

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50187243 (17-ethinyl-3,17-estradiol | 17-ethinyl-3,17-oestra...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 8.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Inhibition of human SULT1A1 expressed in Escherichia coli assessed as p-nitrophenol sulfation at 100 nM by Michaelis-Menten equation analysis | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50187243 (17-ethinyl-3,17-estradiol | 17-ethinyl-3,17-oestra...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Inhibition of human SULT1A1 expressed in Escherichia coli assessed as 17beta-estradiol sulfation at 100 nM by Michaelis-Menten equation analysis | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50187243 (17-ethinyl-3,17-estradiol | 17-ethinyl-3,17-oestra...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Inhibition of human SULT1A1 expressed in Escherichia coli assessed as beta-naphthol sulfation at 100 nM by Michaelis-Menten equation analysis | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM22870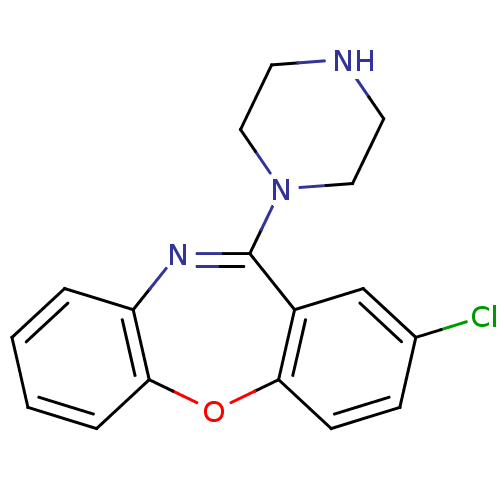 (13-chloro-10-(piperazin-1-yl)-2-oxa-9-azatricyclo[...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 3.50E+4 | -23.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 2 |
Albert Einstein College of Medicine | Assay Description Reaction conditions were as follows: SULT1A1 or 2A1 (50 nM), PnP (3.0 or 100 uM, respectively; 2 x Km PnP), amoxipine or protriptyline (0, 50, 100, o... | J Biol Chem 288: 34494-501 (2013) Article DOI: 10.1074/jbc.M113.510974 BindingDB Entry DOI: 10.7270/Q2XP73S3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50176062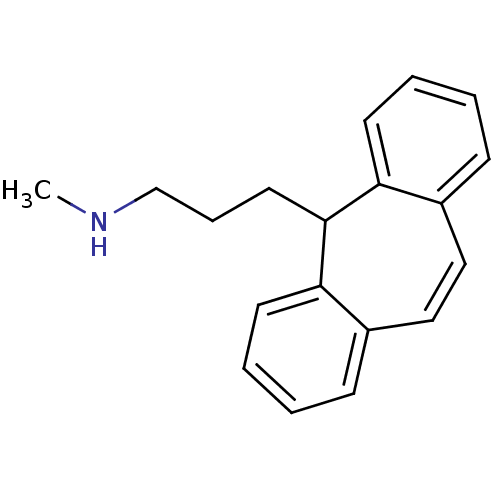 (3-(5H-dibenzo[a,d][7]annulen-5-yl)-N-methylpropan-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents | Article PubMed | 5.50E+4 | -22.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 2 |
Albert Einstein College of Medicine | Assay Description Reaction conditions were as follows: SULT1A1 or 2A1 (50 nM), PnP (3.0 or 100 uM, respectively; 2 x Km PnP), amoxipine or protriptyline (0, 50, 100, o... | J Biol Chem 288: 34494-501 (2013) Article DOI: 10.1074/jbc.M113.510974 BindingDB Entry DOI: 10.7270/Q2XP73S3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50187243 (17-ethinyl-3,17-estradiol | 17-ethinyl-3,17-oestra...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Inhibition of SULT1A1 in human MCF7 cells assessed as 17beta-estradiol sulfation | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Rattus norvegicus) | BDBM7460 (2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chrome...) | MMDB KEGG B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Rattus norvegicus) | BDBM23420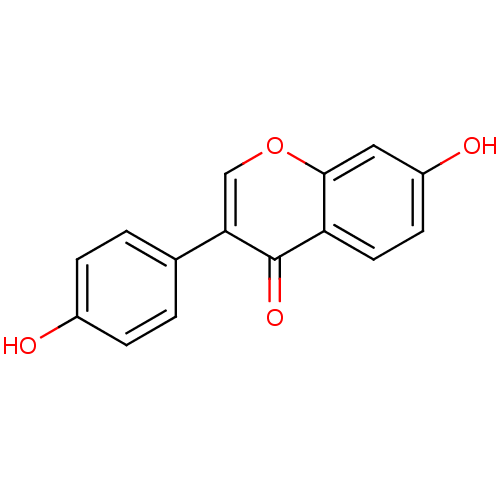 (7,4′-Dihydroxy-isoflavone (3a) | 7-hydroxy-3...) | MMDB KEGG B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Rattus norvegicus) | BDBM7462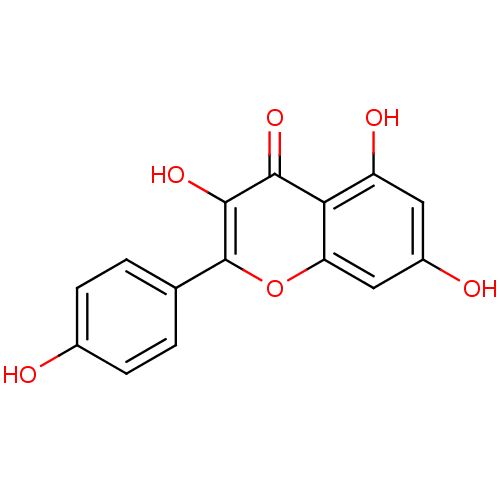 (3,5,7-trihydroxy-2-(4-hydroxyphenyl)-4H-chromen-4-...) | MMDB KEGG B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Rattus norvegicus) | BDBM19459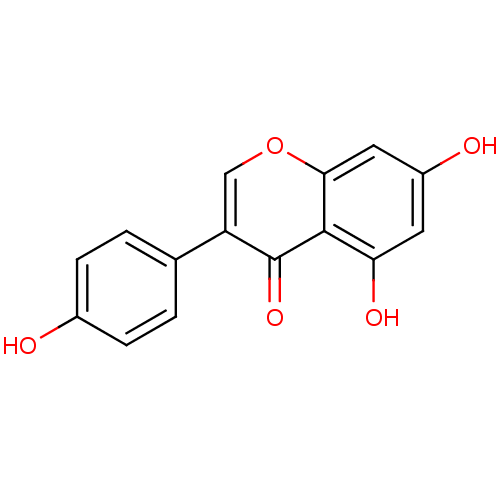 (5,7-dihydroxy-3-(4-hydroxyphenyl)-4H-chromen-4-one...) | MMDB KEGG B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM17292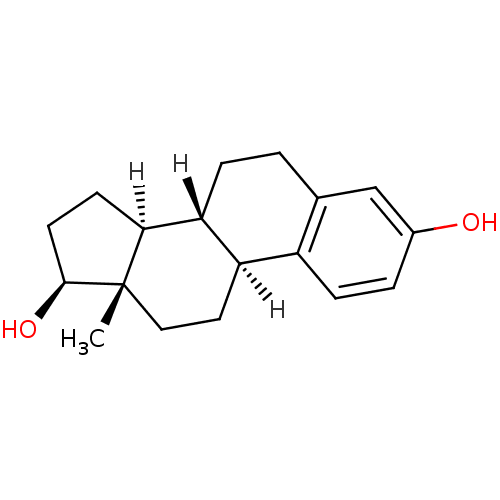 ((1S,10R,11S,14S,15S)-15-methyltetracyclo[8.7.0.0^{...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Binding affinity to human SULT1A1 expressed in Escherichia coli assessed as change in intrinsic fluorescence in presence of 3',5'-phosphoadenosine | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM17292 ((1S,10R,11S,14S,15S)-15-methyltetracyclo[8.7.0.0^{...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Binding affinity to human SULT1A1 expressed in Escherichia coli assessed as change in intrinsic fluorescence | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50187243 (17-ethinyl-3,17-estradiol | 17-ethinyl-3,17-oestra...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Binding affinity to human SULT1A1 expressed in Escherichia coli assessed as change in intrinsic fluorescence | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sulfotransferase 1A1 (Homo sapiens (Human)) | BDBM50187243 (17-ethinyl-3,17-estradiol | 17-ethinyl-3,17-oestra...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | 4.30 | n/a | n/a | n/a | n/a | n/a |
University of Alabama at Birmingham Curated by ChEMBL | Assay Description Binding affinity to human SULT1A1 expressed in Escherichia coli assessed as change in intrinsic fluorescence in presence of 3',5'-phosphoadenosine | Drug Metab Dispos 40: 1588-95 (2012) Article DOI: 10.1124/dmd.112.045583 BindingDB Entry DOI: 10.7270/Q2T43VTP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||