 Found 9771 hits with Last Name = 'zhang' and Initial = 's'
Found 9771 hits with Last Name = 'zhang' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM50605126

(CHEMBL5187340)Show SMILES COc1cc(ccc1NC(=O)[C@@H]1N[C@@H](CC(C)(C)C)[C@@](CNCC2CCNCC2)([C@H]1c1cccc(Cl)c1F)c1ccc(Cl)cc1F)C(O)=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00095
BindingDB Entry DOI: 10.7270/Q2V69PP3 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50144258

(4-[(8-Benzo[1,3]dioxol-5-ylmethyl-8-aza-bicyclo[3....)Show SMILES [#6]-[#6]-[#7](-[#6]-[#6])-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1ccc2-[#8]-[#6]-[#8]-c2c1)\c1ccccc1 Show InChI InChI=1S/C33H36N2O3/c1-3-34(4-2)33(36)26-13-11-25(12-14-26)32(24-8-6-5-7-9-24)27-19-28-15-16-29(20-27)35(28)21-23-10-17-30-31(18-23)38-22-37-30/h5-14,17-18,28-29H,3-4,15-16,19-22H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor |
Bioorg Med Chem Lett 14: 2109-12 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.051
BindingDB Entry DOI: 10.7270/Q25B01X4 |
More data for this
Ligand-Target Pair | |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM50162774

(ABT-199 | US11420968, Example ABT-199 | Venetoclax)Show SMILES CC1(C)CCC(CN2CCN(CC2)c2ccc(C(=O)NS(=O)(=O)c3ccc(NCC4CCOCC4)c(c3)N(=O)=O)c(Oc3cnc4[nH]ccc4c3)c2)=C(C1)c1ccc(Cl)cc1 |c:57| Show InChI InChI=1S/C45H50ClN7O7S/c1-45(2)15-11-33(39(26-45)31-3-5-34(46)6-4-31)29-51-17-19-52(20-18-51)35-7-9-38(42(24-35)60-36-23-32-12-16-47-43(32)49-28-36)44(54)50-61(57,58)37-8-10-40(41(25-37)53(55)56)48-27-30-13-21-59-22-14-30/h3-10,12,16,23-25,28,30,48H,11,13-15,17-22,26-27,29H2,1-2H3,(H,47,49)(H,50,54) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114327
BindingDB Entry DOI: 10.7270/Q2NP28GR |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50163573

((R)-3-(2,3-Dihydro-benzofuran-5-yl)-2-(5-pyridin-2...)Show SMILES Oc1c2CN([C@@H](c2nc2ccccc12)c1ccc2OCCc2c1)c1ncc(cn1)-c1ccccn1 Show InChI InChI=1S/C28H21N5O2/c34-27-20-5-1-2-7-23(20)32-25-21(27)16-33(26(25)18-8-9-24-17(13-18)10-12-35-24)28-30-14-19(15-31-28)22-6-3-4-11-29-22/h1-9,11,13-15,26H,10,12,16H2,(H,32,34)/t26-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of phosphodiesterase 5 in rat fetal lung fibroblast (RFL-6) cells |
J Med Chem 48: 2126-33 (2005)
Article DOI: 10.1021/jm0401098
BindingDB Entry DOI: 10.7270/Q2WH2QR5 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50144236

(4-[(R)-(S)-8-Aza-bicyclo[3.2.1]oct-(3Z)-ylidene-ph...)Show SMILES CCN(CC)C(=O)c1ccc(cc1)C(=C1\C[C@@H]2CC[C@H](C1)N2)\c1ccccc1 Show InChI InChI=1S/C25H30N2O/c1-3-27(4-2)25(28)20-12-10-19(11-13-20)24(18-8-6-5-7-9-18)21-16-22-14-15-23(17-21)26-22/h5-13,22-23,26H,3-4,14-17H2,1-2H3/b24-21-/t22-,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor |
Bioorg Med Chem Lett 14: 2109-12 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.051
BindingDB Entry DOI: 10.7270/Q25B01X4 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50144229
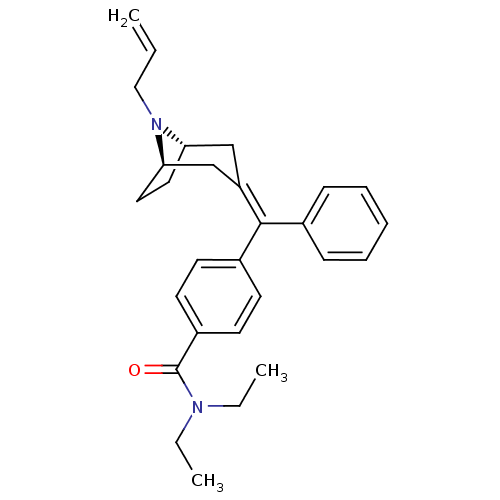
(4-{[(1S,5R)-8-Allyl-8-aza-bicyclo[3.2.1]oct-(3Z)-y...)Show SMILES CCN(CC)C(=O)c1ccc(cc1)C(=C1\C[C@@H]2CC[C@H](C1)N2CC=C)\c1ccccc1 |THB:22:21:14.15.20:17.18| Show InChI InChI=1S/C28H34N2O/c1-4-18-30-25-16-17-26(30)20-24(19-25)27(21-10-8-7-9-11-21)22-12-14-23(15-13-22)28(31)29(5-2)6-3/h4,7-15,25-26H,1,5-6,16-20H2,2-3H3/b27-24-/t25-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Effective concentration against stimulation of [35S]-GTP-gammaS, binding in CHO cells transfected with the human opioid receptor delta 1 |
Bioorg Med Chem Lett 14: 2109-12 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.051
BindingDB Entry DOI: 10.7270/Q25B01X4 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50393719
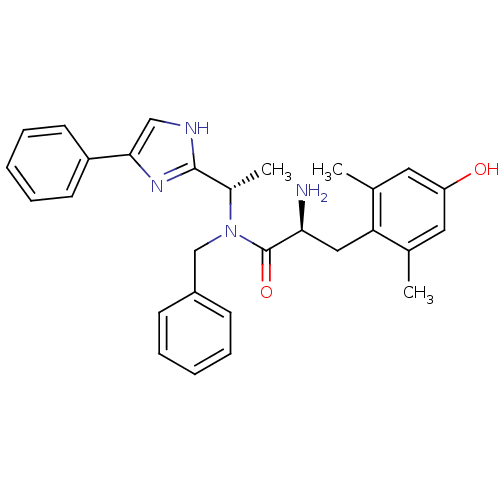
(CHEMBL2159118)Show SMILES C[C@H](N(Cc1ccccc1)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1nc(c[nH]1)-c1ccccc1 |r| Show InChI InChI=1S/C29H32N4O2/c1-19-14-24(34)15-20(2)25(19)16-26(30)29(35)33(18-22-10-6-4-7-11-22)21(3)28-31-17-27(32-28)23-12-8-5-9-13-23/h4-15,17,21,26,34H,16,18,30H2,1-3H3,(H,31,32)/t21-,26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat mu opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50393725
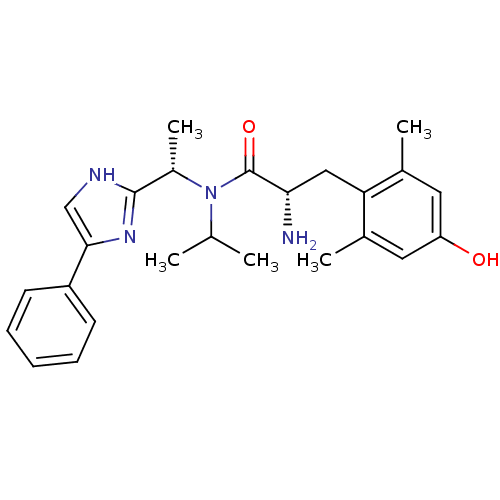
(CHEMBL2159117)Show SMILES CC(C)N([C@@H](C)c1nc(c[nH]1)-c1ccccc1)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C25H32N4O2/c1-15(2)29(18(5)24-27-14-23(28-24)19-9-7-6-8-10-19)25(31)22(26)13-21-16(3)11-20(30)12-17(21)4/h6-12,14-15,18,22,30H,13,26H2,1-5H3,(H,27,28)/t18-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat mu opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278693
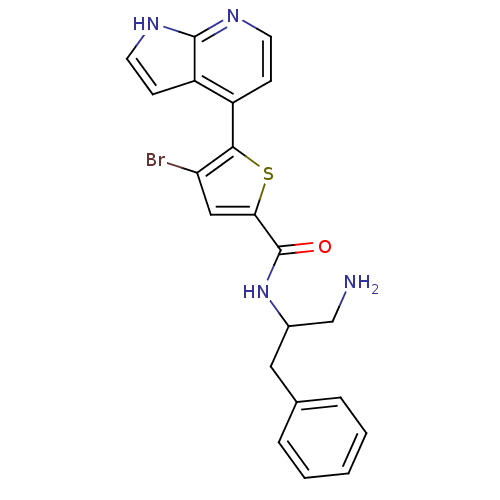
((+/-)-N-(1-amino-3-phenylpropan-2-yl)-4-bromo-5-(1...)Show SMILES NCC(Cc1ccccc1)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 Show InChI InChI=1S/C21H19BrN4OS/c22-17-11-18(21(27)26-14(12-23)10-13-4-2-1-3-5-13)28-19(17)15-6-8-24-20-16(15)7-9-25-20/h1-9,11,14H,10,12,23H2,(H,24,25)(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182939
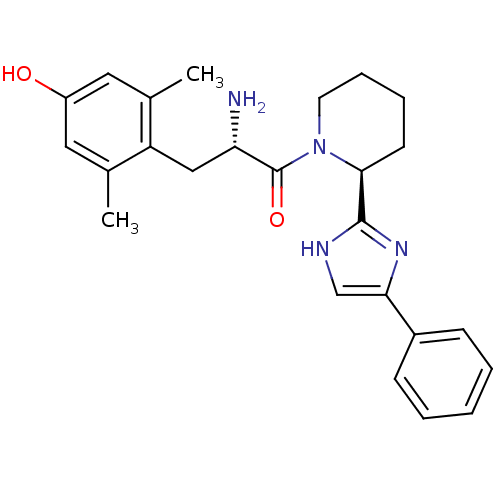
((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-1-((S...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CCCC[C@H]1c1nc(c[nH]1)-c1ccccc1 Show InChI InChI=1S/C25H30N4O2/c1-16-12-19(30)13-17(2)20(16)14-21(26)25(31)29-11-7-6-10-23(29)24-27-15-22(28-24)18-8-4-3-5-9-18/h3-5,8-9,12-13,15,21,23,30H,6-7,10-11,14,26H2,1-2H3,(H,27,28)/t21-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain |
Bioorg Med Chem Lett 16: 2505-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.082
BindingDB Entry DOI: 10.7270/Q29G5MDZ |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278098
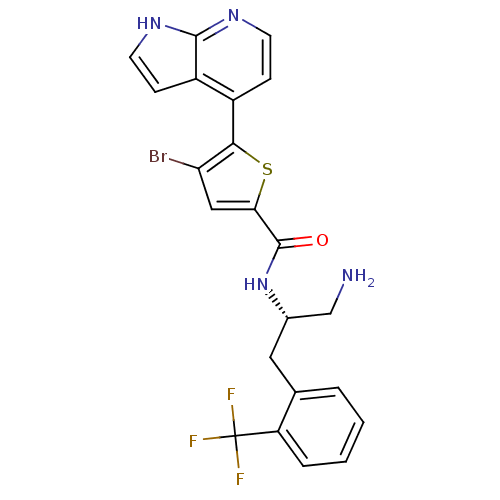
(CHEMBL520788 | N-((S)-1-amino-3-(2-(trifluoromethy...)Show SMILES NC[C@H](Cc1ccccc1C(F)(F)F)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C22H18BrF3N4OS/c23-17-10-18(32-19(17)14-5-7-28-20-15(14)6-8-29-20)21(31)30-13(11-27)9-12-3-1-2-4-16(12)22(24,25)26/h1-8,10,13H,9,11,27H2,(H,28,29)(H,30,31)/t13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182939

((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-1-((S...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CCCC[C@H]1c1nc(c[nH]1)-c1ccccc1 Show InChI InChI=1S/C25H30N4O2/c1-16-12-19(30)13-17(2)20(16)14-21(26)25(31)29-11-7-6-10-23(29)24-27-15-22(28-24)18-8-4-3-5-9-18/h3-5,8-9,12-13,15,21,23,30H,6-7,10-11,14,26H2,1-2H3,(H,27,28)/t21-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat mu opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50277113
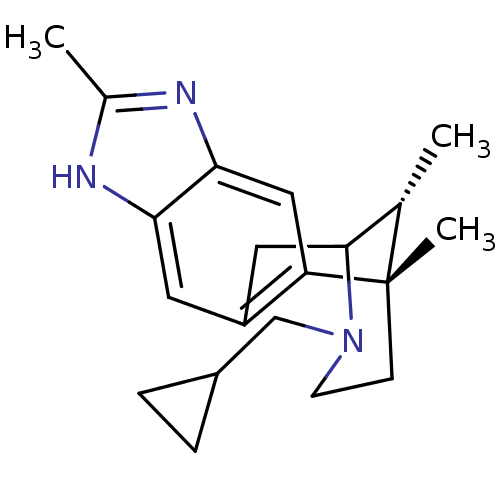
((1S,16R)-13-(cyclopropylmethyl)-1,6,16-trimethyl-5...)Show SMILES C[C@H]1C2Cc3cc4[nH]c(C)nc4cc3[C@@]1(C)CCN2CC1CC1 |r,TLB:12:13:1:18.17.16,19:18:1:4.13.3| Show InChI InChI=1S/C20H27N3/c1-12-19-9-15-8-17-18(22-13(2)21-17)10-16(15)20(12,3)6-7-23(19)11-14-4-5-14/h8,10,12,14,19H,4-7,9,11H2,1-3H3,(H,21,22)/t12-,19?,20-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rensselaer Polytechnic Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 19: 365-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.076
BindingDB Entry DOI: 10.7270/Q2M0458C |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182943
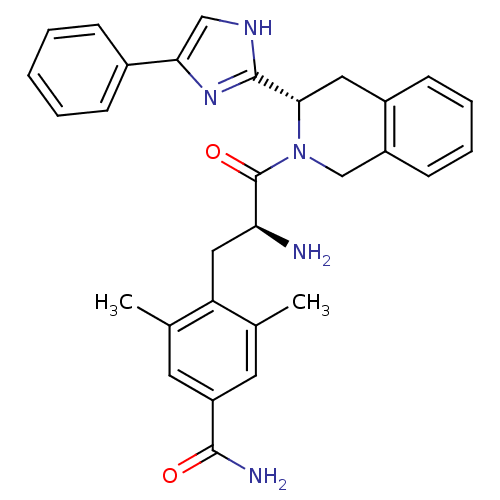
(4-((S)-2-amino-3-oxo-3-((S)-3-(4-phenyl-1H-imidazo...)Show SMILES Cc1cc(cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c[nH]1)-c1ccccc1)C(N)=O Show InChI InChI=1S/C30H31N5O2/c1-18-12-23(28(32)36)13-19(2)24(18)15-25(31)30(37)35-17-22-11-7-6-10-21(22)14-27(35)29-33-16-26(34-29)20-8-4-3-5-9-20/h3-13,16,25,27H,14-15,17,31H2,1-2H3,(H2,32,36)(H,33,34)/t25-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from delta opioid receptor in rat brain |
Bioorg Med Chem Lett 16: 2505-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.082
BindingDB Entry DOI: 10.7270/Q29G5MDZ |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278837
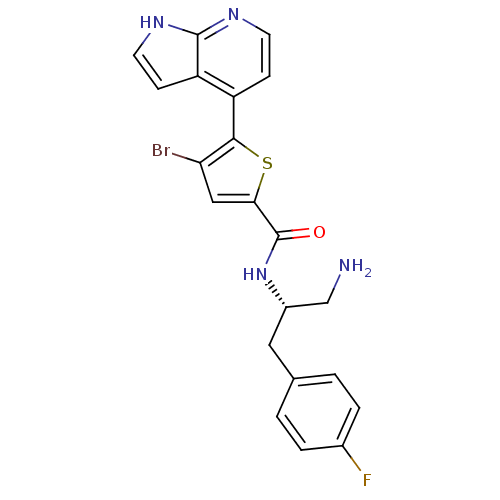
(CHEMBL496690 | N-((S)-1-amino-3-(4-fluorophenyl)pr...)Show SMILES NC[C@H](Cc1ccc(F)cc1)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C21H18BrFN4OS/c22-17-10-18(29-19(17)15-5-7-25-20-16(15)6-8-26-20)21(28)27-14(11-24)9-12-1-3-13(23)4-2-12/h1-8,10,14H,9,11,24H2,(H,25,26)(H,27,28)/t14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182943

(4-((S)-2-amino-3-oxo-3-((S)-3-(4-phenyl-1H-imidazo...)Show SMILES Cc1cc(cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c[nH]1)-c1ccccc1)C(N)=O Show InChI InChI=1S/C30H31N5O2/c1-18-12-23(28(32)36)13-19(2)24(18)15-25(31)30(37)35-17-22-11-7-6-10-21(22)14-27(35)29-33-16-26(34-29)20-8-4-3-5-9-20/h3-13,16,25,27H,14-15,17,31H2,1-2H3,(H2,32,36)(H,33,34)/t25-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat delta opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50277101
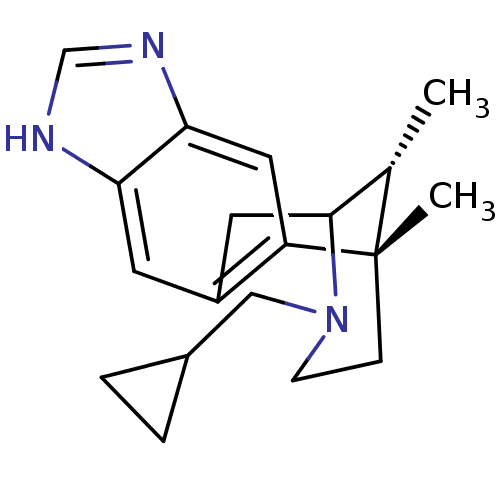
((1S,16R)-13-(cyclopropylmethyl)-1,16-dimethyl-5,7,...)Show SMILES C[C@H]1C2Cc3cc4[nH]cnc4cc3[C@@]1(C)CCN2CC1CC1 |r,TLB:11:12:1:17.16.15,18:17:1:4.12.3| Show InChI InChI=1S/C19H25N3/c1-12-18-8-14-7-16-17(21-11-20-16)9-15(14)19(12,2)5-6-22(18)10-13-3-4-13/h7,9,11-13,18H,3-6,8,10H2,1-2H3,(H,20,21)/t12-,18?,19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rensselaer Polytechnic Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 19: 365-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.076
BindingDB Entry DOI: 10.7270/Q2M0458C |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50001023
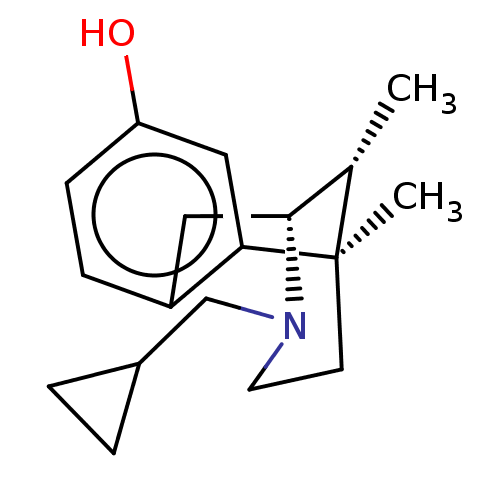
((2R,6R,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,...)Show SMILES C[C@H]1[C@H]2Cc3ccc(O)cc3[C@]1(C)CCN2CC1CC1 |TLB:16:15:1:10.4.3| Show InChI InChI=1S/C18H25NO/c1-12-17-9-14-5-6-15(20)10-16(14)18(12,2)7-8-19(17)11-13-3-4-13/h5-6,10,12-13,17,20H,3-4,7-9,11H2,1-2H3/t12-,17+,18+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rensselaer Polytechnic Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 19: 365-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.076
BindingDB Entry DOI: 10.7270/Q2M0458C |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182954
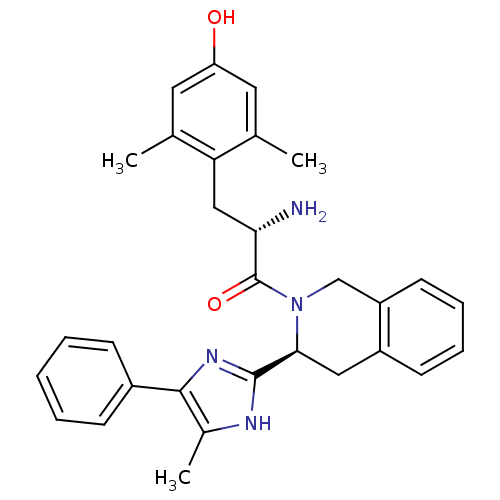
((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-1-((S...)Show SMILES Cc1[nH]c(nc1-c1ccccc1)[C@@H]1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C Show InChI InChI=1S/C30H32N4O2/c1-18-13-24(35)14-19(2)25(18)16-26(31)30(36)34-17-23-12-8-7-11-22(23)15-27(34)29-32-20(3)28(33-29)21-9-5-4-6-10-21/h4-14,26-27,35H,15-17,31H2,1-3H3,(H,32,33)/t26-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from delta opioid receptor in rat brain |
Bioorg Med Chem Lett 16: 2505-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.082
BindingDB Entry DOI: 10.7270/Q29G5MDZ |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182942
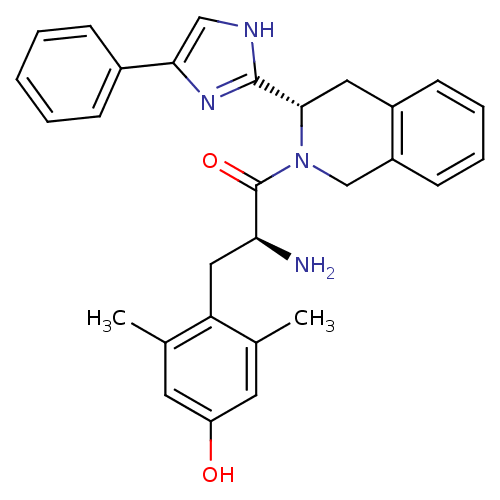
((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-1-((S...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c[nH]1)-c1ccccc1 Show InChI InChI=1S/C29H30N4O2/c1-18-12-23(34)13-19(2)24(18)15-25(30)29(35)33-17-22-11-7-6-10-21(22)14-27(33)28-31-16-26(32-28)20-8-4-3-5-9-20/h3-13,16,25,27,34H,14-15,17,30H2,1-2H3,(H,31,32)/t25-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from delta opioid receptor in rat brain |
Bioorg Med Chem Lett 16: 2505-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.082
BindingDB Entry DOI: 10.7270/Q29G5MDZ |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278099
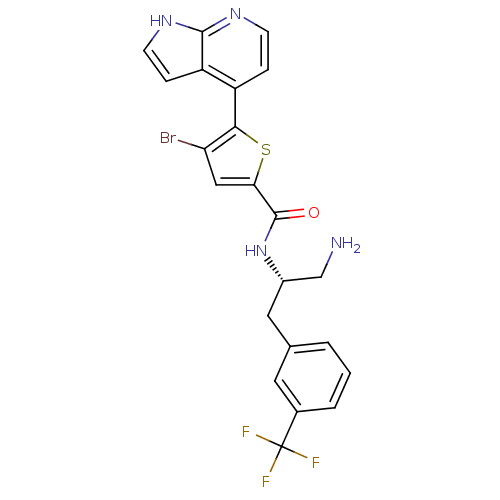
(CHEMBL482536 | N-((S)-1-amino-3-(3-(trifluoromethy...)Show SMILES NC[C@H](Cc1cccc(c1)C(F)(F)F)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C22H18BrF3N4OS/c23-17-10-18(32-19(17)15-4-6-28-20-16(15)5-7-29-20)21(31)30-14(11-27)9-12-2-1-3-13(8-12)22(24,25)26/h1-8,10,14H,9,11,27H2,(H,28,29)(H,30,31)/t14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278099

(CHEMBL482536 | N-((S)-1-amino-3-(3-(trifluoromethy...)Show SMILES NC[C@H](Cc1cccc(c1)C(F)(F)F)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C22H18BrF3N4OS/c23-17-10-18(32-19(17)15-4-6-28-20-16(15)5-7-29-20)21(31)30-14(11-27)9-12-2-1-3-13(8-12)22(24,25)26/h1-8,10,14H,9,11,27H2,(H,28,29)(H,30,31)/t14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50393716
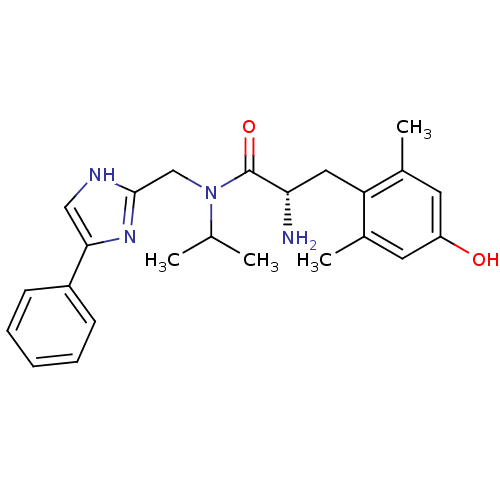
(CHEMBL2159115)Show SMILES CC(C)N(Cc1nc(c[nH]1)-c1ccccc1)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C24H30N4O2/c1-15(2)28(14-23-26-13-22(27-23)18-8-6-5-7-9-18)24(30)21(25)12-20-16(3)10-19(29)11-17(20)4/h5-11,13,15,21,29H,12,14,25H2,1-4H3,(H,26,27)/t21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat mu opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50393717
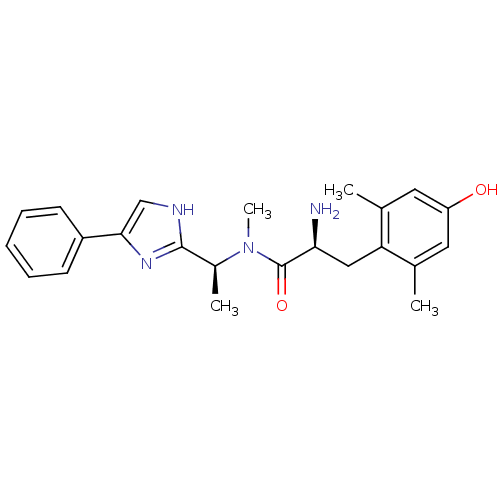
(CHEMBL2159116)Show SMILES C[C@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1nc(c[nH]1)-c1ccccc1 |r| Show InChI InChI=1S/C23H28N4O2/c1-14-10-18(28)11-15(2)19(14)12-20(24)23(29)27(4)16(3)22-25-13-21(26-22)17-8-6-5-7-9-17/h5-11,13,16,20,28H,12,24H2,1-4H3,(H,25,26)/t16-,20-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat mu opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182942

((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl)-1-((S...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c[nH]1)-c1ccccc1 Show InChI InChI=1S/C29H30N4O2/c1-18-12-23(34)13-19(2)24(18)15-25(30)29(35)33-17-22-11-7-6-10-21(22)14-27(33)28-31-16-26(32-28)20-8-4-3-5-9-20/h3-13,16,25,27,34H,14-15,17,30H2,1-2H3,(H,31,32)/t25-,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat delta opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50154039
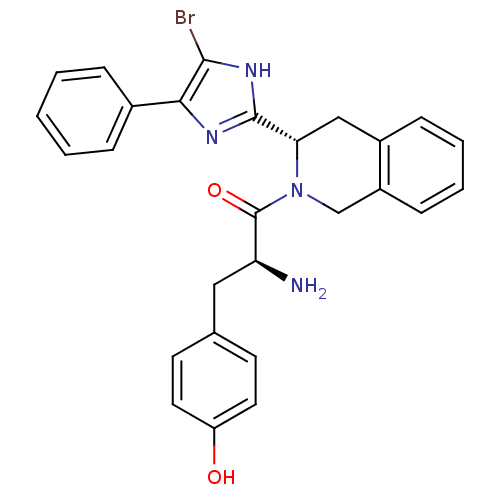
(2-Amino-1-[3-(5-bromo-4-phenyl-1H-imidazol-2-yl)-3...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2C[C@H]1c1nc(c(Br)[nH]1)-c1ccccc1 Show InChI InChI=1S/C27H25BrN4O2/c28-25-24(18-6-2-1-3-7-18)30-26(31-25)23-15-19-8-4-5-9-20(19)16-32(23)27(34)22(29)14-17-10-12-21(33)13-11-17/h1-13,22-23,33H,14-16,29H2,(H,30,31)/t22-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for delta opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50163576
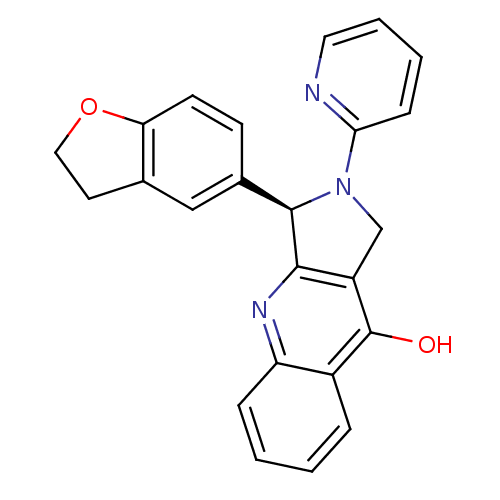
((R)-3-(2,3-Dihydro-benzofuran-5-yl)-2-pyridin-2-yl...)Show SMILES Oc1c2CN([C@@H](c2nc2ccccc12)c1ccc2OCCc2c1)c1ccccn1 Show InChI InChI=1S/C24H19N3O2/c28-24-17-5-1-2-6-19(17)26-22-18(24)14-27(21-7-3-4-11-25-21)23(22)16-8-9-20-15(13-16)10-12-29-20/h1-9,11,13,23H,10,12,14H2,(H,26,28)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of phosphodiesterase 5 in rat fetal lung fibroblast (RFL-6) cells |
J Med Chem 48: 2126-33 (2005)
Article DOI: 10.1021/jm0401098
BindingDB Entry DOI: 10.7270/Q2WH2QR5 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182946
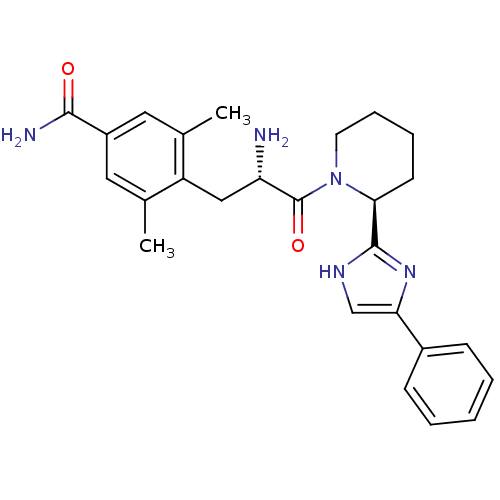
(4-((S)-2-amino-3-oxo-3-((S)-2-(4-phenyl-1H-imidazo...)Show SMILES Cc1cc(cc(C)c1C[C@H](N)C(=O)N1CCCC[C@H]1c1nc(c[nH]1)-c1ccccc1)C(N)=O Show InChI InChI=1S/C26H31N5O2/c1-16-12-19(24(28)32)13-17(2)20(16)14-21(27)26(33)31-11-7-6-10-23(31)25-29-15-22(30-25)18-8-4-3-5-9-18/h3-5,8-9,12-13,15,21,23H,6-7,10-11,14,27H2,1-2H3,(H2,28,32)(H,29,30)/t21-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development, L.L.C.
Curated by ChEMBL
| Assay Description
Binding affinity to rat mu opioid receptor |
Bioorg Med Chem Lett 22: 4869-72 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.042
BindingDB Entry DOI: 10.7270/Q2F76DP8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50182946

(4-((S)-2-amino-3-oxo-3-((S)-2-(4-phenyl-1H-imidazo...)Show SMILES Cc1cc(cc(C)c1C[C@H](N)C(=O)N1CCCC[C@H]1c1nc(c[nH]1)-c1ccccc1)C(N)=O Show InChI InChI=1S/C26H31N5O2/c1-16-12-19(24(28)32)13-17(2)20(16)14-21(27)26(33)31-11-7-6-10-23(31)25-29-15-22(30-25)18-8-4-3-5-9-18/h3-5,8-9,12-13,15,21,23H,6-7,10-11,14,27H2,1-2H3,(H2,28,32)(H,29,30)/t21-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain |
Bioorg Med Chem Lett 16: 2505-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.082
BindingDB Entry DOI: 10.7270/Q29G5MDZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50144281
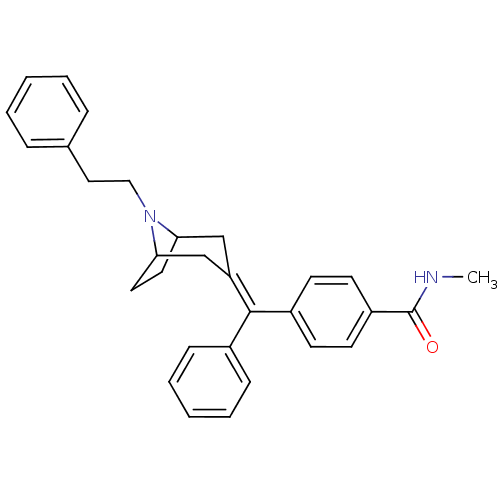
(CHEMBL63309 | N-Methyl-4-[(8-phenethyl-8-aza-bicyc...)Show SMILES [#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1\[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-[#6]-c1ccccc1)\c1ccccc1 Show InChI InChI=1S/C30H32N2O/c1-31-30(33)25-14-12-24(13-15-25)29(23-10-6-3-7-11-23)26-20-27-16-17-28(21-26)32(27)19-18-22-8-4-2-5-9-22/h2-15,27-28H,16-21H2,1H3,(H,31,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards opioid receptor mu 1 |
Bioorg Med Chem Lett 14: 2113-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.052
BindingDB Entry DOI: 10.7270/Q21J997Q |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50163581

(2-[2,3'']Bipyridinyl-6''-yl-3-(2,3-dihydro-benzofu...)Show SMILES Oc1c2CN(C(c2nc2ccccc12)c1ccc2OCCc2c1)c1ccc(cn1)-c1ccccn1 Show InChI InChI=1S/C29H22N4O2/c34-29-21-5-1-2-7-24(21)32-27-22(29)17-33(28(27)19-8-10-25-18(15-19)12-14-35-25)26-11-9-20(16-31-26)23-6-3-4-13-30-23/h1-11,13,15-16,28H,12,14,17H2,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of phosphodiesterase 5 in rat fetal lung fibroblast (RFL-6) cells |
J Med Chem 48: 2126-33 (2005)
Article DOI: 10.1021/jm0401098
BindingDB Entry DOI: 10.7270/Q2WH2QR5 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50144284

(CHEMBL68412 | N-Ethyl-4-[(3-hydroxy-phenyl)-(8-thi...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1\[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-c1cccs1)\c1cccc(-[#8])c1 Show InChI InChI=1S/C28H30N2O2S/c1-2-29-28(32)20-10-8-19(9-11-20)27(21-5-3-6-25(31)17-21)22-15-23-12-13-24(16-22)30(23)18-26-7-4-14-33-26/h3-11,14,17,23-24,31H,2,12-13,15-16,18H2,1H3,(H,29,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.152 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards opioid receptor delta 1 |
Bioorg Med Chem Lett 14: 2113-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.052
BindingDB Entry DOI: 10.7270/Q21J997Q |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50017698
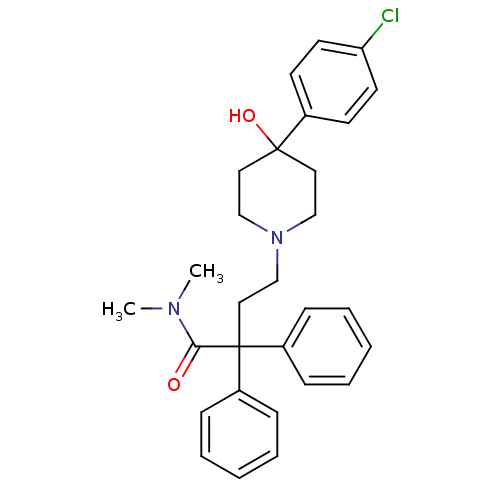
(4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...)Show SMILES CN(C)C(=O)C(CCN1CCC(O)(CC1)c1ccc(Cl)cc1)(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C29H33ClN2O2/c1-31(2)27(33)29(24-9-5-3-6-10-24,25-11-7-4-8-12-25)19-22-32-20-17-28(34,18-21-32)23-13-15-26(30)16-14-23/h3-16,34H,17-22H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Binding affinity for Mu opioid receptor of rat brain |
J Med Chem 47: 5009-20 (2004)
Article DOI: 10.1021/jm030548r
BindingDB Entry DOI: 10.7270/Q2H131GF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50001023

((2R,6R,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,...)Show SMILES C[C@H]1[C@H]2Cc3ccc(O)cc3[C@]1(C)CCN2CC1CC1 |TLB:16:15:1:10.4.3| Show InChI InChI=1S/C18H25NO/c1-12-17-9-14-5-6-15(20)10-16(14)18(12,2)7-8-19(17)11-13-3-4-13/h5-6,10,12-13,17,20H,3-4,7-9,11H2,1-2H3/t12-,17+,18+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rensselaer Polytechnic Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 19: 365-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.076
BindingDB Entry DOI: 10.7270/Q2M0458C |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50545673
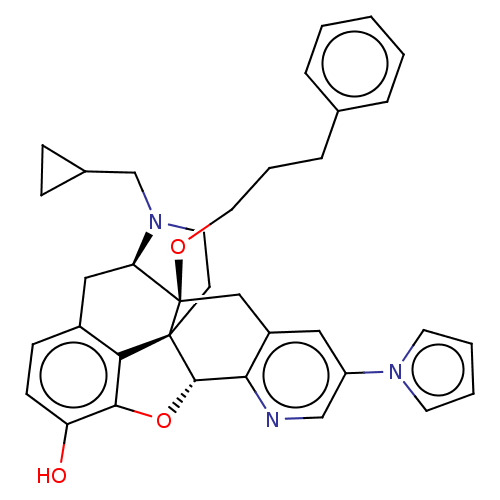
(CHEMBL4634079)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC6CC6)CC[C@@]14[C@]5(Cc1cc(cnc21)-n1cccc1)OCCCc1ccccc1)ccc3O |r,THB:10:9:17:4.5.6| Show InChI InChI=1S/C36H37N3O3/c40-29-13-12-26-20-30-36(41-18-6-9-24-7-2-1-3-8-24)21-27-19-28(38-15-4-5-16-38)22-37-32(27)34-35(36,31(26)33(29)42-34)14-17-39(30)23-25-10-11-25/h1-5,7-8,12-13,15-16,19,22,25,30,34,40H,6,9-11,14,17-18,20-21,23H2/t30-,34+,35+,36-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.169 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham
Curated by ChEMBL
| Assay Description
Displacement of [3H]-diprenorphine from human delta opioid receptor expressed in CHO cell membranes incubated for 1 hr by competition radioligand bin... |
J Med Chem 63: 7663-7694 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00503
BindingDB Entry DOI: 10.7270/Q2B56P91 |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50163570

(2-[5-(3-Benzyl-3H-imidazol-4-yl)-pyridin-2-yl]-3-(...)Show SMILES Oc1c2CN(C(c2nc2ccccc12)c1ccc2OCCc2c1)c1ccc(cn1)-c1cncn1Cc1ccccc1 Show InChI InChI=1S/C34H27N5O2/c40-34-26-8-4-5-9-28(26)37-32-27(34)20-39(33(32)24-10-12-30-23(16-24)14-15-41-30)31-13-11-25(17-36-31)29-18-35-21-38(29)19-22-6-2-1-3-7-22/h1-13,16-18,21,33H,14-15,19-20H2,(H,37,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of phosphodiesterase 5 in rat fetal lung fibroblast (RFL-6) cells |
J Med Chem 48: 2126-33 (2005)
Article DOI: 10.1021/jm0401098
BindingDB Entry DOI: 10.7270/Q2WH2QR5 |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50163577

(3-Benzofuran-5-yl-2-(5-pyridin-2-yl-pyrimidin-2-yl...)Show SMILES Oc1c2CN(C(c2nc2ccccc12)c1ccc2occc2c1)c1ncc(cn1)-c1ccccn1 Show InChI InChI=1S/C28H19N5O2/c34-27-20-5-1-2-7-23(20)32-25-21(27)16-33(26(25)18-8-9-24-17(13-18)10-12-35-24)28-30-14-19(15-31-28)22-6-3-4-11-29-22/h1-15,26H,16H2,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of phosphodiesterase 5 in rat fetal lung fibroblast (RFL-6) cells |
J Med Chem 48: 2126-33 (2005)
Article DOI: 10.1021/jm0401098
BindingDB Entry DOI: 10.7270/Q2WH2QR5 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50155124
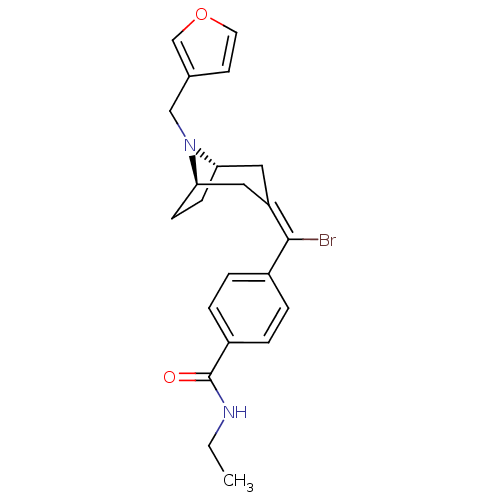
(4-{Bromo-[(1S,5R)-8-furan-3-ylmethyl-8-aza-bicyclo...)Show SMILES CCNC(=O)c1ccc(cc1)C(\Br)=C1\C[C@@H]2CC[C@H](C1)N2Cc1ccoc1 |THB:21:20:13.14.19:17.16| Show InChI InChI=1S/C22H25BrN2O2/c1-2-24-22(26)17-5-3-16(4-6-17)21(23)18-11-19-7-8-20(12-18)25(19)13-15-9-10-27-14-15/h3-6,9-10,14,19-20H,2,7-8,11-13H2,1H3,(H,24,26)/b21-18-/t19-,20+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Binding affinity against mu opioid receptor in mouse hot plate test |
Bioorg Med Chem Lett 14: 5493-8 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.004
BindingDB Entry DOI: 10.7270/Q2X34WZV |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50088712

(4-{[(1S,3S,5R)-8-(Benzo[1,3]dioxole-5-carbonyl)-8-...)Show SMILES CCN(CC)C(=O)c1ccc(cc1)N([C@H]1C[C@@H]2CC[C@H](C1)N2C(=O)c1ccc2OCOc2c1)c1ccccc1 |THB:22:21:14.15.20:18.17| Show InChI InChI=1S/C32H35N3O4/c1-3-33(4-2)31(36)22-10-13-25(14-11-22)34(24-8-6-5-7-9-24)28-19-26-15-16-27(20-28)35(26)32(37)23-12-17-29-30(18-23)39-21-38-29/h5-14,17-18,26-28H,3-4,15-16,19-21H2,1-2H3/t26-,27+,28- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-DPDPE radioligand binding to rat opioid receptor delta 1 site from rat brain membranes |
Bioorg Med Chem Lett 10: 1109-11 (2000)
BindingDB Entry DOI: 10.7270/Q2X0668B |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50316183
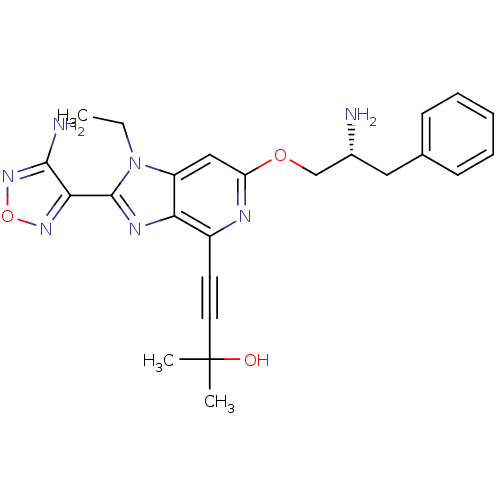
(4-(2-(4-amino-1,2,5-oxadiazol-3-yl)-6-((R)-2-amino...)Show SMILES CCn1c(nc2c(nc(OC[C@H](N)Cc3ccccc3)cc12)C#CC(C)(C)O)-c1nonc1N |r| Show InChI InChI=1S/C24H27N7O3/c1-4-31-18-13-19(33-14-16(25)12-15-8-6-5-7-9-15)27-17(10-11-24(2,3)32)20(18)28-23(31)21-22(26)30-34-29-21/h5-9,13,16,32H,4,12,14,25H2,1-3H3,(H2,26,30)/t16-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT |
Bioorg Med Chem Lett 19: 1508-11 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.002
BindingDB Entry DOI: 10.7270/Q2XK8FP7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50316184

(4-(2-(4-amino-1,2,5-oxadiazol-3-yl)-6-((S)-3-amino...)Show SMILES CCn1c(nc2c(nc(O[C@@H](CCN)c3ccccc3)cc12)C#CC(C)(C)O)-c1nonc1N |r| Show InChI InChI=1S/C24H27N7O3/c1-4-31-17-14-19(33-18(11-13-25)15-8-6-5-7-9-15)27-16(10-12-24(2,3)32)20(17)28-23(31)21-22(26)30-34-29-21/h5-9,14,18,32H,4,11,13,25H2,1-3H3,(H2,26,30)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT |
Bioorg Med Chem Lett 19: 1508-11 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.002
BindingDB Entry DOI: 10.7270/Q2XK8FP7 |
More data for this
Ligand-Target Pair | |
Sigma non-opioid intracellular receptor 1
(Cavia porcellus (Guinea pig)) | BDBM50001019
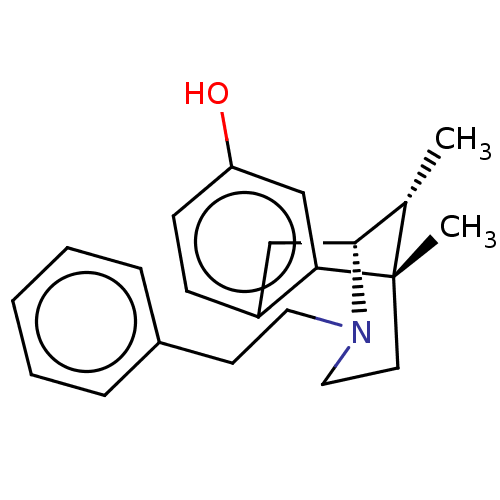
(6,11-Dimethyl-3-phenethyl-1,2,3,4,5,6-hexahydro-2,...)Show SMILES C[C@H]1[C@H]2Cc3ccc(O)cc3[C@@]1(C)CCN2CCc1ccccc1 |TLB:16:15:1:10.4.3| Show InChI InChI=1S/C22H27NO/c1-16-21-14-18-8-9-19(24)15-20(18)22(16,2)11-13-23(21)12-10-17-6-4-3-5-7-17/h3-9,15-16,21,24H,10-14H2,1-2H3/t16-,21+,22-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114649
BindingDB Entry DOI: 10.7270/Q2F47T3Z |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50370143
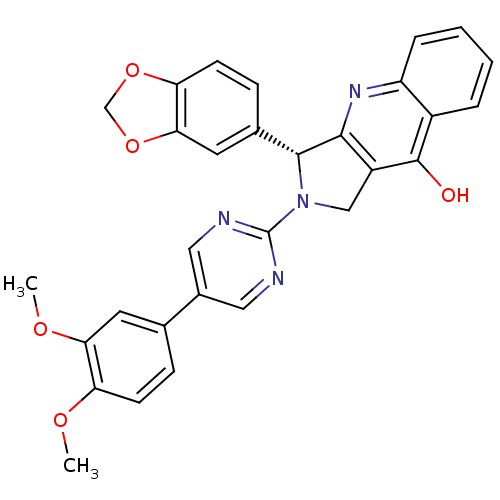
(CHEMBL1744059)Show SMILES COc1ccc(cc1OC)-c1cnc(nc1)N1Cc2c(nc3ccccc3c2O)[C@H]1c1ccc2OCOc2c1 |r| Show InChI InChI=1S/C30H24N4O5/c1-36-23-9-7-17(11-25(23)37-2)19-13-31-30(32-14-19)34-15-21-27(33-22-6-4-3-5-20(22)29(21)35)28(34)18-8-10-24-26(12-18)39-16-38-24/h3-14,28H,15-16H2,1-2H3,(H,33,35)/t28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
Phosphodiesterase 5 activity of human corpus cavernosum |
J Med Chem 45: 4094-6 (2002)
BindingDB Entry DOI: 10.7270/Q2R49RGM |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50118249
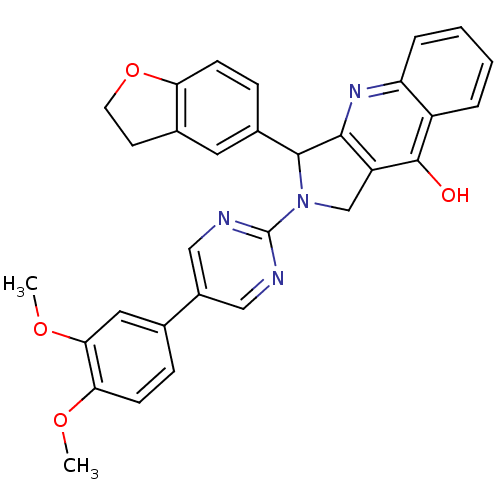
(3-(2,3-Dihydro-benzofuran-5-yl)-2-[5-(3,4-dimethox...)Show SMILES COc1ccc(cc1OC)-c1cnc(nc1)N1Cc2c(nc3ccccc3c2O)C1c1ccc2OCCc2c1 Show InChI InChI=1S/C31H26N4O4/c1-37-26-10-7-18(14-27(26)38-2)21-15-32-31(33-16-21)35-17-23-28(34-24-6-4-3-5-22(24)30(23)36)29(35)20-8-9-25-19(13-20)11-12-39-25/h3-10,13-16,29H,11-12,17H2,1-2H3,(H,34,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
Phosphodiesterase 5 activity of human corpus cavernosum |
J Med Chem 45: 4094-6 (2002)
BindingDB Entry DOI: 10.7270/Q2R49RGM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50581486

(CHEMBL5082165) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]DAMGO from MOR in Sprague-Dawley rat brain membranes measured after 90 mins by scintillation counting method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113879
BindingDB Entry DOI: 10.7270/Q26W9FZS |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Rattus norvegicus) | BDBM50163578

(3-(2,3-Dihydro-benzofuran-5-yl)-2-(5-pyridin-4-yl-...)Show SMILES Oc1c2CN(C(c2nc2ccccc12)c1ccc2OCCc2c1)c1ncc(cn1)-c1ccncc1 Show InChI InChI=1S/C28H21N5O2/c34-27-21-3-1-2-4-23(21)32-25-22(27)16-33(26(25)19-5-6-24-18(13-19)9-12-35-24)28-30-14-20(15-31-28)17-7-10-29-11-8-17/h1-8,10-11,13-15,26H,9,12,16H2,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of phosphodiesterase 5 in rat fetal lung fibroblast (RFL-6) cells |
J Med Chem 48: 2126-33 (2005)
Article DOI: 10.1021/jm0401098
BindingDB Entry DOI: 10.7270/Q2WH2QR5 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50144275
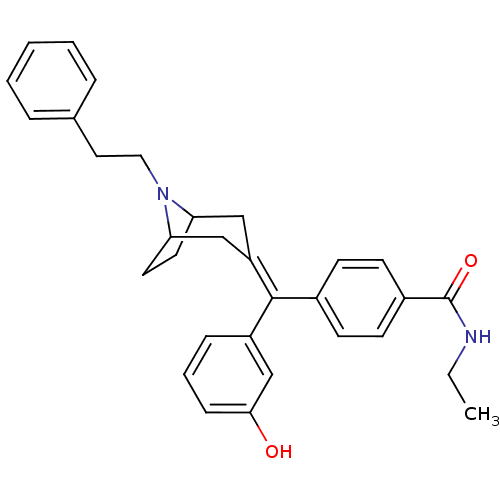
(CHEMBL419621 | N-Ethyl-4-[(3-hydroxy-phenyl)-(8-ph...)Show SMILES [#6]-[#6]-[#7]-[#6](=O)-c1ccc(cc1)-[#6](=[#6]-1/[#6]-[#6]-2-[#6]-[#6]-[#6](-[#6]-1)-[#7]-2-[#6]-[#6]-c1ccccc1)\c1cccc(-[#8])c1 Show InChI InChI=1S/C31H34N2O2/c1-2-32-31(35)24-13-11-23(12-14-24)30(25-9-6-10-29(34)21-25)26-19-27-15-16-28(20-26)33(27)18-17-22-7-4-3-5-8-22/h3-14,21,27-28,34H,2,15-20H2,1H3,(H,32,35) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.222 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards opioid receptor mu 1 |
Bioorg Med Chem Lett 14: 2113-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.052
BindingDB Entry DOI: 10.7270/Q21J997Q |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50277118
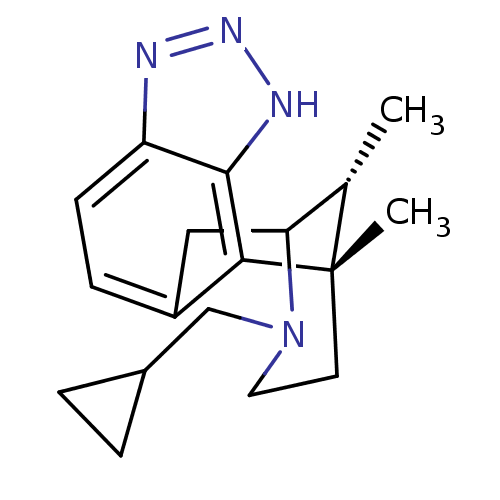
((1S,16R)-13-(cyclopropylmethyl)-1,16-dimethyl-4,5,...)Show SMILES C[C@H]1C2Cc3ccc4nn[nH]c4c3[C@@]1(C)CCN2CC1CC1 |r,TLB:11:12:1:17.16.15,18:17:1:4.12.3| Show InChI InChI=1S/C18H24N4/c1-11-15-9-13-5-6-14-17(20-21-19-14)16(13)18(11,2)7-8-22(15)10-12-3-4-12/h5-6,11-12,15H,3-4,7-10H2,1-2H3,(H,19,20,21)/t11-,15?,18-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rensselaer Polytechnic Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 19: 365-8 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.076
BindingDB Entry DOI: 10.7270/Q2M0458C |
More data for this
Ligand-Target Pair | |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50118248
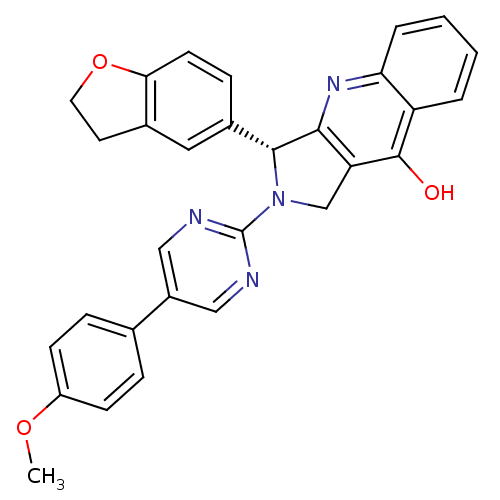
(3-(2,3-Dihydro-benzofuran-5-yl)-2-[5-(4-methoxy-ph...)Show SMILES COc1ccc(cc1)-c1cnc(nc1)N1Cc2c(nc3ccccc3c2O)[C@H]1c1ccc2OCCc2c1 Show InChI InChI=1S/C30H24N4O3/c1-36-22-9-6-18(7-10-22)21-15-31-30(32-16-21)34-17-24-27(33-25-5-3-2-4-23(25)29(24)35)28(34)20-8-11-26-19(14-20)12-13-37-26/h2-11,14-16,28H,12-13,17H2,1H3,(H,33,35)/t28-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development
Curated by ChEMBL
| Assay Description
Phosphodiesterase 5 activity of human corpus cavernosum |
J Med Chem 45: 4094-6 (2002)
BindingDB Entry DOI: 10.7270/Q2R49RGM |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50144249

(CHEMBL302801 | N,N-Diethyl-4-{[(1S,5R)-8-phenethyl...)Show SMILES CCN(CC)C(=O)c1ccc(cc1)C(=C1\C[C@@H]2CC[C@H](C1)N2CCc1ccccc1)\c1ccccc1 |THB:22:21:14.15.20:17.18| Show InChI InChI=1S/C33H38N2O/c1-3-34(4-2)33(36)28-17-15-27(16-18-28)32(26-13-9-6-10-14-26)29-23-30-19-20-31(24-29)35(30)22-21-25-11-7-5-8-12-25/h5-18,30-31H,3-4,19-24H2,1-2H3/b32-29-/t30-,31+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards opioid receptor delta 1 |
Bioorg Med Chem Lett 14: 2113-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.052
BindingDB Entry DOI: 10.7270/Q21J997Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data