

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Wt: 614.6 BDBM19 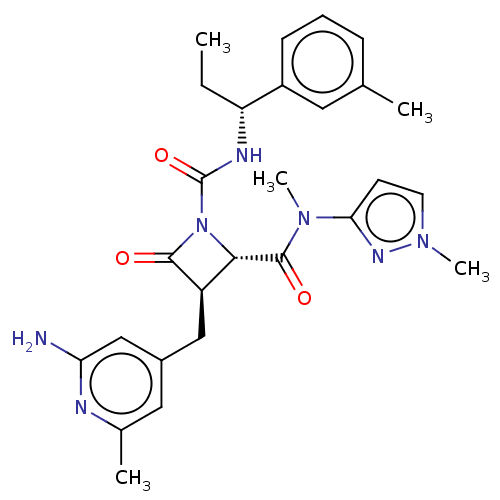 Purchase Purchase | Wt: 360.2 BDBM3032 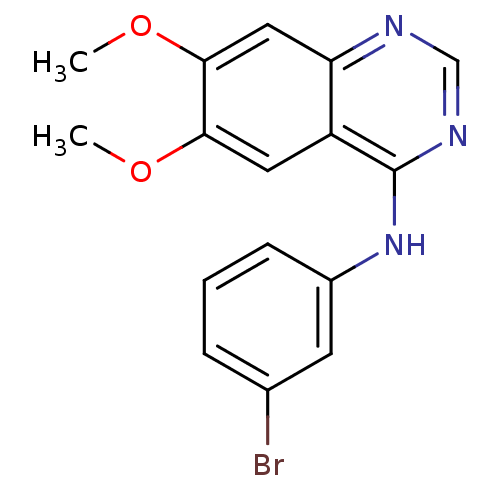 Purchase Purchase | Wt: 466.5 BDBM2579  Purchase Purchase | Wt: 374.2 BDBM3563  Purchase Purchase | Wt: 360.2 BDBM3567 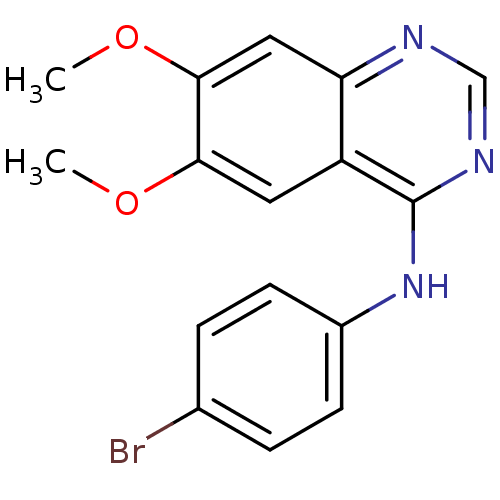 Purchase Purchase |
| Wt: 281.3 BDBM3530  Purchase Purchase | Wt: 302.1 BDBM4078  Purchase Purchase | Wt: 530.4 BDBM4552 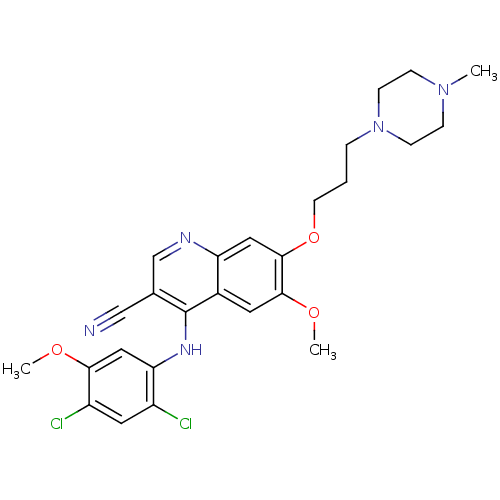 Purchase Purchase | Wt: 268.2 BDBM4367 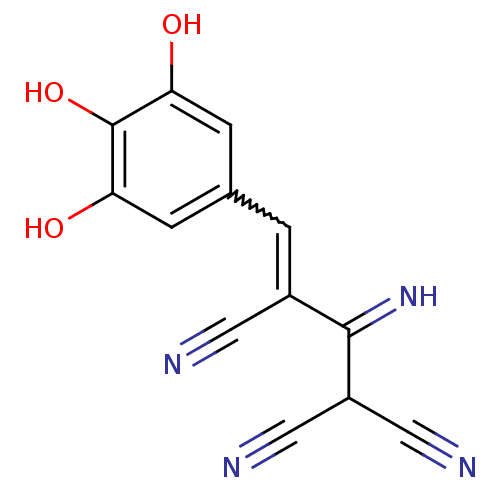 | Wt: 315.7 BDBM4620 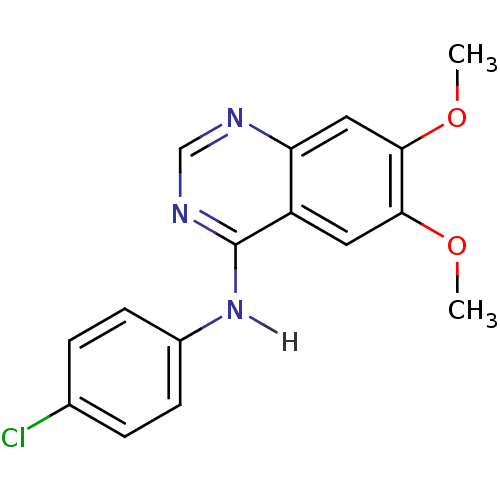 Purchase Purchase |
| Wt: 333.7 BDBM4621  Purchase Purchase | Wt: 297.3 BDBM4622 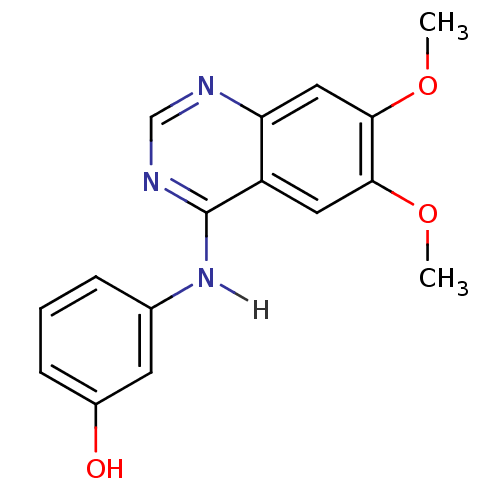 Purchase Purchase | Wt: 317.2 BDBM4625 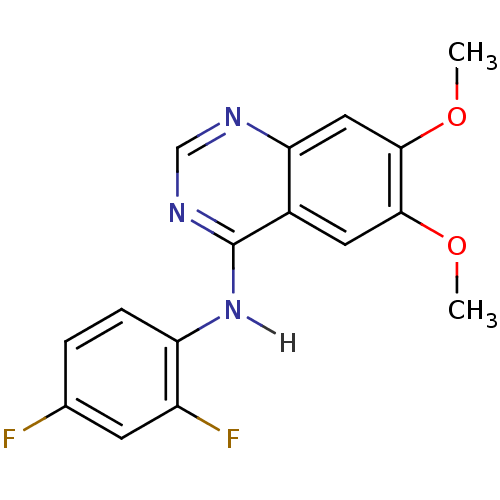 Purchase Purchase | Wt: 333.7 BDBM4626 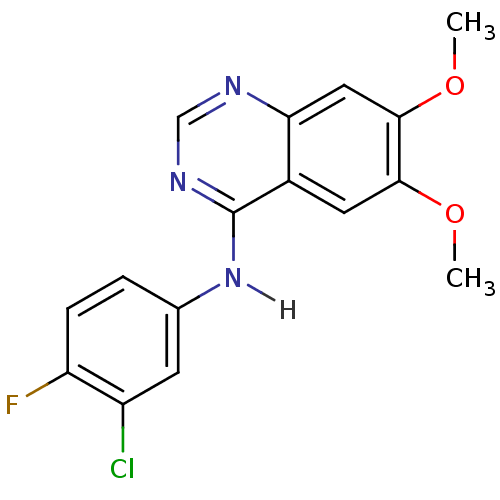 Purchase Purchase | Wt: 475.3 BDBM21 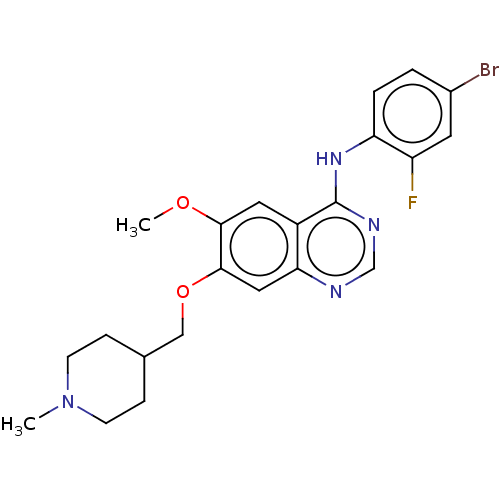 Purchase Purchase |
| Displayed 1 to 15 (of 296 total ) | Next | Last >> |
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM4078 (6,7,13,14-tetrahydroxy-2,9-dioxatetracyclo[6.6.2.0...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 2.07E+5 | -5.02 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Ataturk University | Assay Description Carbonic anhydrase activity was assayed by following the change in absorbance at 348 nm of 4-nitrophenylacetate (NPA) to 4-nitrophenylate ion using s... | Chem Biol Drug Des 75: 515-20 (2010) Article DOI: 10.1111/j.1747-0285.2010.00965.x BindingDB Entry DOI: 10.7270/Q23F4N46 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ribosomal protein S6 kinase alpha-6 (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | PCBioAssay | 2.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences Curated by PubChem BioAssay | Assay Description Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q27P8WRB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mast/stem cell growth factor receptor Kit (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | PCBioAssay | 2.60E+5 | -4.89 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Ambit Biosciences Curated by PubChem BioAssay | Assay Description Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q2NC5ZHH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mast/stem cell growth factor receptor Kit (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | PCBioAssay | 2.90E+5 | -4.82 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Ambit Biosciences Curated by PubChem BioAssay | Assay Description Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q2NC5ZHH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ribosomal protein S6 kinase alpha-1 (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | PCBioAssay | 4.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences Curated by PubChem BioAssay | Assay Description Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q2765CQQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mast/stem cell growth factor receptor Kit (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | PCBioAssay | 2.00E+6 | -3.68 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Ambit Biosciences Curated by PubChem BioAssay | Assay Description Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q2NC5ZHH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM3032 (CHEMBL1204168 | CHEMBL29197 | N-(3-bromophenyl)-6,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 40: 3601-16 (1997) Article DOI: 10.1021/jm970124v BindingDB Entry DOI: 10.7270/Q2H70D0N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase transforming protein Abl (Abelson murine leukemia virus) | BDBM3032 (CHEMBL1204168 | CHEMBL29197 | N-(3-bromophenyl)-6,...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 40: 3601-16 (1997) Article DOI: 10.1021/jm970124v BindingDB Entry DOI: 10.7270/Q2H70D0N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM3032 (CHEMBL1204168 | CHEMBL29197 | N-(3-bromophenyl)-6,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 40: 3601-16 (1997) Article DOI: 10.1021/jm970124v BindingDB Entry DOI: 10.7270/Q2H70D0N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein kinase C alpha type (Bos taurus (bovine)) | BDBM3032 (CHEMBL1204168 | CHEMBL29197 | N-(3-bromophenyl)-6,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 40: 3601-16 (1997) Article DOI: 10.1021/jm970124v BindingDB Entry DOI: 10.7270/Q2H70D0N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1 (Homo sapiens (Human)) | BDBM3032 (CHEMBL1204168 | CHEMBL29197 | N-(3-bromophenyl)-6,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharmaceuticals | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 40: 3601-16 (1997) Article DOI: 10.1021/jm970124v BindingDB Entry DOI: 10.7270/Q2H70D0N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Myosin light chain kinase, smooth muscle (Gallus gallus (chicken)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 10 | n/a | n/a | n/a | n/a | 7.2 | 30 |
Tumor Biology Center | Assay Description The activity of MLCK is measured by its ability to transfer 32P from [gamma-32P]ATP to myosin light-chain. | Bioorg Med Chem Lett 5: 55-60 (1995) Article DOI: 10.1016/0960-894X(94)00458-R BindingDB Entry DOI: 10.7270/Q2CF9N8C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein kinase C beta type (Rattus norvegicus (rat)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Tumor Biology Center | Assay Description The activity of PKC, activated by phosphatidylserine and Ca2+, is measured by its ability to transfer phosphate from [gamma-32P]ATP to lysine-rich hi... | Bioorg Med Chem Lett 5: 55-60 (1995) Article DOI: 10.1016/0960-894X(94)00458-R BindingDB Entry DOI: 10.7270/Q2CF9N8C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-dependent protein kinase catalytic subunit alpha (Bos taurus (bovine)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Tumor Biology Center | Assay Description The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. | Bioorg Med Chem Lett 5: 55-60 (1995) Article DOI: 10.1016/0960-894X(94)00458-R BindingDB Entry DOI: 10.7270/Q2CF9N8C | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| cGMP-dependent protein kinase 1 (Bos taurus (bovine)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Tumor Biology Center | Assay Description The activity of cGPK, activated by cGMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. | Bioorg Med Chem Lett 5: 55-60 (1995) Article DOI: 10.1016/0960-894X(94)00458-R BindingDB Entry DOI: 10.7270/Q2CF9N8C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 2 (Mus musculus (mouse)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Tumor Biology Center | Assay Description The activity of TPK is measured by its ability to transfer 32P from [gamma-32P]ATP to Poly(Glu,Tyr; 4:l). | Bioorg Med Chem Lett 5: 55-60 (1995) Article DOI: 10.1016/0960-894X(94)00458-R BindingDB Entry DOI: 10.7270/Q2CF9N8C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein kinase C beta type (Rattus norvegicus (rat)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Roche Products Limited | Assay Description The activity of PKC, activated by phosphatidylerine and Ca2+, is measured by its ability to transfer phosphate from [gamma-32P]ATP to lysine-rich his... | J Med Chem 35: 177-84 (1992) Article DOI: 10.1021/jm00079a024 BindingDB Entry DOI: 10.7270/Q2K64G8V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein kinase C beta type (Rattus norvegicus (rat)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Roche Products Limited | Assay Description The activity of PKC, activated by phosphatidylerine and Ca2+, is measured by its ability to transfer phosphate from [gamma-32P]ATP to lysine-rich his... | J Med Chem 36: 21-9 (1993) Article DOI: 10.1021/jm00053a003 BindingDB Entry DOI: 10.7270/Q29P2ZTD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-dependent protein kinase catalytic subunit alpha (Bos taurus (bovine)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 121 | n/a | n/a | n/a | n/a | 8.5 | 37 |
Roche Products Limited | Assay Description The activity of PKA, activated by cAMP, is measured by its ability to transfer phosphate from [gamma-32P]ATP to histone. | J Med Chem 36: 21-9 (1993) Article DOI: 10.1021/jm00053a003 BindingDB Entry DOI: 10.7270/Q29P2ZTD | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM3530 (6,7-dimethoxy-N-phenylquinazolin-4-amine | CHEMBL5...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 39: 267-76 (1996) Article DOI: 10.1021/jm9503613 BindingDB Entry DOI: 10.7270/Q25T3HPR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM3563 (CHEMBL447230 | N-(3-bromophenyl)-6,7-dimethoxy-N-m...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 152 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 39: 267-76 (1996) Article DOI: 10.1021/jm9503613 BindingDB Entry DOI: 10.7270/Q25T3HPR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM3567 (CHEMBL328106 | N-(4-bromophenyl)-6,7-dimethoxyquin...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.960 | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research | Assay Description IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... | J Med Chem 39: 267-76 (1996) Article DOI: 10.1021/jm9503613 BindingDB Entry DOI: 10.7270/Q25T3HPR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM3032 (CHEMBL1204168 | CHEMBL29197 | N-(3-bromophenyl)-6,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research | Assay Description The EGF-R kinase autophosphorylation activity was measured by DELFIA/time-resolved fluorometry with excitation at 340 nm and emission at 615 nm. Po... | J Med Chem 43: 3244-56 (2000) Article DOI: 10.1021/jm000206a BindingDB Entry DOI: 10.7270/Q22V2D9M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM4552 (4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research | Assay Description Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra... | J Med Chem 44: 3965-77 (2001) Article DOI: 10.1021/jm0102250 BindingDB Entry DOI: 10.7270/Q2TD9VHP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Homo sapiens (Human)) | BDBM4620 (Anilinoquinazoline deriv. 3 | N-(4-chlorophenyl)-6...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Homo sapiens (Human)) | BDBM4621 (Anilinoquinazoline deriv. 4 | CHEMBL150315 | N-(4-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Homo sapiens (Human)) | BDBM4622 (3-[(6,7-dimethoxyquinazolin-4-yl)amino]phenol | An...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Homo sapiens (Human)) | BDBM4625 (Anilinoquinazoline deriv. 8 | N-(2,4-difluoropheny...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.20E+4 | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Homo sapiens (Human)) | BDBM4626 (Anilinoquinazoline deriv. 9 | CHEMBL301018 | N-(3-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM4620 (Anilinoquinazoline deriv. 3 | N-(4-chlorophenyl)-6...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM4621 (Anilinoquinazoline deriv. 4 | CHEMBL150315 | N-(4-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM4622 (3-[(6,7-dimethoxyquinazolin-4-yl)amino]phenol | An...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM4625 (Anilinoquinazoline deriv. 8 | N-(2,4-difluoropheny...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM4626 (Anilinoquinazoline deriv. 9 | CHEMBL301018 | N-(3-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM4620 (Anilinoquinazoline deriv. 3 | N-(4-chlorophenyl)-6...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit FGFR-1 TK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM4621 (Anilinoquinazoline deriv. 4 | CHEMBL150315 | N-(4-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit FGFR-1 TK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM4622 (3-[(6,7-dimethoxyquinazolin-4-yl)amino]phenol | An...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit FGFR-1 TK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM4625 (Anilinoquinazoline deriv. 8 | N-(2,4-difluoropheny...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.80E+4 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit FGFR-1 TK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM4626 (Anilinoquinazoline deriv. 9 | CHEMBL301018 | N-(3-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit FGFR-1 TK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 42: 5369-89 (1999) Article DOI: 10.1021/jm990345w BindingDB Entry DOI: 10.7270/Q2F47MBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 1 (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 45: 1300-12 (2002) Article DOI: 10.1021/jm011022e BindingDB Entry DOI: 10.7270/Q29C6VMW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit VEGF-R RTK activity. The compounds were incubated with enzyme 20 min at room... | J Med Chem 45: 1300-12 (2002) Article DOI: 10.1021/jm011022e BindingDB Entry DOI: 10.7270/Q29C6VMW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fibroblast growth factor receptor 1 (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit FGFR-1 TK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 45: 1300-12 (2002) Article DOI: 10.1021/jm011022e BindingDB Entry DOI: 10.7270/Q29C6VMW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM21 (CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | 25 |
AstraZeneca | Assay Description An ELISA assay was used to determine the ability of inhibitor to inhibit EGF-R RTK activity. The compounds were incubated with enzyme 20 min at room ... | J Med Chem 45: 1300-12 (2002) Article DOI: 10.1021/jm011022e BindingDB Entry DOI: 10.7270/Q29C6VMW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM4552 (4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Wyeth Research | Assay Description Kinase assays were performed using the europium/APC detection format (LANCE, Perkin Elmer). HTRF is based on the proximity of europium cryptate (dono... | J Med Chem 47: 6666-8 (2004) Article DOI: 10.1021/jm049237m BindingDB Entry DOI: 10.7270/Q2CV4FXN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM4552 (4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Wyeth Research | Assay Description Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra... | J Med Chem 48: 5909-20 (2005) Article DOI: 10.1021/jm050512u BindingDB Entry DOI: 10.7270/Q2833Q6N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM4552 (4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Wyeth-Ayerst Research | Assay Description Kinase assays were performed using the europium/APC detection format (LANCE, Perkin Elmer). HTRF is based on the proximity of europium cryptate (dono... | Bioorg Med Chem Lett 15: 1743-7 (2005) Article DOI: 10.1016/j.bmcl.2005.01.004 BindingDB Entry DOI: 10.7270/Q2VX0DRH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-dependent kinase 4/G1/S-specific cyclin-D1 (Homo sapiens (Human)) | BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 59 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Lilly Research Laboratories | Assay Description In vitro CDK assay using purified enzyme, was incubated at room temperature with substrate, and test compounds in the presence of ATP/[gamma-33P]ATP.... | Bioorg Med Chem Lett 13: 2261-7 (2003) Article DOI: 10.1016/s0960-894x(03)00461-x BindingDB Entry DOI: 10.7270/Q29W0CPW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM4078 (6,7,13,14-tetrahydroxy-2,9-dioxatetracyclo[6.6.2.0...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | 7.4 | 30 |
Pfizer | Assay Description IC50 is the inhibitor concentration, which inhibits 50% of pp60c-src activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] ... | J Med Chem 37: 2224-31 (1994) Article DOI: 10.1021/jm00040a015 BindingDB Entry DOI: 10.7270/Q27M0648 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-dependent protein kinase catalytic subunit alpha (Bos taurus (bovine)) | BDBM4078 (6,7,13,14-tetrahydroxy-2,9-dioxatetracyclo[6.6.2.0...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 600 | n/a | n/a | n/a | n/a | 7.4 | 30 |
Pfizer | Assay Description IC50 is the inhibitor concentration, which inhibits 50% of PKA activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] label... | J Med Chem 37: 2224-31 (1994) Article DOI: 10.1021/jm00040a015 BindingDB Entry DOI: 10.7270/Q27M0648 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Homo sapiens (Human)) | BDBM4552 (4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | 7.5 | 37 |
Wyeth Research | Assay Description Kinase assays were performed using the europium/APC detection format (LANCE, Perkin Elmer). HTRF is based on the proximity of europium cryptate (dono... | J Med Chem 48: 3891-902 (2005) Article DOI: 10.1021/jm050175p BindingDB Entry DOI: 10.7270/Q2T43R88 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| << First | Previous | Displayed 51 to 100 (of 4231 total ) | Next | Last >> |
| Cell (A) | Syringe (B) | Cell Links | Syringe Links | Cell + Syr Links | ΔG° kcal/mole | -TΔS° kcal/mole | ΔH° kcal/mole | log K | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -8.53 | 13.2 | -21.7 | 6.25 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -7.85 | 11.7 | -19.4 | 5.76 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -6.83 | 6.58 | -13.4 | 5.01 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -6.22 | 4.77 | -11.0 | 4.56 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -5.52 | 3.59 | -9.03 | 4.17 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -4.82 | 0 | -4.82 | 3.53 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -6.62 | 12.9 | -19.5 | 4.89 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -6.39 | 6.91 | -17.3 | 4.69 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
| RNA(23-mer) (NULL) | BDBM19 ((2S,3S,4R,5R,6R)-5-amino-2-(aminomethyl)-6-{[(2R,3...) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -5.96 | 5.68 | -11.7 | 4.37 | 7 | 25 | |
The Ohio State University | Nucleic Acids Res 28: 2935-42 (2000) | |||||||||
BDBM2579 ((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...) | Serine/threonine-protein kinase PIM (Homo sapiens (Human)) | CHEBI KEGG MMDB PC cid PC sid PDB | DrugBank GoogleScholar KEGG PDB | -11.5 | -3.33 | -8.09 | 8.85 | 7.5 | 10 | |
Oxford University | J Med Chem 48: 7604-14 (2005) | |||||||||