

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395732 (CHEMBL2164462) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395730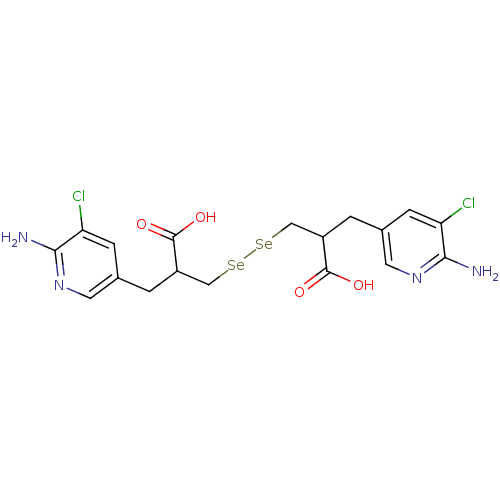 (CHEMBL2164450) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50388528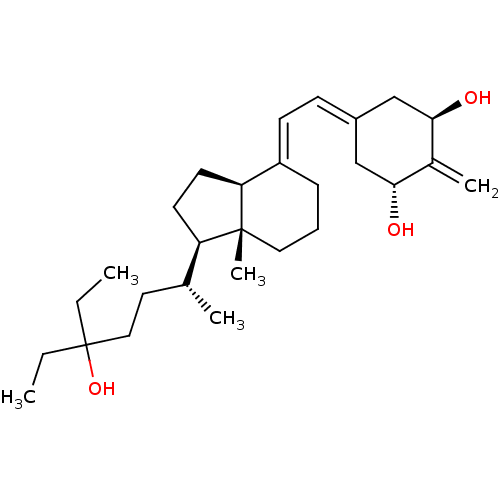 (CHEMBL2058272) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.0290 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]1alpha,25-dihydroxyvitamin D3 from human recombinant GST-tagged vitamin D3 receptor LBD expressed in Escherichia coli BL21 after ... | J Med Chem 55: 4373-81 (2012) Article DOI: 10.1021/jm300230a BindingDB Entry DOI: 10.7270/Q25M66R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50200182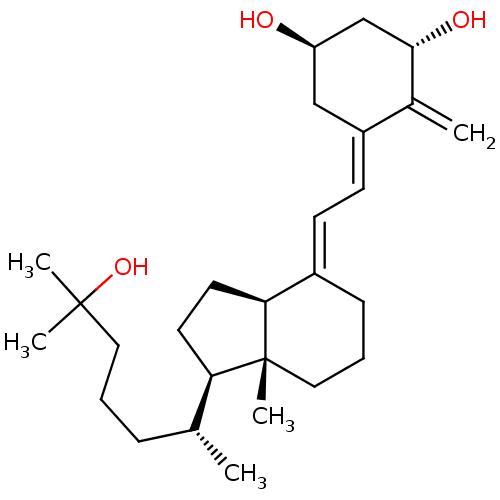 ((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1,25D3 from C-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli BL21 (DE3) after 16 hrs by radioligand... | J Med Chem 61: 6339-6349 (2018) Article DOI: 10.1021/acs.jmedchem.8b00774 BindingDB Entry DOI: 10.7270/Q2CF9SNZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50200182 ((1S,3R,5Z,7E)-9,10-secocholesta-5,7,10(19)-triene-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013605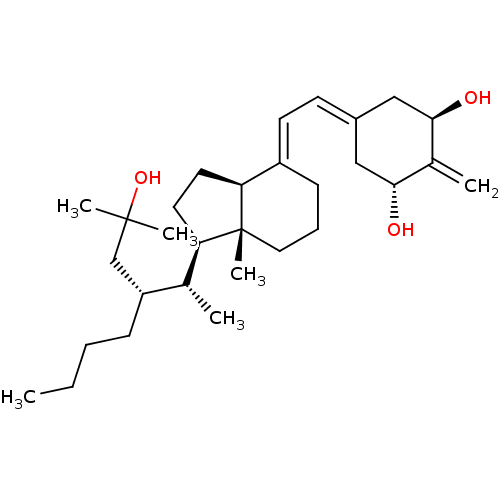 (CHEMBL3264162) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Sus scrofa) | BDBM50395732 (CHEMBL2164462) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of pig pancreatic carboxypeptidase B using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins b... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013603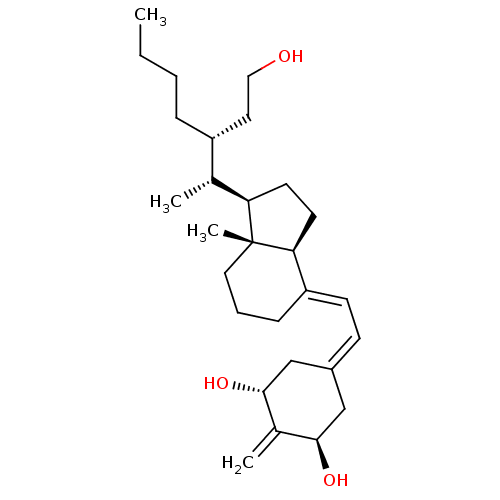 (CHEMBL3264160) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013612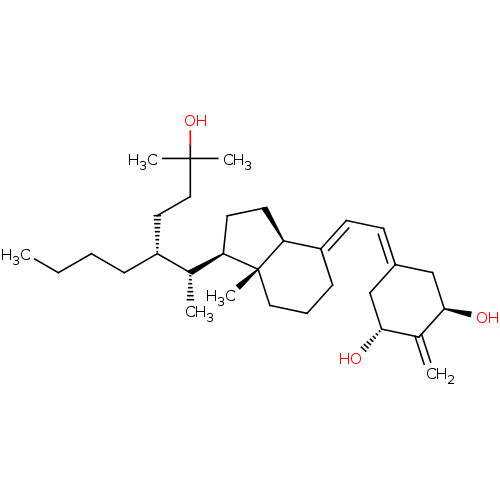 (CHEMBL3264166) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50388529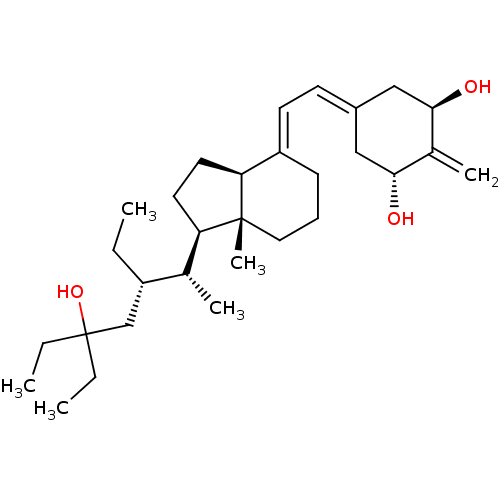 (CHEMBL2058273) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]1alpha,25-dihydroxyvitamin D3 from human recombinant GST-tagged vitamin D3 receptor LBD expressed in Escherichia coli BL21 after ... | J Med Chem 55: 4373-81 (2012) Article DOI: 10.1021/jm300230a BindingDB Entry DOI: 10.7270/Q25M66R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50388530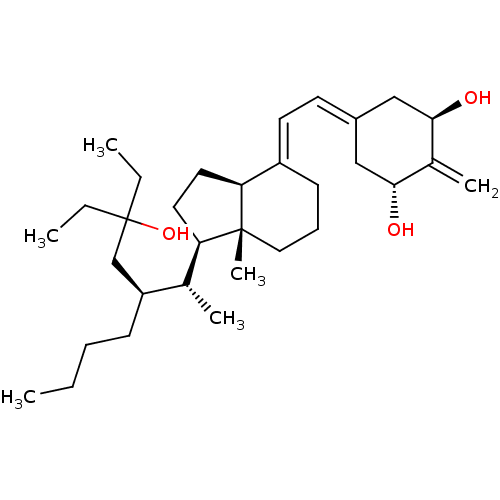 (CHEMBL2058274) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 0.410 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]1alpha,25-dihydroxyvitamin D3 from human recombinant GST-tagged vitamin D3 receptor LBD expressed in Escherichia coli BL21 after ... | J Med Chem 55: 4373-81 (2012) Article DOI: 10.1021/jm300230a BindingDB Entry DOI: 10.7270/Q25M66R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013607 (CHEMBL3264164) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.890 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Sus scrofa) | BDBM50395737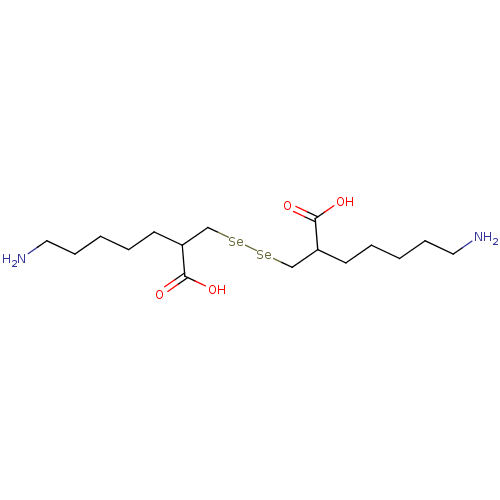 (CHEMBL2164457) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of pig pancreatic carboxypeptidase B using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins b... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Sus scrofa) | BDBM50395735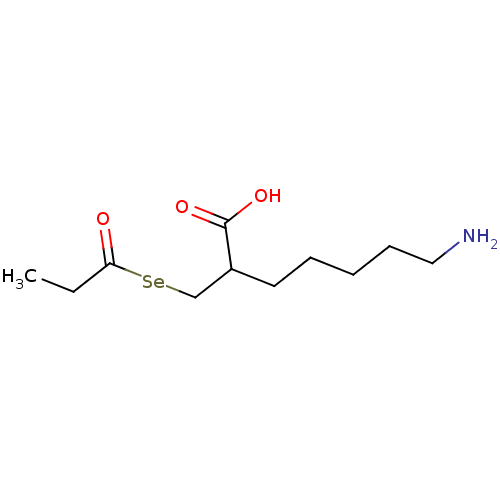 (CHEMBL2164459) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of pig pancreatic carboxypeptidase B using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins b... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395735 (CHEMBL2164459) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50281164 (CHEMBL4176277) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1,25D3 from C-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli BL21 (DE3) after 16 hrs by radioligand... | J Med Chem 61: 6339-6349 (2018) Article DOI: 10.1021/acs.jmedchem.8b00774 BindingDB Entry DOI: 10.7270/Q2CF9SNZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50275212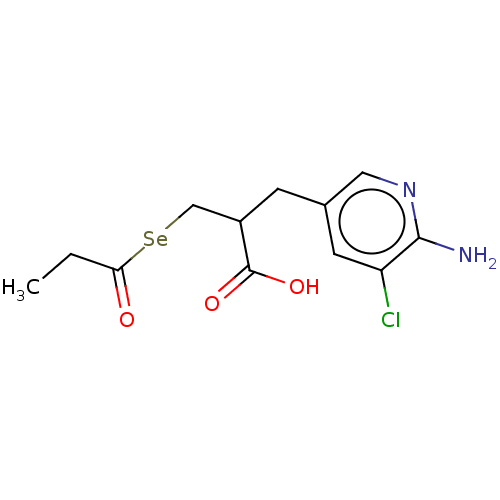 (CHEMBL4127473) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human plasma activated thrombin-activatable fibrinolysis inhibitor after 10 mins in presence of DTT | Bioorg Med Chem Lett 28: 2256-2260 (2018) Article DOI: 10.1016/j.bmcl.2018.05.042 BindingDB Entry DOI: 10.7270/Q25M686N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013606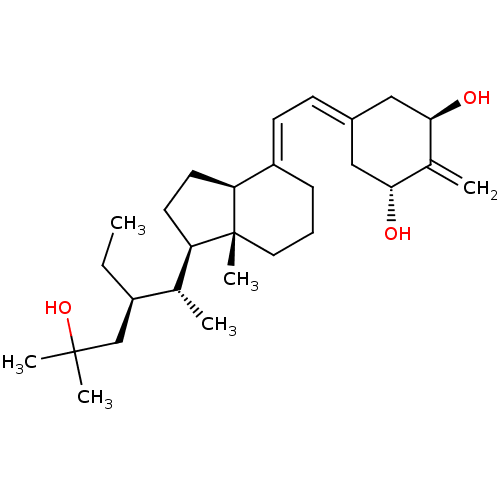 (CHEMBL3264163) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013642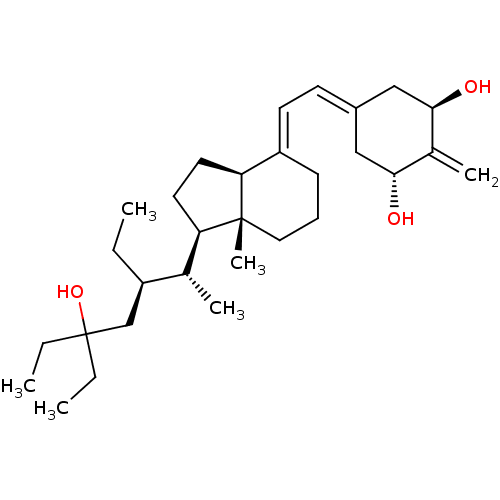 (CHEMBL3264169) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50281165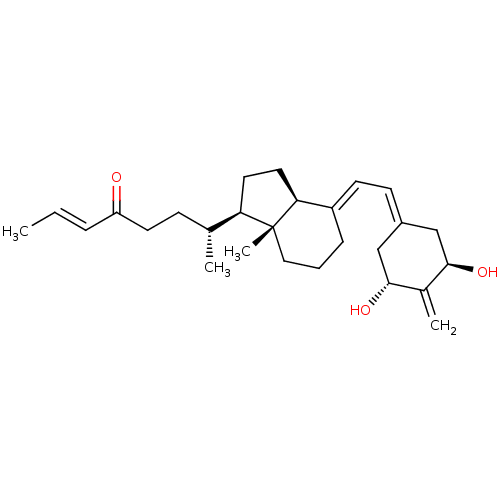 (CHEMBL4176693) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1,25D3 from C-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli BL21 (DE3) after 16 hrs by radioligand... | J Med Chem 61: 6339-6349 (2018) Article DOI: 10.1021/acs.jmedchem.8b00774 BindingDB Entry DOI: 10.7270/Q2CF9SNZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013618 (CHEMBL3264168) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50281163 (CHEMBL4166076) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1,25D3 from C-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli BL21 (DE3) after 16 hrs by radioligand... | J Med Chem 61: 6339-6349 (2018) Article DOI: 10.1021/acs.jmedchem.8b00774 BindingDB Entry DOI: 10.7270/Q2CF9SNZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395737 (CHEMBL2164457) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522329 (CHEMBL4452246) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50281166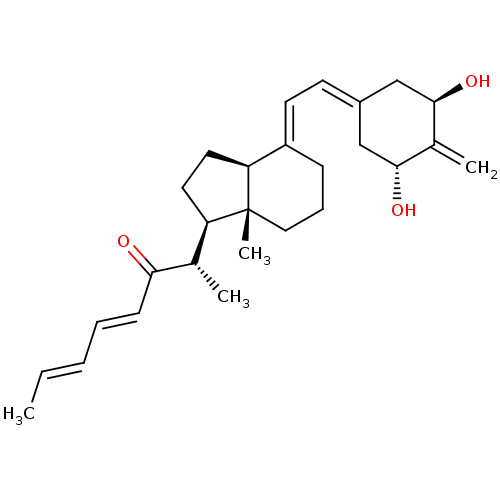 (CHEMBL4161578) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1,25D3 from C-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli BL21 (DE3) after 16 hrs by radioligand... | J Med Chem 61: 6339-6349 (2018) Article DOI: 10.1021/acs.jmedchem.8b00774 BindingDB Entry DOI: 10.7270/Q2CF9SNZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013617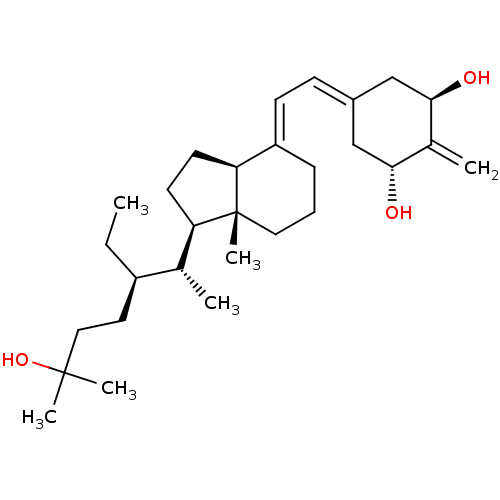 (CHEMBL3264167) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1 (Homo sapiens (Human)) | BDBM50522329 (CHEMBL4452246) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS SAM-PARP domain (1024 to 1327 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522343 (CHEMBL4453770) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522351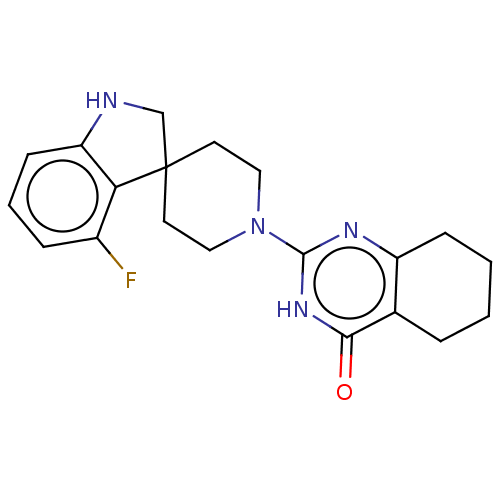 (CHEMBL4454164) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50013604 (CHEMBL3264161) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]-1-alpha,25-dihydroxyvitamin D3 from N-terminal GST-tagged human recombinant VDR LBD expressed in Escherichia coli Rosetta2 (DE3)... | J Med Chem 57: 4351-67 (2014) Article DOI: 10.1021/jm500392t BindingDB Entry DOI: 10.7270/Q24M962V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vitamin D3 receptor (Homo sapiens (Human)) | BDBM50292705 ((25R)-25-Adamantyl-1alpha,25-dihydroxy-2-methylene...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Tokyo Medical and Dental University Curated by ChEMBL | Assay Description Antagonist activity at VDR expressed in COS7 cells assessed as inhibition of 1,25-Dihydroxyvitamin D3-induced response by transient transcription ass... | J Med Chem 51: 5320-9 (2008) Article DOI: 10.1021/jm8004477 BindingDB Entry DOI: 10.7270/Q28C9W2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Sus scrofa) | BDBM50395733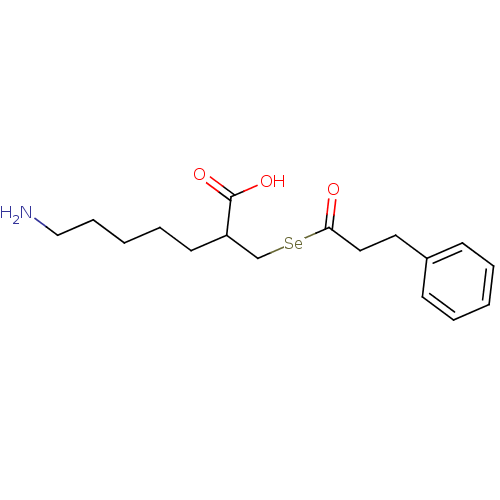 (CHEMBL2164461) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of pig pancreatic carboxypeptidase B using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins b... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Sus scrofa) | BDBM50395734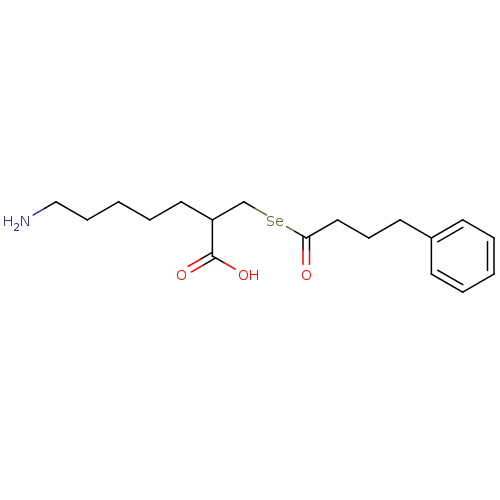 (CHEMBL2164460) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of pig pancreatic carboxypeptidase B using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins b... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522370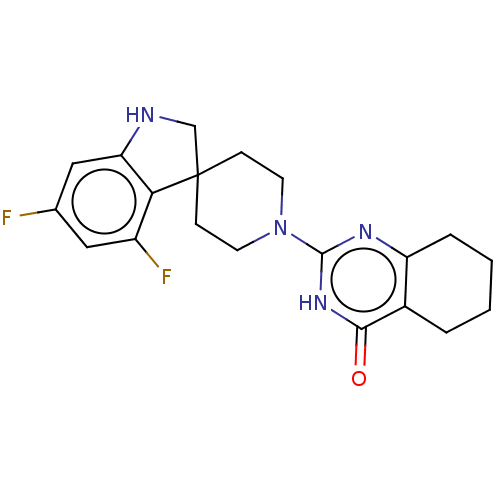 (CHEMBL4533577) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50188594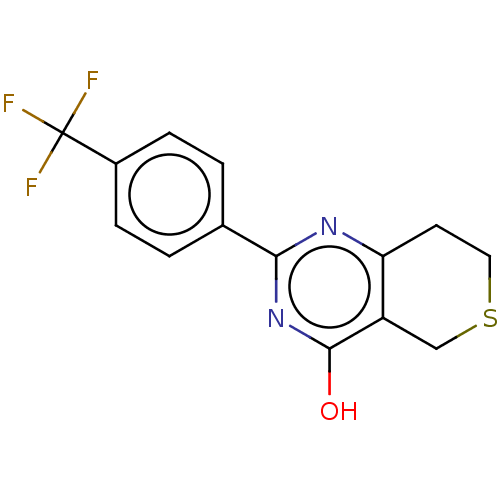 (CHEBI:62878 | CHEMBL1086580) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1 (Homo sapiens (Human)) | BDBM50522343 (CHEMBL4453770) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS SAM-PARP domain (1024 to 1327 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395734 (CHEMBL2164460) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1 (Homo sapiens (Human)) | BDBM50522351 (CHEMBL4454164) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS SAM-PARP domain (1024 to 1327 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1 (Homo sapiens (Human)) | BDBM50522359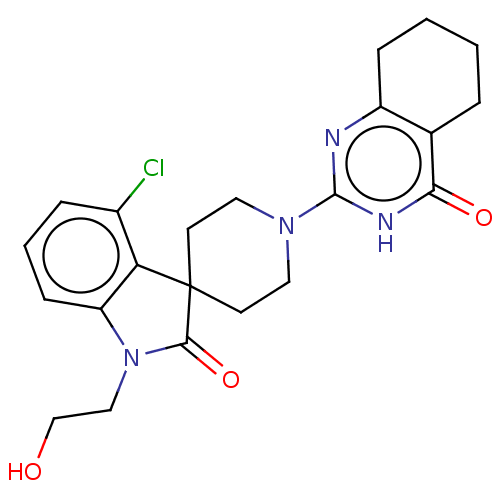 (CHEMBL4483558) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS SAM-PARP domain (1024 to 1327 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1/tankyrase-2 (Homo sapiens (Human)) | BDBM50427989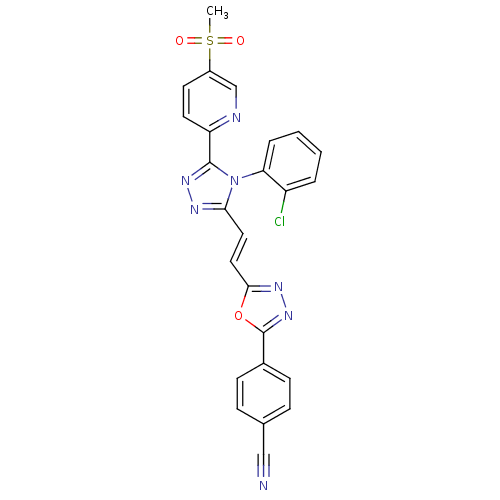 (CHEMBL2325503) | PDB KEGG UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of TNKS/TNKS2 (unknown origin) expressed in HEK293 cells assessed as reduction in Wnt-signaling measured after 24 hrs by TCF-luciferase re... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522355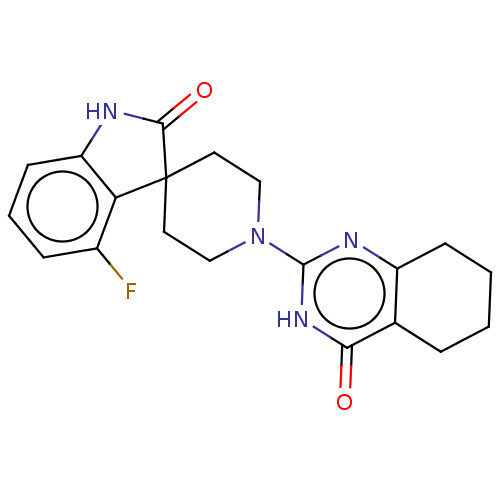 (CHEMBL4449621) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522342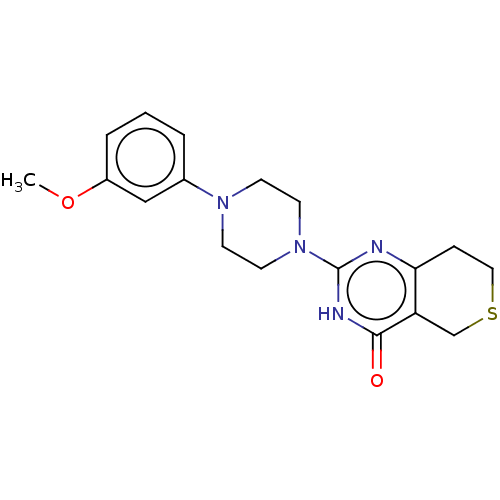 (CHEMBL4538780) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B (Sus scrofa) | BDBM50395728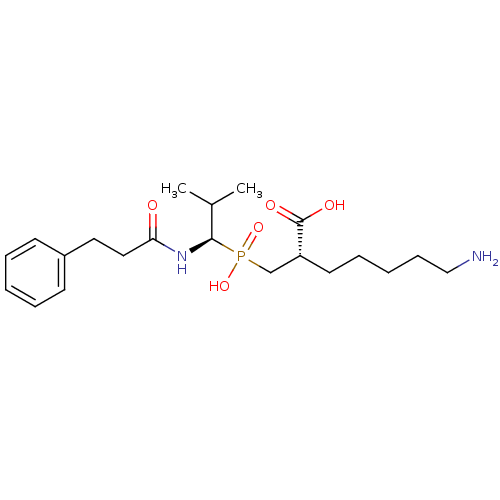 (CHEMBL2164463) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of pig pancreatic carboxypeptidase B using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins b... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522366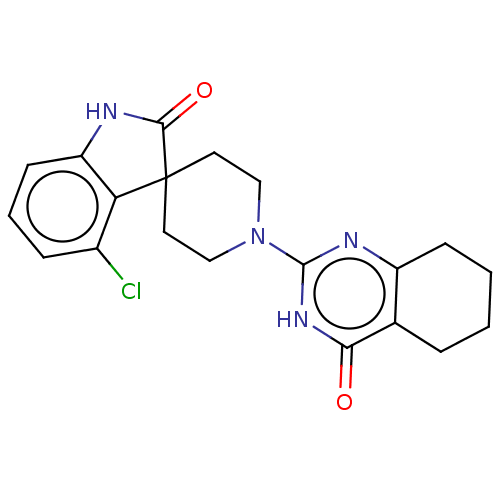 (CHEMBL4441837) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395733 (CHEMBL2164461) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522353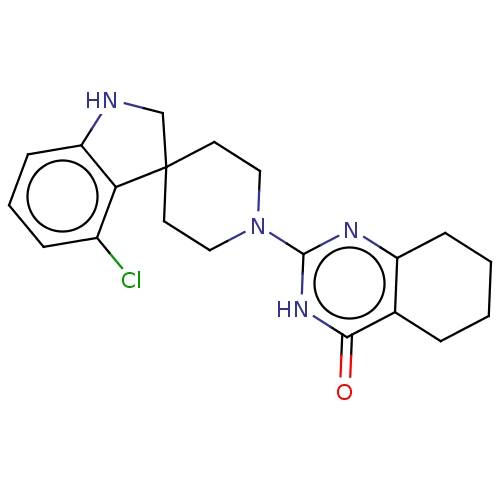 (CHEMBL4441713) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1 (Homo sapiens (Human)) | BDBM50522337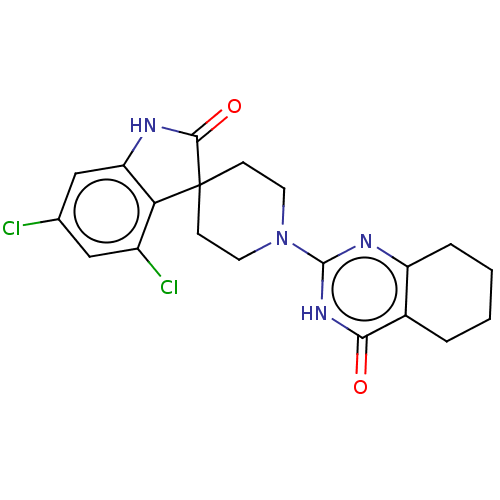 (CHEMBL4469038) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS SAM-PARP domain (1024 to 1327 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-1 (Homo sapiens (Human)) | BDBM50522370 (CHEMBL4533577) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS SAM-PARP domain (1024 to 1327 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxypeptidase B2 (Homo sapiens (Human)) | BDBM50395728 (CHEMBL2164463) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem | Article PubMed | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Showa Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human activated TAFI using Hip-Arg as substrate incubated for 10 mins prior to substrate addition measured after 30 mins by spectrophot... | J Med Chem 55: 7696-705 (2012) Article DOI: 10.1021/jm300735t BindingDB Entry DOI: 10.7270/Q2N58NHV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Poly [ADP-ribose] polymerase tankyrase-2 (Homo sapiens (Human)) | BDBM50522326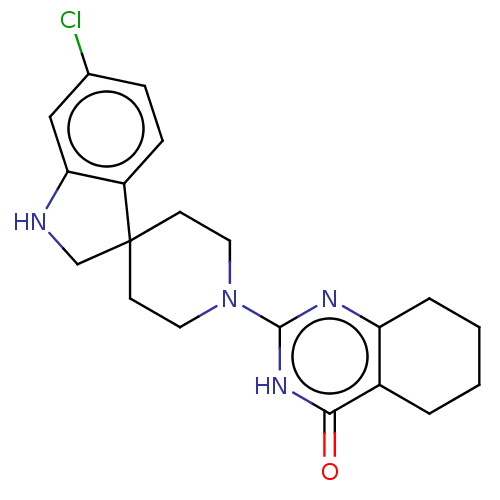 (CHEMBL4469043) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
RIKEN Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal TEV cleavage site-fused/FLAG-poly his-tagged TNKS2 SAM-PARP domain (613 to 1166 residues) expressed in Esc... | J Med Chem 62: 3407-3427 (2019) Article DOI: 10.1021/acs.jmedchem.8b01888 BindingDB Entry DOI: 10.7270/Q2S46WC3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 330 total ) | Next | Last >> |