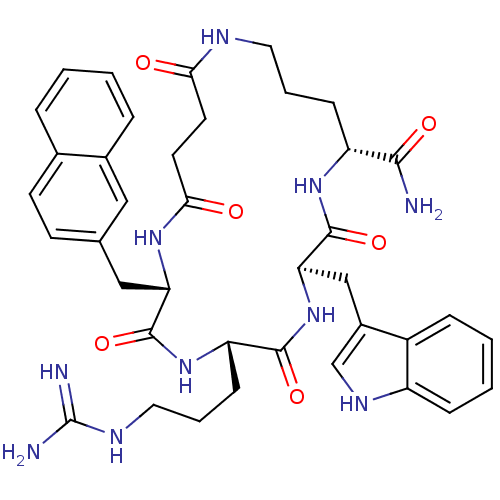
(5-(3-Guanidino-propyl)-8-(1H-indol-3-ylmethyl)-2-n...)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)CCC(=O)NCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC1=O)C(N)=O Show InChI InChI=1S/C39H48N10O6/c40-35(52)29-11-5-17-43-33(50)15-16-34(51)46-31(20-23-13-14-24-7-1-2-8-25(24)19-23)37(54)48-30(12-6-18-44-39(41)42)36(53)49-32(38(55)47-29)21-26-22-45-28-10-4-3-9-27(26)28/h1-4,7-10,13-14,19,22,29-32,45H,5-6,11-12,15-18,20-21H2,(H2,40,52)(H,43,50)(H,46,51)(H,47,55)(H,48,54)(H,49,53)(H4,41,42,44)/t29-,30+,31+,32-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 4 (hMC4R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Protein kinase C gamma type
(Homo sapiens (Human)) | BDBM50391386

(CHEMBL2148106)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cccc(O)c1 |r| Show InChI InChI=1S/C32H48O10/c1-18(11-12-24(38-7)22-9-8-10-23(34)13-22)29-20(3)26-16-32(41-29)30(5,6)15-19(2)31(37,42-32)17-28(36)39-25(21(4)33)14-27(35)40-26/h8-10,13,18-21,24-26,29,33-34,37H,11-12,14-17H2,1-7H3/t18-,19+,20-,21+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKCgamma C1A domain |
J Med Chem 55: 5614-26 (2012)
Article DOI: 10.1021/jm300566h
BindingDB Entry DOI: 10.7270/Q200036J |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM21121

((2S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-N-[(...)Show SMILES CCC(=O)N(C1CCN(CC1)C(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1ccccc1 Show InChI InChI=1S/C39H50N6O6/c1-5-36(48)45(29-14-10-7-11-15-29)30-16-18-44(19-17-30)39(51)34(22-28-12-8-6-9-13-28)43-35(47)24-41-37(49)27(4)42-38(50)33(40)23-32-25(2)20-31(46)21-26(32)3/h6-15,20-21,27,30,33-34,46H,5,16-19,22-24,40H2,1-4H3,(H,41,49)(H,42,50)(H,43,47)/t27-,33+,34+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.380 | -53.8 | n/a | n/a | 0.880 | n/a | n/a | 7.4 | 25 |
University of Arizona at Tucson
| Assay Description
Log IC50 values for each test compound were determined from nonlinear regression analysis of data collected from two independent experiments performe... |
J Med Chem 50: 5528-32 (2007)
Article DOI: 10.1021/jm061465o
BindingDB Entry DOI: 10.7270/Q2D50K8P |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50492535

(CHEMBL2408013)Show SMILES CCC(=O)N(C1CCN(CC1)C(=O)[C@@H](Cc1ccc(Cl)cc1)NC(=O)[C@@H]1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1ccccc1 |r| Show InChI InChI=1S/C44H50ClN5O5/c1-4-41(52)50(34-12-6-5-7-13-34)35-18-20-48(21-19-35)44(55)39(24-30-14-16-33(45)17-15-30)47-42(53)40-25-31-10-8-9-11-32(31)27-49(40)43(54)38(46)26-37-28(2)22-36(51)23-29(37)3/h5-17,22-23,35,38-40,51H,4,18-21,24-27,46H2,1-3H3,(H,47,53)/t38-,39+,40-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in human/mouse HN9.10 cell membranes after 3 hrs by liquid scintillation countin... |
ACS Med Chem Lett 4: 656-659 (2013)
Article DOI: 10.1021/ml400115n
BindingDB Entry DOI: 10.7270/Q2CN76VX |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50105883
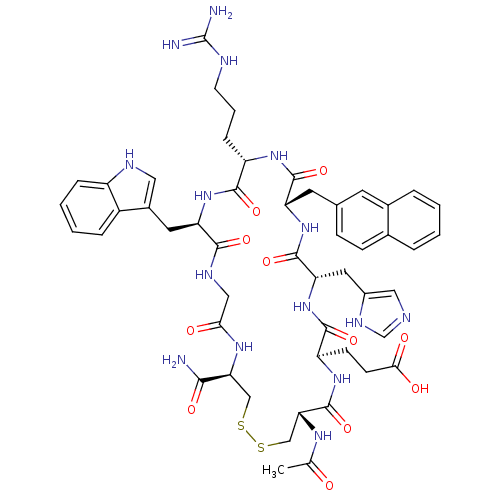
(3-[25-Acetylamino-4-carbamoyl-13-(3-guanidino-prop...)Show SMILES CC(=O)N[C@@H]1CSSC[C@H](NC(=O)CNC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)[C@@H](CCC(O)=O)NC1=O)C(N)=O Show InChI InChI=1S/C51H63N15O11S2/c1-27(67)60-41-25-79-78-24-40(44(52)71)61-42(68)23-58-45(72)38(19-31-21-57-34-10-5-4-9-33(31)34)65-46(73)35(11-6-16-56-51(53)54)62-48(75)37(18-28-12-13-29-7-2-3-8-30(29)17-28)64-49(76)39(20-32-22-55-26-59-32)66-47(74)36(63-50(41)77)14-15-43(69)70/h2-5,7-10,12-13,17,21-22,26,35-41,57H,6,11,14-16,18-20,23-25H2,1H3,(H2,52,71)(H,55,59)(H,58,72)(H,60,67)(H,61,68)(H,62,75)(H,63,77)(H,64,76)(H,65,73)(H,66,74)(H,69,70)(H4,53,54,56)/t35-,36+,37-,38+,39-,40-,41+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against human melanocortin receptor 5 (hMC5R) (concentration of the peptide at 50% specific binding) |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50327943

(Aplysiatoxin | CHEMBL1256416)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cc(O)ccc1Br |r| Show InChI InChI=1S/C32H47BrO10/c1-17(8-11-24(39-7)22-12-21(35)9-10-23(22)33)29-19(3)26-15-32(42-29)30(5,6)14-18(2)31(38,43-32)16-28(37)40-25(20(4)34)13-27(36)41-26/h9-10,12,17-20,24-26,29,34-35,38H,8,11,13-16H2,1-7H3/t17-,18+,19-,20+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKC alpha C1A peptide |
Bioorg Med Chem Lett 20: 6064-6 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.051
BindingDB Entry DOI: 10.7270/Q2FJ2H04 |
More data for this
Ligand-Target Pair | |
Bombesin
(Frog) | BDBM85488

(DPhe6,BetaAla11,Phe13,Nle14-Bn(6-14))Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](N)Cc1ccccc1)C(C)C)C(N)=O Show InChI InChI=1S/C57H76N14O10/c1-6-7-21-42(49(60)73)66-55(79)44(26-36-18-12-9-13-19-36)69-56(80)46(28-38-30-61-31-63-38)68-50(74)33(4)65-57(81)48(32(2)3)71-51(75)34(5)64-54(78)45(27-37-29-62-41-22-15-14-20-39(37)41)70-53(77)43(23-24-47(59)72)67-52(76)40(58)25-35-16-10-8-11-17-35/h8-20,22,29-34,40,42-46,48,62H,6-7,21,23-28,58H2,1-5H3,(H2,59,72)(H2,60,73)(H,61,63)(H,64,78)(H,65,81)(H,66,79)(H,67,76)(H,68,74)(H,69,80)(H,70,77)(H,71,75)/t33-,34-,40+,42-,43-,44-,45-,46-,48-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by PDSP Ki Database
| |
Biochemistry 38: 7307-20 (1999)
Article DOI: 10.1021/bi990204w
BindingDB Entry DOI: 10.7270/Q29022BG |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50327943

(Aplysiatoxin | CHEMBL1256416)Show SMILES CO[C@@H](CC[C@H](C)[C@H]1O[C@@]23C[C@H](OC(=O)C[C@@H](OC(=O)C[C@](O)(O2)[C@H](C)CC3(C)C)[C@@H](C)O)[C@@H]1C)c1cc(O)ccc1Br |r| Show InChI InChI=1S/C32H47BrO10/c1-17(8-11-24(39-7)22-12-21(35)9-10-23(22)33)29-19(3)26-15-32(42-29)30(5,6)14-18(2)31(38,43-32)16-28(37)40-25(20(4)34)13-27(36)41-26/h9-10,12,17-20,24-26,29,34-35,38H,8,11,13-16H2,1-7H3/t17-,18+,19-,20+,24-,25+,26?,29+,31-,32-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University
Curated by ChEMBL
| Assay Description
Inhibition of [3H]PDBu binding to PKC delta C1B peptide |
Bioorg Med Chem Lett 20: 6064-6 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.051
BindingDB Entry DOI: 10.7270/Q2FJ2H04 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50491852

(CHEMBL2387339)Show SMILES CC[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N(C1CCN(CCc2ccccc2)CC1)c1ccccc1 |r| Show InChI InChI=1S/C51H66N8O7/c1-4-43(56-48(63)42(52)31-38-20-22-41(60)23-21-38)49(64)53-33-46(61)55-45(32-37-16-10-6-11-17-37)51(66)57-44(30-35(2)3)50(65)54-34-47(62)59(39-18-12-7-13-19-39)40-25-28-58(29-26-40)27-24-36-14-8-5-9-15-36/h5-23,35,40,42-45,60H,4,24-34,52H2,1-3H3,(H,53,64)(H,54,65)(H,55,61)(H,56,63)(H,57,66)/t42-,43+,44-,45-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cell membranes |
Bioorg Med Chem Lett 23: 3434-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.03.065
BindingDB Entry DOI: 10.7270/Q2T72MCM |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50492538

(CHEMBL2408012)Show SMILES CCC(=O)N(C1CCN(CC1)C(=O)[C@H](Cc1ccc(Cl)cc1)NC(=O)[C@@H]1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1ccccc1 |r| Show InChI InChI=1S/C44H50ClN5O5/c1-4-41(52)50(34-12-6-5-7-13-34)35-18-20-48(21-19-35)44(55)39(24-30-14-16-33(45)17-15-30)47-42(53)40-25-31-10-8-9-11-32(31)27-49(40)43(54)38(46)26-37-28(2)22-36(51)23-29(37)3/h5-17,22-23,35,38-40,51H,4,18-21,24-27,46H2,1-3H3,(H,47,53)/t38-,39-,40-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in human/mouse HN9.10 cell membranes after 3 hrs by liquid scintillation countin... |
ACS Med Chem Lett 4: 656-659 (2013)
Article DOI: 10.1021/ml400115n
BindingDB Entry DOI: 10.7270/Q2CN76VX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data