

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM190667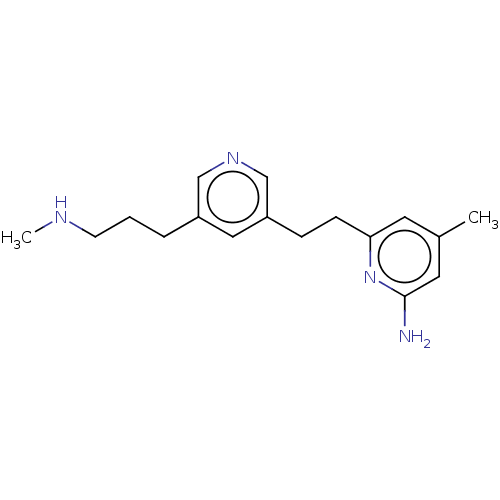 (US10759791, Compound 14j | nNOS inhibitor, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | US Patent | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM152712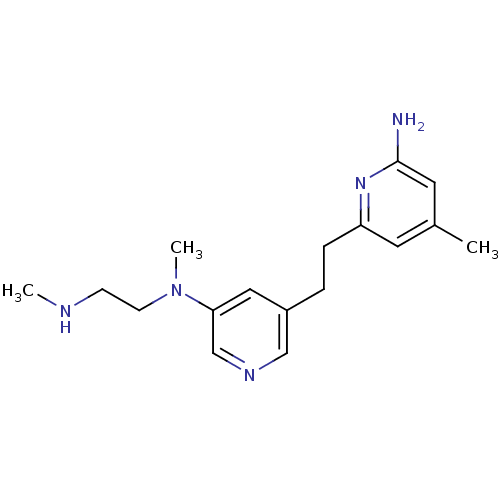 (4-methyl-6-[2-(5-{methyl[2-(methylamino)ethyl]amin...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB US Patent | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50386178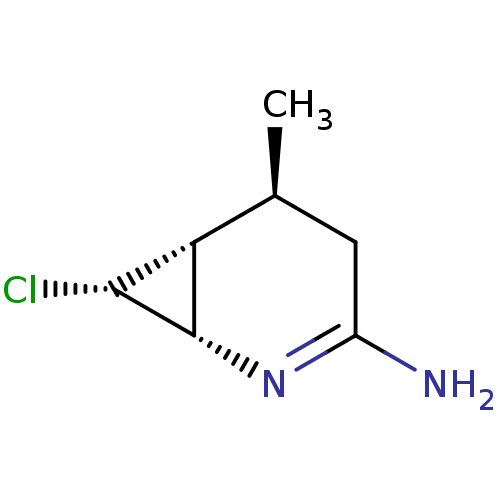 (CHEMBL1800346 | ONO-1714) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nycomed GmbH Curated by ChEMBL | Assay Description Inhibition of human eNOS assessed as inhibition of [3H]L-arginine to [3H]L-citrulline conversion by scintillation counting | Bioorg Med Chem Lett 21: 4228-32 (2011) Article DOI: 10.1016/j.bmcl.2011.05.073 BindingDB Entry DOI: 10.7270/Q2H996GJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM190668 (US10759791, Compound 14k | nNOS inhibitor, 6) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB US Patent | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50225106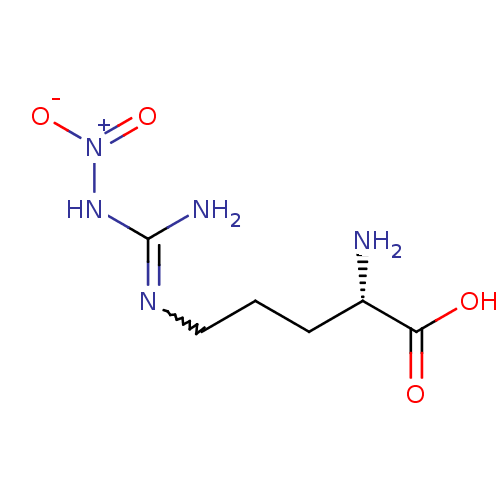 ((2S)-2-amino-5-{[(E)-amino(nitroimino)methyl]amino...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM50014713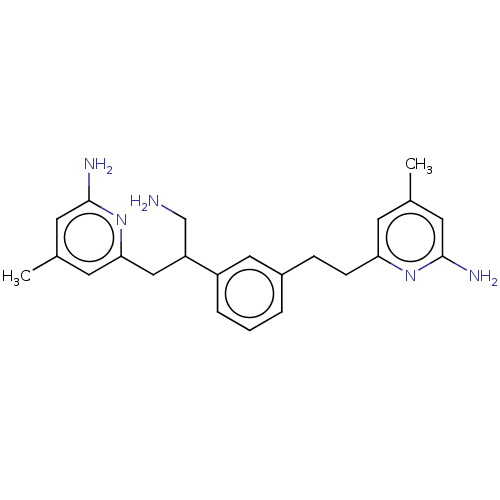 (CHEMBL3262022 | US10759791, Compound 6 | US9951014...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | US Patent | 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM190665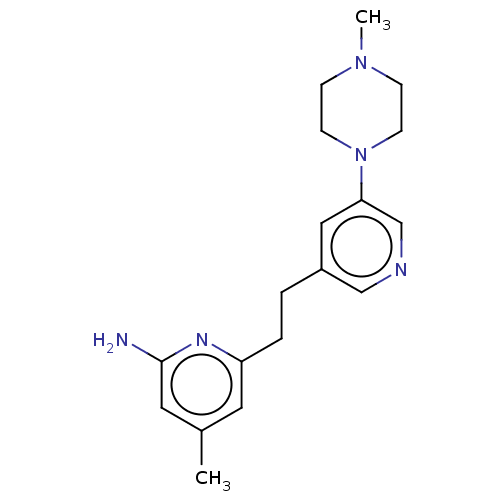 (US10759791, Compound 14g | nNOS inhibitor, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB US Patent | 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM190666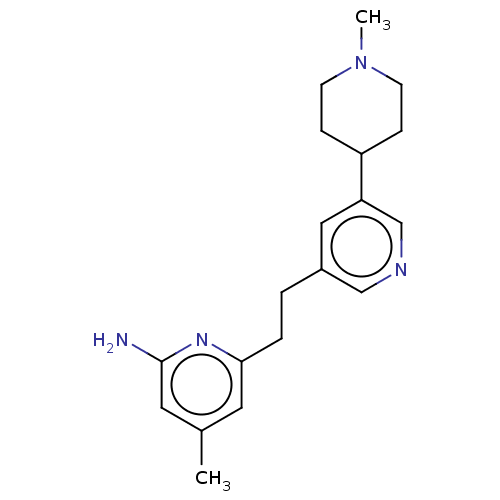 (US10759791, Compound 14h | nNOS inhibitor, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | US Patent | 72 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258602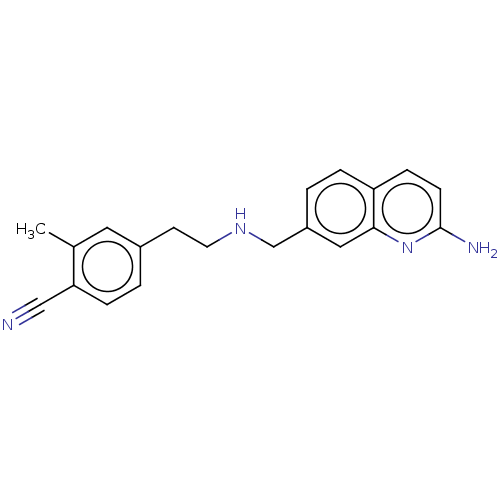 (CHEMBL4068062) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 92 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258601 (CHEMBL4089246) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 115 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM50180872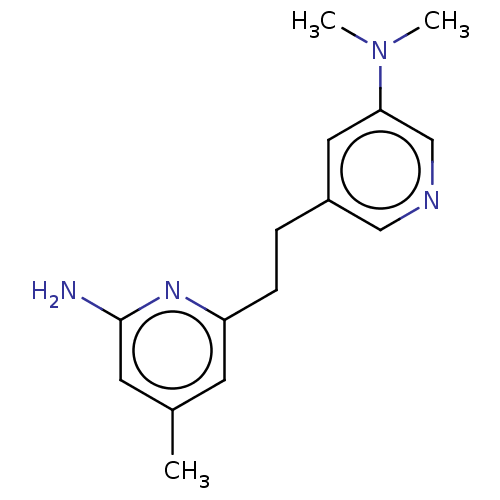 (CHEMBL3819046 | US10759791, Compound 14a) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 139 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50289680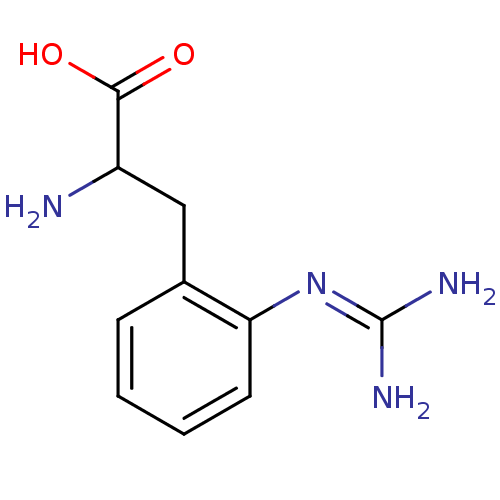 (2-Amino-3-(2-guanidino-phenyl)-propionic acid | CH...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50289681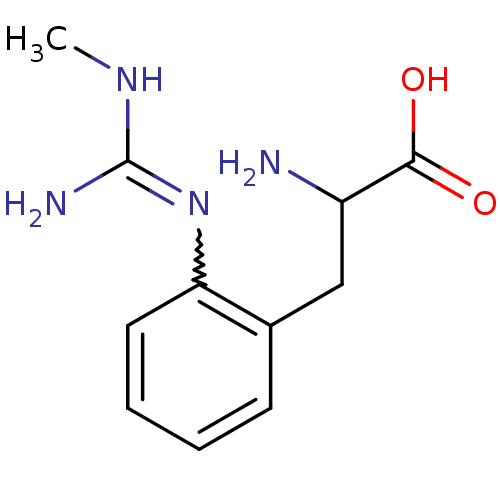 (2-Amino-3-[2-(N'-methyl-guanidino)-phenyl]-propion...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258603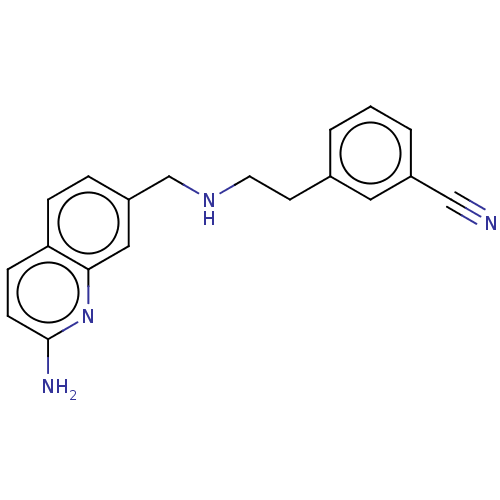 (CHEMBL4090184) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 273 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065823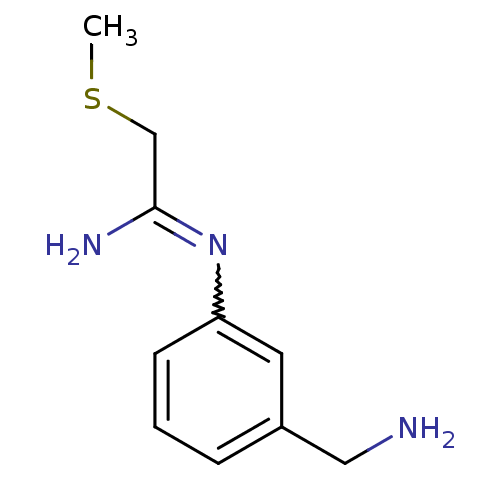 (CHEMBL555794 | N-(3-Aminomethyl-phenyl)-2-methylsu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50095181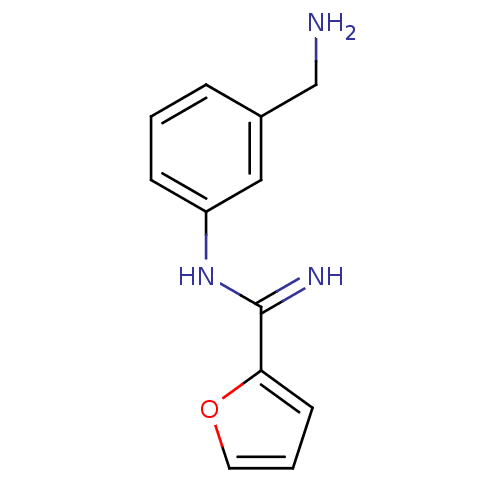 (CHEMBL96680 | N-(3-Aminomethyl-phenyl)-furan-2-car...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition against endothelial Nitric Oxide Synthase(eNOS) | Bioorg Med Chem Lett 10: 2771-4 (2000) BindingDB Entry DOI: 10.7270/Q23B60NS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065807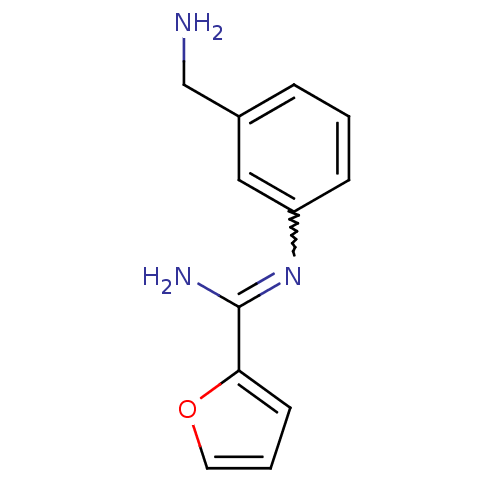 (CHEMBL552825 | N-(3-Aminomethyl-phenyl)-furan-2-ca...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065843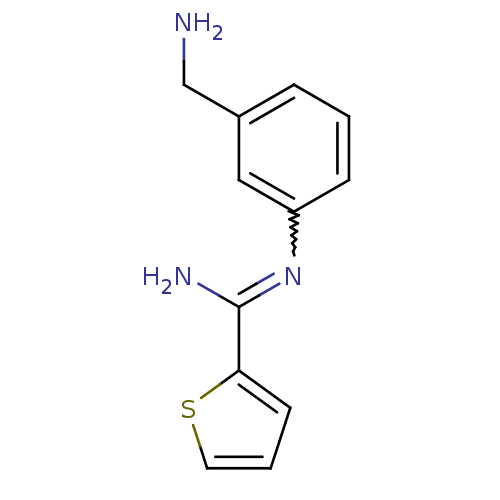 (CHEMBL553081 | CHEMBL555715 | N-(3-Aminomethyl-phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50230993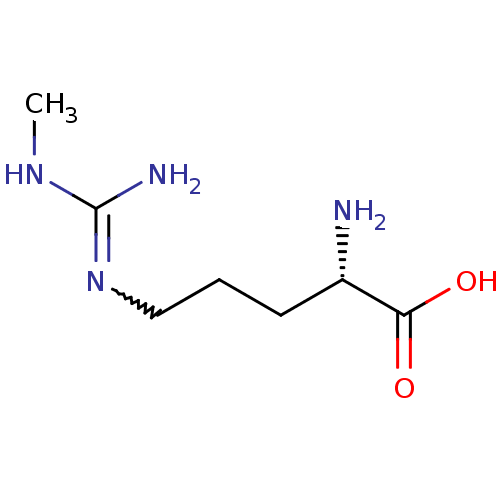 ((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50230993 ((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058459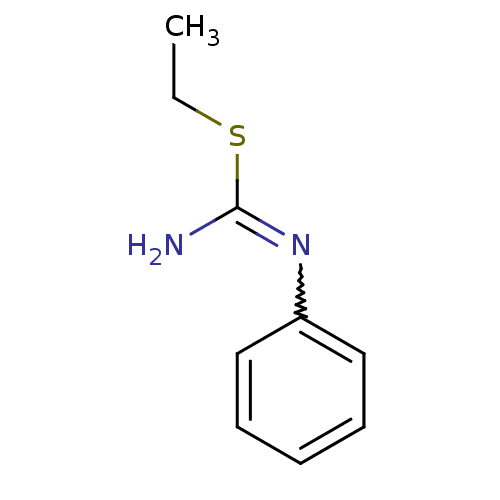 (2-Ethyl-1-phenyl-isothiourea; hydriodide | CHEMBL4...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50095172 (CHEMBL262040 | N-(3-Aminomethyl-phenyl)-thiophene-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition against endothelial Nitric Oxide Synthase(eNOS) | Bioorg Med Chem Lett 10: 2771-4 (2000) BindingDB Entry DOI: 10.7270/Q23B60NS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258687 (CHEMBL4092027) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 468 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50449048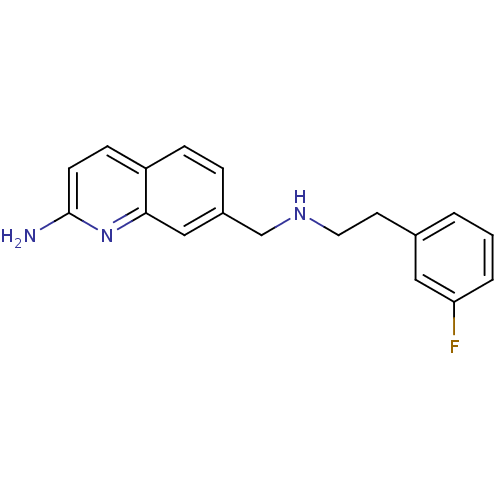 (CHEMBL3126213 | US9212144, 5 (Ex. 8)) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB US Patent | 485 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Northwestern University US Patent | Assay Description To test for enzyme inhibition, the hemoglobin capture assay was used to measure nitric oxide production. The assay was performed at 37� C. in HEPES b... | US Patent US9212144 (2015) BindingDB Entry DOI: 10.7270/Q2K0732P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50449048 (CHEMBL3126213 | US9212144, 5 (Ex. 8)) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 485 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of bovine recombinant eNOS expressed in Escherichia coli using L-arginine as substrate assessed as NO production by hemoglobin capture ass... | J Med Chem 57: 1513-30 (2014) Article DOI: 10.1021/jm401838x BindingDB Entry DOI: 10.7270/Q2MP54SZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50095177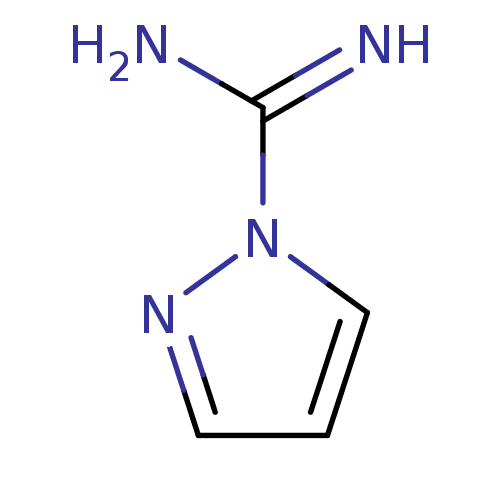 (CHEMBL503356 | CHEMBL542185 | Pyrazole-1-carboxami...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | PubMed | 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition against endothelial Nitric Oxide Synthase(eNOS) | Bioorg Med Chem Lett 10: 2771-4 (2000) BindingDB Entry DOI: 10.7270/Q23B60NS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Rattus norvegicus) | BDBM50180871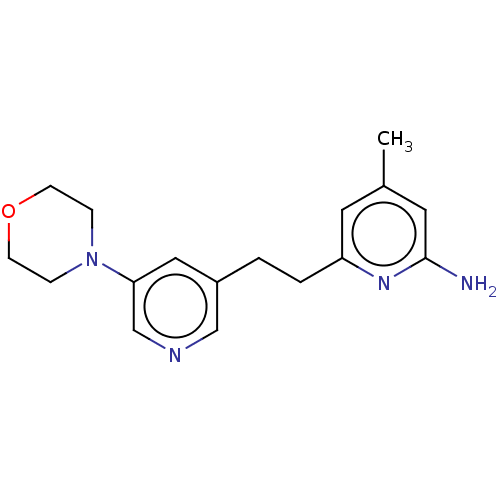 (CHEMBL3819644 | US10759791, Compound 14c) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | US Patent | 547 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258722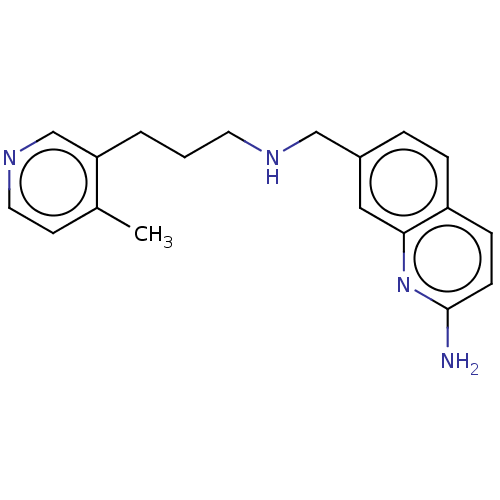 (CHEMBL4092616) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | 562 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258688 (CHEMBL4062164) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 581 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50225106 ((2S)-2-amino-5-{[(E)-amino(nitroimino)methyl]amino...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 720 | -35.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
Northwestern University | Assay Description Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... | J Med Chem 50: 2089-99 (2007) Article DOI: 10.1021/jm061305c BindingDB Entry DOI: 10.7270/Q2W0947W | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50203163 ((S)-2-((S)-4-amino-2-((S)-2,4-diamino-4-oxobutanam...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of bovine recombinant eNOS expressed in Escherichia coli assessed as nitric oxide production by hemoglobin capture assay | Bioorg Med Chem 15: 1928-38 (2007) Article DOI: 10.1016/j.bmc.2007.01.001 BindingDB Entry DOI: 10.7270/Q2QF8SJB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50225106 ((2S)-2-amino-5-{[(E)-amino(nitroimino)methyl]amino...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of bovine recombinant eNOS | J Med Chem 53: 7804-24 (2010) Article DOI: 10.1021/jm100947x BindingDB Entry DOI: 10.7270/Q2NC61FT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50061232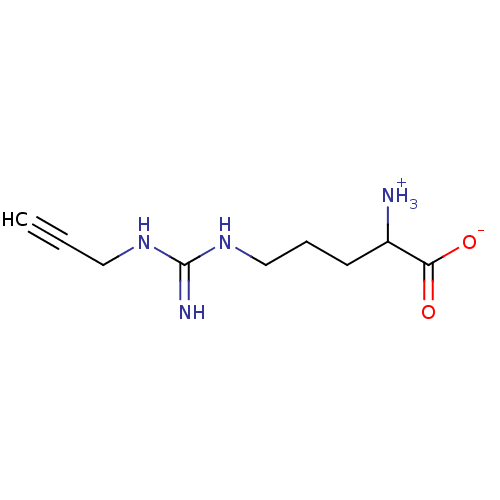 (2-Amino-5-(N'-prop-2-ynyl-guanidino)-pentanoic aci...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Compound was tested for binding affinity against Endothelial nitric oxide synthase | J Med Chem 40: 3869-70 (1998) Article DOI: 10.1021/jm970550g BindingDB Entry DOI: 10.7270/Q2C829ZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50072297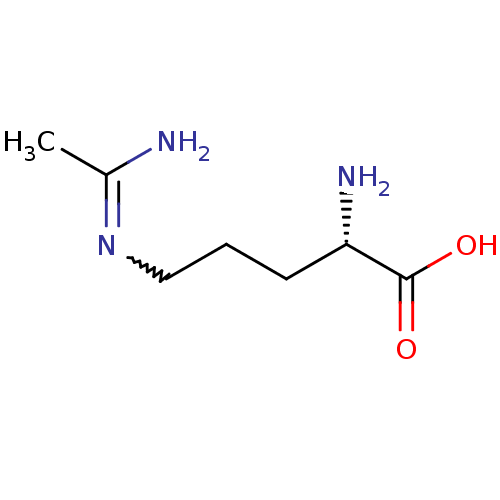 ((S)-5-Acetimidoylamino-2-amino-pent | (S)-5-Acetim...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article | 810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM196648 (US9212144, 3 | US9242957, 65) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB US Patent | 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description The compounds were evaluated for in vitro inhibition against three NOS isozymes: rat nNOS, bovine eNOS and marine iNOS using known literature methods... | US Patent US9242957 (2016) BindingDB Entry DOI: 10.7270/Q24B3047 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM196648 (US9212144, 3 | US9242957, 65) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB US Patent | 900 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Northwestern University US Patent | Assay Description To test for enzyme inhibition, the hemoglobin capture assay was used to measure nitric oxide production. The assay was performed at 37� C. in HEPES b... | US Patent US9212144 (2015) BindingDB Entry DOI: 10.7270/Q2K0732P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058469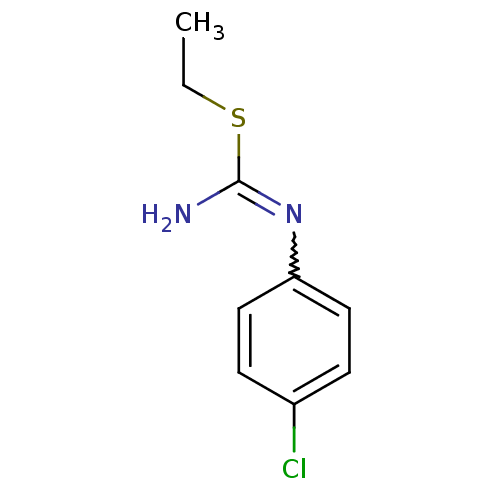 (1-(4-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50448833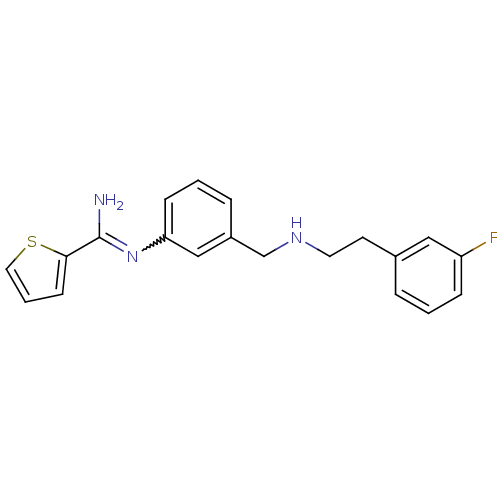 (CHEMBL3125048) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS overexpressed in Escherichia coli using L-arginine as substrate assessed as nitric oxide formation measured for... | J Med Chem 57: 686-700 (2014) Article DOI: 10.1021/jm401252e BindingDB Entry DOI: 10.7270/Q2SF2XNP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50448833 (CHEMBL3125048) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of bovine recombinant eNOS expressed in Escherichia coli using L-arginine as substrate assessed as NO production by hemoglobin capture ass... | J Med Chem 57: 1513-30 (2014) Article DOI: 10.1021/jm401838x BindingDB Entry DOI: 10.7270/Q2MP54SZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50438638 (CHEMBL2414426) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 979 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate assessed as formation of nitric oxide measured up t... | Bioorg Med Chem 21: 5323-31 (2013) Article DOI: 10.1016/j.bmc.2013.06.014 BindingDB Entry DOI: 10.7270/Q2PZ5B7N | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058443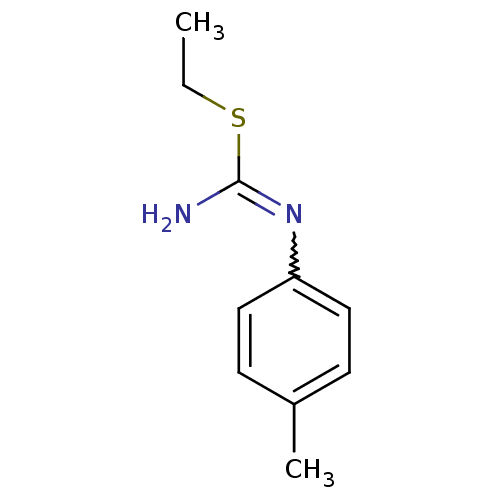 (2-Ethyl-1-p-tolyl-isothiourea; hydrochloride | CHE...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50258688 (CHEMBL4062164) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 1.03E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant human eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258623 (CHEMBL4071836) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 1.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065813 (CHEMBL555584 | N-(3-Aminomethyl-phenyl)-2-fluoro-a...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50023386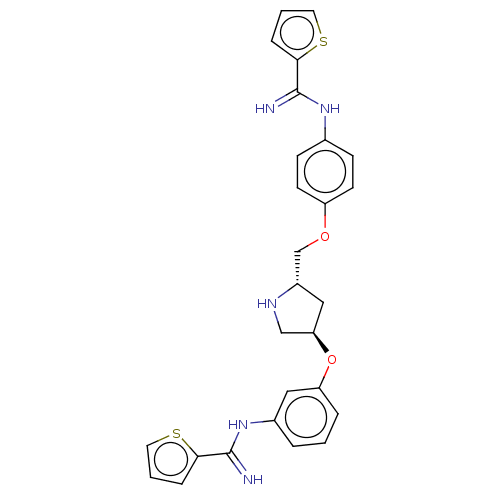 (CHEMBL3327299) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of bovine eNOS using L-arginine substrate | Bioorg Med Chem Lett 24: 4504-10 (2014) Article DOI: 10.1016/j.bmcl.2014.07.079 BindingDB Entry DOI: 10.7270/Q2542Q57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM202653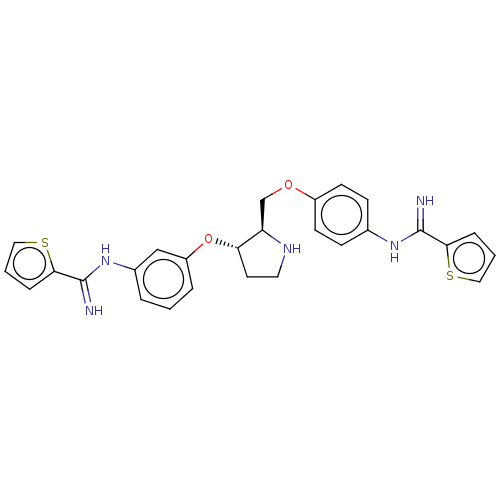 (US9242957, QJ-III-33) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 1.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description The compounds were evaluated for in vitro inhibition against three NOS isozymes: rat nNOS, bovine eNOS and marine iNOS using known literature methods... | US Patent US9242957 (2016) BindingDB Entry DOI: 10.7270/Q24B3047 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50258600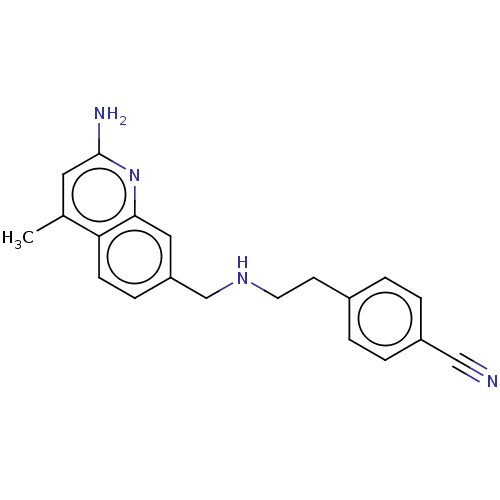 (CHEMBL4064297) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 1.22E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50341684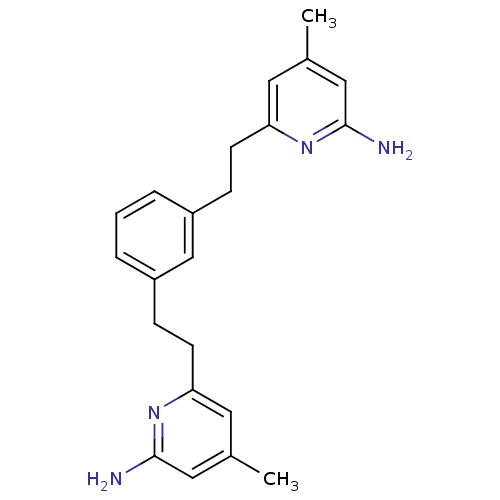 (6,6'-(2,2'-(1,3-Phenylene)bis(ethane-2,1-diyl))bis...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of bovine recombinant eNOS expressed in Escherichia coli by UV-vis spectrometric analysis | J Med Chem 54: 2039-48 (2011) Article DOI: 10.1021/jm101071n BindingDB Entry DOI: 10.7270/Q2571CB0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50341684 (6,6'-(2,2'-(1,3-Phenylene)bis(ethane-2,1-diyl))bis...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | US Patent | 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50341684 (6,6'-(2,2'-(1,3-Phenylene)bis(ethane-2,1-diyl))bis...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | US Patent | 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Enzyme inhibition was evaluated by measuring NO production with the hemoglobin capture assay, which was performed with purified NOSs in 96-well plate... | US Patent US10759791 (2020) BindingDB Entry DOI: 10.7270/Q2ZW1PZG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 1828 total ) | Next | Last >> |