 Found 608 hits for monomerid = 14971,14972,14973,14974,14975,14976,15020,35318,35319,35320,35321,50207861,50237710
Found 608 hits for monomerid = 14971,14972,14973,14974,14975,14976,15020,35318,35319,35320,35321,50207861,50237710 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Mast/stem cell growth factor receptor Kit
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Ephrin type-B receptor 2
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 2 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Ephrin type-B receptor 1
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 29.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 1 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Ephrin type-B receptor 4
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 41.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 9 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 15
(Mus musculus) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of mouse carbonic anhydrase 15 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 7
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 99 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 7 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 14
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 223 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 14 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 12
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 302 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 12 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 3
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 443 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 3 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 4
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 446 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 4 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 6
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 461 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 6 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2.14E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.95E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lyn
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 13
(Mus musculus (mouse)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of mouse carbonic anhydrase 13 by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase FRK
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.72E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Cytoplasmic tyrosine-protein kinase BMX
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.81E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 5A, mitochondrial
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 5.49E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 5A by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 7.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 7.22E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Fgr
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 7.74E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 8.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
BDNF/NT-3 growth factors receptor
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 8.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ZAP-70
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 8.37E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK2
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 8.54E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Ret proto-oncogene
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 8.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Macrophage-stimulating protein receptor
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 8.99E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Non-receptor tyrosine-protein kinase TYK2
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | PDB
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 9.44E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Blk
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 103: 3153-8 (2006)
Article DOI: 10.1073/pnas.0511292103
BindingDB Entry DOI: 10.7270/Q21Z430B |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 5B, mitochondrial
(Homo sapiens (Human)) | BDBM50237710

(4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)-5-(trifl...)Show SMILES Cc1cn(cn1)-c1cc(NC(=O)c2ccc(C)c(Nc3nccc(n3)-c3cccnc3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C28H22F3N7O/c1-17-5-6-19(10-25(17)37-27-33-9-7-24(36-27)20-4-3-8-32-14-20)26(39)35-22-11-21(28(29,30)31)12-23(13-22)38-15-18(2)34-16-38/h3-16H,1-2H3,(H,35,39)(H,33,36,37) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.49E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tampere and Tampere University Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human carbonic anhydrase 5B by stopped flow CO2 hydration assay |
Bioorg Med Chem Lett 19: 4102-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.06.002
BindingDB Entry DOI: 10.7270/Q23778S7 |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM14971
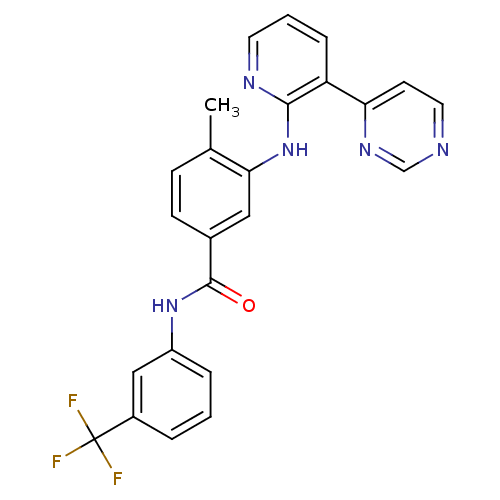
(4-Methyl-3-(3-(pyrimidin-4-yl)pyridin-2-ylamino)-N...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1cccc(c1)C(F)(F)F Show InChI InChI=1S/C24H18F3N5O/c1-15-7-8-16(23(33)31-18-5-2-4-17(13-18)24(25,26)27)12-21(15)32-22-19(6-3-10-29-22)20-9-11-28-14-30-20/h2-14H,1H3,(H,29,32)(H,31,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM14972

(4-Methyl-N-phenyl-3-(3-(pyrimidin-4-yl)pyridin-2-y...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1ccccc1 Show InChI InChI=1S/C23H19N5O/c1-16-9-10-17(23(29)27-18-6-3-2-4-7-18)14-21(16)28-22-19(8-5-12-25-22)20-11-13-24-15-26-20/h2-15H,1H3,(H,25,28)(H,27,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.22E+3 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM14973
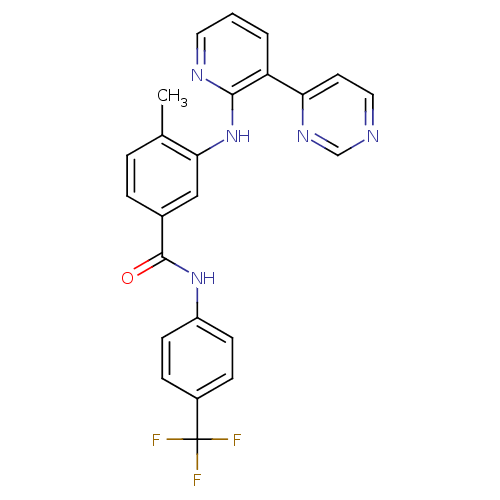
(4-Methyl-3-(3-(pyrimidin-4-yl)pyridin-2-ylamino)-N...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C24H18F3N5O/c1-15-4-5-16(23(33)31-18-8-6-17(7-9-18)24(25,26)27)13-21(15)32-22-19(3-2-11-29-22)20-10-12-28-14-30-20/h2-14H,1H3,(H,29,32)(H,31,33) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 504 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM14974

(CHEMBL221484 | N-(3-(3-(Dimethylamino)propyl)-5-(t...)Show SMILES CN(C)CCCc1cc(NC(=O)c2ccc(C)c(Nc3ncccc3-c3ccncn3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C29H29F3N6O/c1-19-8-9-21(16-26(19)37-27-24(7-4-11-34-27)25-10-12-33-18-35-25)28(39)36-23-15-20(6-5-13-38(2)3)14-22(17-23)29(30,31)32/h4,7-12,14-18H,5-6,13H2,1-3H3,(H,34,37)(H,36,39) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM14975
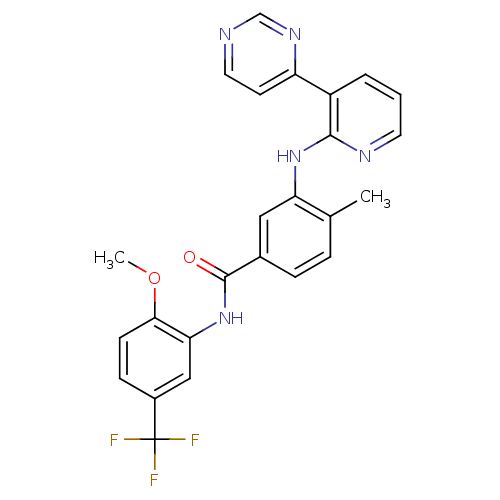
(N-[2-methoxy-5-(trifluoromethyl)phenyl]-4-methyl-3...)Show SMILES COc1ccc(cc1NC(=O)c1ccc(C)c(Nc2ncccc2-c2ccncn2)c1)C(F)(F)F Show InChI InChI=1S/C25H20F3N5O2/c1-15-5-6-16(24(34)33-21-13-17(25(26,27)28)7-8-22(21)35-2)12-20(15)32-23-18(4-3-10-30-23)19-9-11-29-14-31-19/h3-14H,1-2H3,(H,30,32)(H,33,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM14976
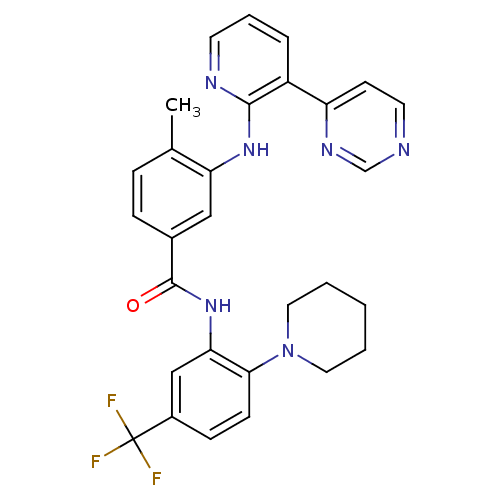
(4-Methyl-N-(2-(piperidin-1-yl)-5-(trifluoromethyl)...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1cc(ccc1N1CCCCC1)C(F)(F)F Show InChI InChI=1S/C29H27F3N6O/c1-19-7-8-20(16-24(19)36-27-22(6-5-12-34-27)23-11-13-33-18-35-23)28(39)37-25-17-21(29(30,31)32)9-10-26(25)38-14-3-2-4-15-38/h5-13,16-18H,2-4,14-15H2,1H3,(H,34,36)(H,37,39) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 181 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM14971

(4-Methyl-3-(3-(pyrimidin-4-yl)pyridin-2-ylamino)-N...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1cccc(c1)C(F)(F)F Show InChI InChI=1S/C24H18F3N5O/c1-15-7-8-16(23(33)31-18-5-2-4-17(13-18)24(25,26)27)12-21(15)32-22-19(6-3-10-29-22)20-9-11-28-14-30-20/h2-14H,1H3,(H,29,32)(H,31,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM14972

(4-Methyl-N-phenyl-3-(3-(pyrimidin-4-yl)pyridin-2-y...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1ccccc1 Show InChI InChI=1S/C23H19N5O/c1-16-9-10-17(23(29)27-18-6-3-2-4-7-18)14-21(16)28-22-19(8-5-12-25-22)20-11-13-24-15-26-20/h2-15H,1H3,(H,25,28)(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 435 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM14973

(4-Methyl-3-(3-(pyrimidin-4-yl)pyridin-2-ylamino)-N...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C24H18F3N5O/c1-15-4-5-16(23(33)31-18-8-6-17(7-9-18)24(25,26)27)13-21(15)32-22-19(3-2-11-29-22)20-10-12-28-14-30-20/h2-14H,1H3,(H,29,32)(H,31,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 177 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM14974

(CHEMBL221484 | N-(3-(3-(Dimethylamino)propyl)-5-(t...)Show SMILES CN(C)CCCc1cc(NC(=O)c2ccc(C)c(Nc3ncccc3-c3ccncn3)c2)cc(c1)C(F)(F)F Show InChI InChI=1S/C29H29F3N6O/c1-19-8-9-21(16-26(19)37-27-24(7-4-11-34-27)25-10-12-33-18-35-25)28(39)36-23-15-20(6-5-13-38(2)3)14-22(17-23)29(30,31)32/h4,7-12,14-18H,5-6,13H2,1-3H3,(H,34,37)(H,36,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM14975

(N-[2-methoxy-5-(trifluoromethyl)phenyl]-4-methyl-3...)Show SMILES COc1ccc(cc1NC(=O)c1ccc(C)c(Nc2ncccc2-c2ccncn2)c1)C(F)(F)F Show InChI InChI=1S/C25H20F3N5O2/c1-15-5-6-16(24(34)33-21-13-17(25(26,27)28)7-8-22(21)35-2)12-20(15)32-23-18(4-3-10-30-23)19-9-11-29-14-31-19/h3-14H,1-2H3,(H,30,32)(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 58 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM14976

(4-Methyl-N-(2-(piperidin-1-yl)-5-(trifluoromethyl)...)Show SMILES Cc1ccc(cc1Nc1ncccc1-c1ccncn1)C(=O)Nc1cc(ccc1N1CCCCC1)C(F)(F)F Show InChI InChI=1S/C29H27F3N6O/c1-19-7-8-20(16-24(19)36-27-22(6-5-12-34-27)23-11-13-33-18-35-23)28(39)37-25-17-21(29(30,31)32)9-10-26(25)38-14-3-2-4-15-38/h5-13,16-18H,2-4,14-15H2,1H3,(H,34,36)(H,37,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen
| Assay Description
The assay uses purified enzyme interacting with biotinylated peptide substrate. HTRF is based on the proximity of europium cryptate (donor fluorophor... |
J Med Chem 50: 611-26 (2007)
Article DOI: 10.1021/jm061107l
BindingDB Entry DOI: 10.7270/Q2MC8X8S |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data






 Purchase
Purchase Purchase
Purchase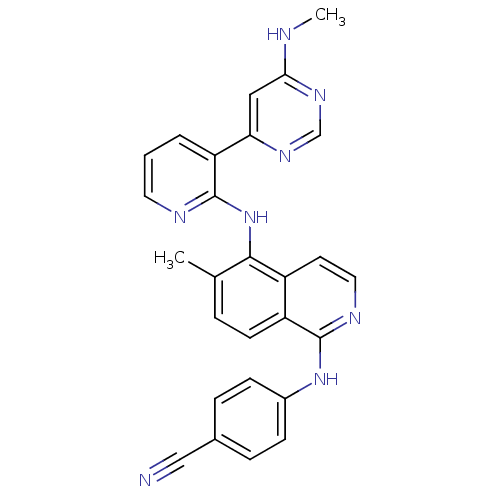

 Purchase
Purchase