

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Wt: 264.3 BDBM19149  Purchase Purchase | Wt: 540.6 BDBM19151 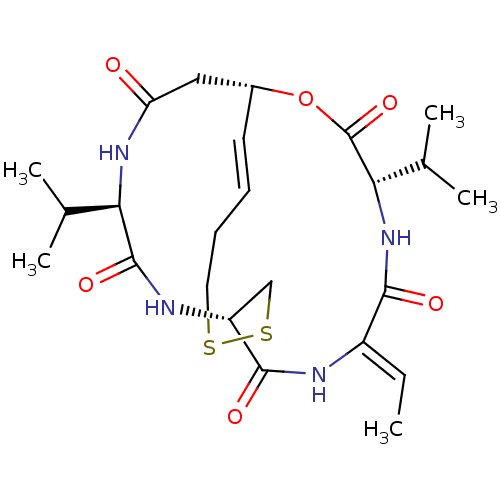 Purchase Purchase | Wt: 376.4 BDBM19410 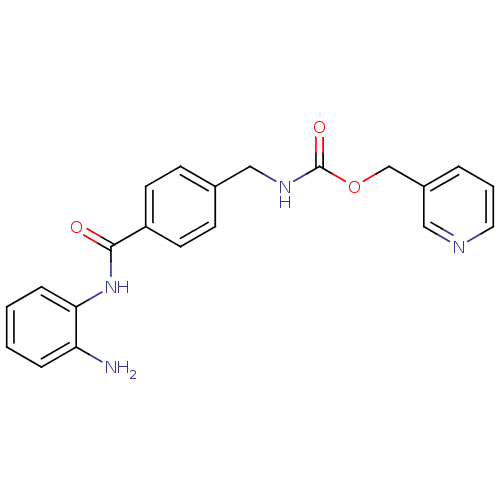 Purchase Purchase | Wt: 351.4 BDBM19423  Purchase Purchase | Wt: 379.4 BDBM19428 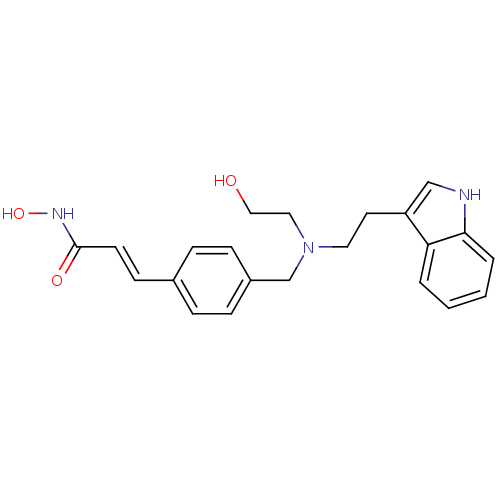 Purchase Purchase |
| Wt: 302.3 BDBM19130  | Wt: 721.8 BDBM22449  Purchase Purchase | Wt: 396.4 BDBM24624 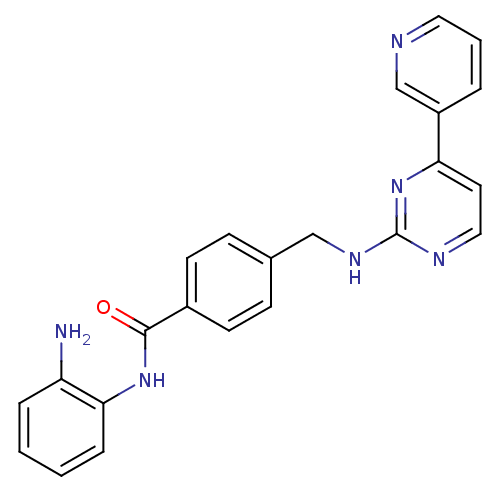 Purchase Purchase | Wt: 252.2 BDBM25153 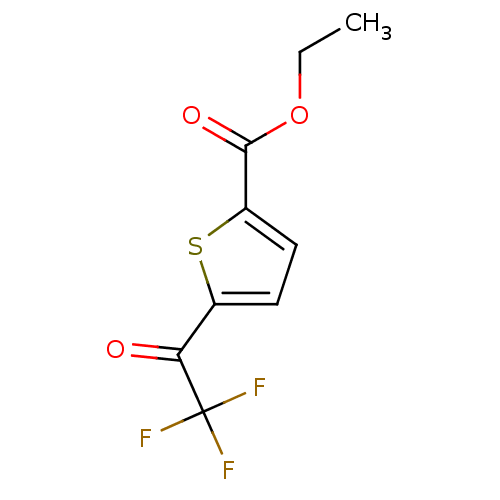 Purchase Purchase | Wt: 236.1 BDBM25154 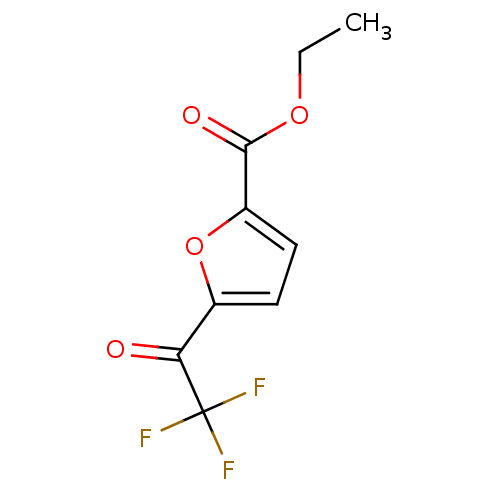 Purchase Purchase |
| Wt: 623.7 BDBM25142 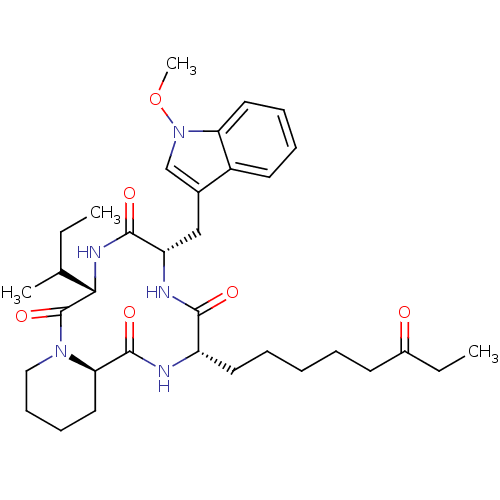 Purchase Purchase | Wt: 318.3 BDBM25150 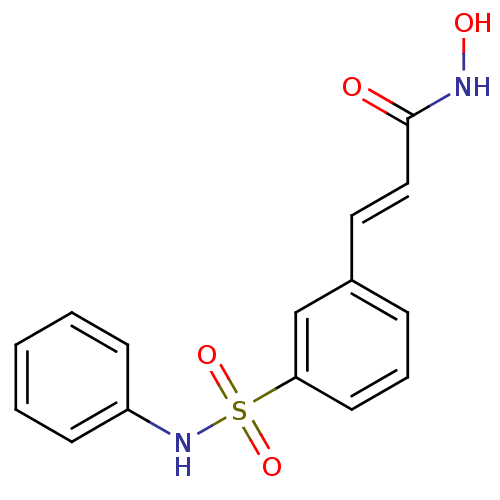 Purchase Purchase | Wt: 273.3 BDBM25169 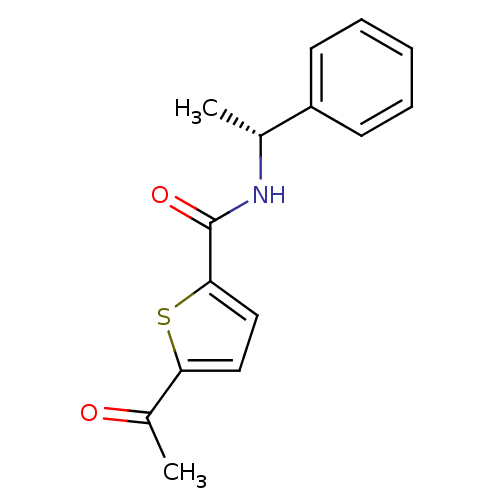 Purchase Purchase | Wt: 88.1 BDBM26109 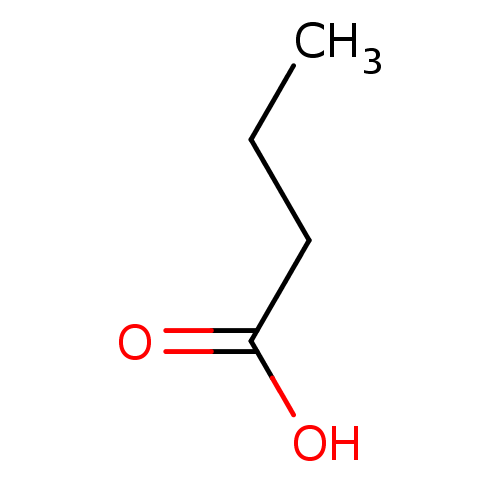 Purchase Purchase | Wt: 349.4 BDBM29589 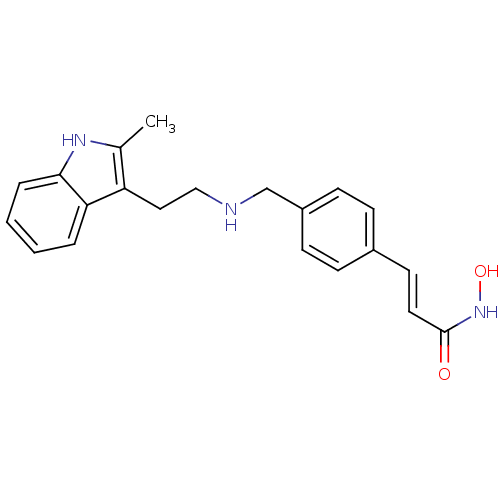 Purchase Purchase |
| Displayed 1 to 15 (of 63 total ) | Next | Last >> |
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | 5.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Heinrich-Heine-Universit£t D£sseldorf Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC8 | Bioorg Med Chem 28: (2020) Article DOI: 10.1016/j.bmc.2019.115108 BindingDB Entry DOI: 10.7270/Q2NV9NSQ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 7 (Homo sapiens (Human)) | BDBM22449 (CHEMBL356769 | N-(4-{(2R,4R,6S)-4-{[(4,5-diphenyl-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 5.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital Curated by ChEMBL | Assay Description Inhibition of human HDAC7 | Bioorg Med Chem 23: 5151-5 (2015) Article DOI: 10.1016/j.bmc.2014.12.066 BindingDB Entry DOI: 10.7270/Q2B859V9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM22449 (CHEMBL356769 | N-(4-{(2R,4R,6S)-4-{[(4,5-diphenyl-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 6.30E+3 | -7.02 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
University of Notre Dame | Assay Description To assess the effect of test compounds on histone deacetylase enzyme function in Vitro, a fluorometric assay was performed using HDAC, which incubate... | J Med Chem 51: 2898-906 (2008) Article DOI: 10.1021/jm7015254 BindingDB Entry DOI: 10.7270/Q2NC5ZG2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 9 (Homo sapiens (Human)) | BDBM19428 ((2E)-N-hydroxy-3-(4-{[(2-hydroxyethyl)[2-(1H-indol...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital Curated by ChEMBL | Assay Description Inhibition of human HDAC9 | Bioorg Med Chem 23: 5151-5 (2015) Article DOI: 10.1016/j.bmc.2014.12.066 BindingDB Entry DOI: 10.7270/Q2B859V9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM22449 (CHEMBL356769 | N-(4-{(2R,4R,6S)-4-{[(4,5-diphenyl-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 9.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital Curated by ChEMBL | Assay Description Inhibition of human HDAC4 | Bioorg Med Chem 23: 5151-5 (2015) Article DOI: 10.1016/j.bmc.2014.12.066 BindingDB Entry DOI: 10.7270/Q2B859V9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 7 (Homo sapiens (Human)) | BDBM19428 ((2E)-N-hydroxy-3-(4-{[(2-hydroxyethyl)[2-(1H-indol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital Curated by ChEMBL | Assay Description Inhibition of human HDAC7 | Bioorg Med Chem 23: 5151-5 (2015) Article DOI: 10.1016/j.bmc.2014.12.066 BindingDB Entry DOI: 10.7270/Q2B859V9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| NAD-dependent protein deacetylase sirtuin-3, mitochondrial (Homo sapiens (Human)) | BDBM29589 (Faridak | LBH-589 | LBH-589B | Panobinostat | US10...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nestle Skin Health R&D Curated by ChEMBL | Assay Description Inhibition of human recombinant SIRT3 using fluoro-lysine sirtuin 2 deacetylase substrate after 45 mins by fluorimetrc method | Bioorg Med Chem Lett 28: 2985-2992 (2018) Article DOI: 10.1016/j.bmcl.2018.06.029 BindingDB Entry DOI: 10.7270/Q2M0484T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| NAD-dependent protein deacetylase sirtuin-7 (Homo sapiens (Human)) | BDBM29589 (Faridak | LBH-589 | LBH-589B | Panobinostat | US10...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nestle Skin Health R&D Curated by ChEMBL | Assay Description Inhibition of SIRT7 (unknown origin) | Bioorg Med Chem Lett 28: 2985-2992 (2018) Article DOI: 10.1016/j.bmcl.2018.06.029 BindingDB Entry DOI: 10.7270/Q2M0484T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| NAD-dependent protein deacetylase sirtuin-1 (Homo sapiens (Human)) | BDBM29589 (Faridak | LBH-589 | LBH-589B | Panobinostat | US10...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nestle Skin Health R&D Curated by ChEMBL | Assay Description Inhibition of human recombinant SIRT1 using fluorogenic HDAC substrate after 20 mins by fluorimetrc method | Bioorg Med Chem Lett 28: 2985-2992 (2018) Article DOI: 10.1016/j.bmcl.2018.06.029 BindingDB Entry DOI: 10.7270/Q2M0484T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| NAD-dependent protein deacetylase sirtuin-2 (Homo sapiens (Human)) | BDBM29589 (Faridak | LBH-589 | LBH-589B | Panobinostat | US10...) | PDB MMDB NCI pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nestle Skin Health R&D Curated by ChEMBL | Assay Description Inhibition of human recombinant SIRT2 using fluoro-lysine sirtuin 2 deacetylase substrate after 60 mins by fluorimetrc method | Bioorg Med Chem Lett 28: 2985-2992 (2018) Article DOI: 10.1016/j.bmcl.2018.06.029 BindingDB Entry DOI: 10.7270/Q2M0484T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 5 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Applied Sciences Darmstadt Curated by ChEMBL | Assay Description Inhibition of human HDAC5 using Boc-Lys(trifluoroacetyl)-AMC as substrate preincubated for 30 mins followed by substrate addition measured after 60 m... | Bioorg Med Chem Lett 27: 1508-1512 (2017) Article DOI: 10.1016/j.bmcl.2017.02.050 BindingDB Entry DOI: 10.7270/Q2JD502H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Applied Sciences Darmstadt Curated by ChEMBL | Assay Description Inhibition of human HDAC4 using Boc-Lys(trifluoroacetyl)-AMC as substrate preincubated for 30 mins followed by substrate addition measured after 60 m... | Bioorg Med Chem Lett 27: 1508-1512 (2017) Article DOI: 10.1016/j.bmcl.2017.02.050 BindingDB Entry DOI: 10.7270/Q2JD502H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | 1.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Heinrich-Heine-University D£sseldorf Curated by ChEMBL | Assay Description Inhibition of C-terminal His-fusion tagged/N-terminal Strep-2 tagged recombinant human HDAC8 (1 to 377 residues) expressed in insect cells using Boc-... | Bioorg Med Chem 27: (2019) Article DOI: 10.1016/j.bmc.2019.07.052 BindingDB Entry DOI: 10.7270/Q2VX0KZ8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| E3 ubiquitin-protein ligase Mdm2 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
East China University of Science and Technology Curated by ChEMBL | Assay Description Inhibition of MDM2 (unknown origin) preincubated for 30 mins by fluorescence polarization assay | J Med Chem 61: 7245-7260 (2018) Article DOI: 10.1021/acs.jmedchem.8b00664 BindingDB Entry DOI: 10.7270/Q2R213Z9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 2.84E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Heinrich-Heine-Universit£t D£sseldorf Curated by ChEMBL | Assay Description Inhibition of recombinant human HDAC4 | Bioorg Med Chem 28: (2020) Article DOI: 10.1016/j.bmc.2019.115108 BindingDB Entry DOI: 10.7270/Q2NV9NSQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 3.01E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Heinrich-Heine-University D£sseldorf Curated by ChEMBL | Assay Description Inhibition of C-terminal His-tagged/N-terminal GST-tagged recombinant human HDAC4 (627 to 1084 residues) expressed in Baculovirus infected insect cel... | Bioorg Med Chem 27: (2019) Article DOI: 10.1016/j.bmc.2019.07.052 BindingDB Entry DOI: 10.7270/Q2VX0KZ8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier family 22 member 20 (Mus musculus) | BDBM26109 (Butyrate | butanoic acid | butanoic acid, 4) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 8.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Curated by ChEMBL | Assay Description Inhibition of mouse Oat6-mediated [3H]ES uptake in Xenopus oocytes after 1 hr | J Biol Chem 282: 23841-53 (2007) Article DOI: 10.1074/jbc.M703467200 BindingDB Entry DOI: 10.7270/Q2W95B35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase-like amidohydrolase (Alcaligenes sp. (strain DSM 11172) (Bordetella sp....) | BDBM19410 (CHEMBL27759 | MS-275 | US11377423, MS-275 | US1167...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of G£ttingen Curated by ChEMBL | Assay Description Displacement of Atto700-HA from Bordetella/Alcaligenes strain FB188 HDAH by fluorescence anisotropy | Bioorg Med Chem Lett 19: 3651-6 (2009) Article DOI: 10.1016/j.bmcl.2009.04.102 BindingDB Entry DOI: 10.7270/Q24T6JFV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase (Homo sapiens (Human)) | BDBM26109 (Butyrate | butanoic acid | butanoic acid, 4) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | Purchase DrugBank KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.36E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hacettepe University Curated by ChEMBL | Assay Description Inhibition of HDAC in human Hela cells nuclear extracts by fluorimetric assay | Bioorg Med Chem 17: 5219-28 (2009) Article DOI: 10.1016/j.bmc.2009.05.042 BindingDB Entry DOI: 10.7270/Q22F7R82 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier family 22 member 6 (Mus musculus) | BDBM26109 (Butyrate | butanoic acid | butanoic acid, 4) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 3.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Curated by ChEMBL | Assay Description Inhibition of mouse Oat1-mediated [3H]PAH uptake in Xenopus oocytes after 1 hr | J Biol Chem 282: 23841-53 (2007) Article DOI: 10.1074/jbc.M703467200 BindingDB Entry DOI: 10.7270/Q2W95B35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 3/Nuclear receptor corepressor 1 (Homo sapiens (Human)) | BDBM24624 (CHEMBL272980 | MGCD-0103 | MGCD0103 | N-(2-aminoph...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
University College London Curated by ChEMBL | Assay Description Inhibition of recombinant HDAC3-NCoR1 (unknown origin) using MAL as substrate incubated for 3 hrs prior to substrate addition measured after 60 mins ... | J Med Chem 56: 6156-74 (2013) Article DOI: 10.1021/jm400634n BindingDB Entry DOI: 10.7270/Q2N87C65 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 3 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 132 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Michigan Curated by ChEMBL | Assay Description Inhibition of HDAC3 (unknown origin) using fluorogenic peptide from p53 residues (379 to 382) (RHKK(Ac)) as substrate by fluorescence assay | ACS Med Chem Lett 4: 779-783 (2013) Article DOI: 10.1021/ml400175d BindingDB Entry DOI: 10.7270/Q2CZ38KR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19410 (CHEMBL27759 | MS-275 | US11377423, MS-275 | US1167...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 540 | n/a | n/a | n/a | n/a | 8.0 | 37 |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 2 (Homo sapiens (Human)) | BDBM19410 (CHEMBL27759 | MS-275 | US11377423, MS-275 | US1167...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | 8.0 | 37 |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19423 (HDAC inhibitor, Compound 1 | N-[2-amino-5-(thiophe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 2 (Homo sapiens (Human)) | BDBM19423 (HDAC inhibitor, Compound 1 | N-[2-amino-5-(thiophe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 3 (Homo sapiens (Human)) | BDBM19423 (HDAC inhibitor, Compound 1 | N-[2-amino-5-(thiophe...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM19423 (HDAC inhibitor, Compound 1 | N-[2-amino-5-(thiophe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19428 ((2E)-N-hydroxy-3-(4-{[(2-hydroxyethyl)[2-(1H-indol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 2 (Homo sapiens (Human)) | BDBM19428 ((2E)-N-hydroxy-3-(4-{[(2-hydroxyethyl)[2-(1H-indol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 3 (Homo sapiens (Human)) | BDBM19428 ((2E)-N-hydroxy-3-(4-{[(2-hydroxyethyl)[2-(1H-indol...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM19428 ((2E)-N-hydroxy-3-(4-{[(2-hydroxyethyl)[2-(1H-indol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
MethylGene Inc | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5543-6 (2007) Article DOI: 10.1021/jm701079h BindingDB Entry DOI: 10.7270/Q2445JS2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Mus musculus) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 81 | n/a | n/a | n/a | n/a | 8.0 | 37 |
Nagoya City University | Assay Description The enzyme activity was assayed using recombinant HDAC and [3H] acetyl-labeled histones as substrate. The released [3H]acetic acid was extracted and ... | J Med Chem 50: 5425-38 (2007) Article DOI: 10.1021/jm7009217 BindingDB Entry DOI: 10.7270/Q2XK8CT6 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | 8.0 | 37 |
Nagoya City University | Assay Description The enzyme activity was assayed using recombinant HDAC and [3H] acetyl-labeled histones as substrate. The released [3H]acetic acid was extracted and ... | J Med Chem 50: 5425-38 (2007) Article DOI: 10.1021/jm7009217 BindingDB Entry DOI: 10.7270/Q2XK8CT6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University | Assay Description The enzyme activity was assayed using recombinant HDAC and [3H] acetyl-labeled histones as substrate. The released [3H]acetic acid was extracted and ... | J Med Chem 50: 5425-38 (2007) Article DOI: 10.1021/jm7009217 BindingDB Entry DOI: 10.7270/Q2XK8CT6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Mus musculus) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 196 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Southampton | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5720-5726 (2007) Article DOI: 10.1021/jm0703800 BindingDB Entry DOI: 10.7270/Q2ST7N4B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Mus musculus) | BDBM19151 ((1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-bis(propa...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 787 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Southampton | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5720-5726 (2007) Article DOI: 10.1021/jm0703800 BindingDB Entry DOI: 10.7270/Q2ST7N4B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 775 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Southampton | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5720-5726 (2007) Article DOI: 10.1021/jm0703800 BindingDB Entry DOI: 10.7270/Q2ST7N4B | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19151 ((1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-bis(propa...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.97 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Southampton | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | J Med Chem 50: 5720-5726 (2007) Article DOI: 10.1021/jm0703800 BindingDB Entry DOI: 10.7270/Q2ST7N4B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | 8.0 | 37 |
Kyushu Institute of Technology | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | Bioorg Med Chem 14: 3438-46 (2006) Article DOI: 10.1016/j.bmc.2005.12.063 BindingDB Entry DOI: 10.7270/Q2P55KRB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 4 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 20 | n/a | n/a | n/a | n/a | 8.0 | 37 |
Kyushu Institute of Technology | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | Bioorg Med Chem 14: 3438-46 (2006) Article DOI: 10.1016/j.bmc.2005.12.063 BindingDB Entry DOI: 10.7270/Q2P55KRB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 6 (Mus musculus) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 28 | n/a | n/a | n/a | n/a | 8.0 | 37 |
Kyushu Institute of Technology | Assay Description For the enzyme assay, the enzyme fraction was added to fluorescent substrate in the presence of test compounds, and the mixture was incubated for add... | Bioorg Med Chem 14: 3438-46 (2006) Article DOI: 10.1016/j.bmc.2005.12.063 BindingDB Entry DOI: 10.7270/Q2P55KRB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 68 | n/a | n/a | n/a | n/a | 7.5 | 25 |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | 7.5 | 25 |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 2 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 164 | n/a | n/a | n/a | n/a | 7.5 | 25 |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 2 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | 7.5 | 25 |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 3 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 3 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 1.52E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone deacetylase 8 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 1.38E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago | Assay Description The inhibitory effects of compounds on histone deacetylase (HDAC) activity were determined using a fluorescence-based assay with electrophoretic sepa... | J Med Chem 51: 3437-48 (2008) Article DOI: 10.1021/jm701606b BindingDB Entry DOI: 10.7270/Q2ZC815S | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| << First | Previous | Displayed 151 to 200 (of 2840 total ) | Next | Last >> |
| Cell (A) | Syringe (B) | Cell Links | Syringe Links | Cell + Syr Links | ΔG° kcal/mole | -TΔS° kcal/mole | ΔH° kcal/mole | log K | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|
| Methyl-accepting chemotaxis protein (McpS) (Pseudomonas putida (Arthrobacter siderocapsulatus)) | BDBM26109 (Butyrate | butanoic acid | butanoic acid, 4) | GoogleScholar | CHEBI DrugBank KEGG MMDB PC cid PC sid PDB | -5.41 | 2.80 | -7.88 | 4.04 | n/a | 20 | |
CSIC | J Biol Chem 285: 23126-36 (2010) | |||||||||
| Histone deacetylase 1/3/5/8 (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | DrugBank GoogleScholar KEGG PDB | CHEBI DrugBank MMDB PC cid PC sid PDB | -8.36 | 2.42 | -10.9 | 6.13 | n/a | 25 | |
North Dakota State University | Biochemistry 52: 8139-49 (2013) | |||||||||
| Histone deacetylase 1/3/5/8 (Homo sapiens (Human)) | BDBM19130 ((2E,4E,6R)-7-[4-(dimethylamino)phenyl]-N-hydroxy-4...) | DrugBank GoogleScholar KEGG PDB | MMDB PC cid PC sid PDB | -8.60 | 0.214 | -8.84 | 6.31 | n/a | 25 | |
North Dakota State University | Biochemistry 52: 8139-49 (2013) | |||||||||
| Histone deacetylase 8 (HDAC8) (Homo sapiens (Human)) | BDBM19149 (CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...) | DrugBank GoogleScholar KEGG PDB | CHEBI DrugBank MMDB PC cid PC sid PDB | -8.36 | 0.0519 | -8.96 | 6.34 | 7.5 | -248 | |
North Dakota State University | Biochemistry 53: 7445-58 (2014) | |||||||||