

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Urotensin-2 receptor (RAT) | BDBM50320463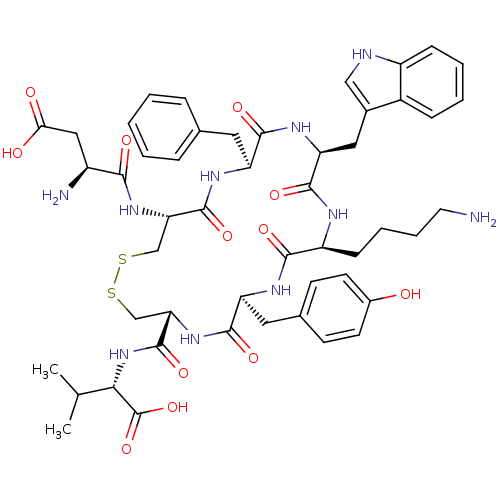 (CHEMBL218994 | D[CFWKYC]V | H-Asp-Cys-Phe-Trp-Lys-...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
Johnson& Johnson Pharmaceutical Research& Development Curated by ChEMBL | Assay Description Agonist activity at rat urotensin 2 receptor expressed in CHO cells assessed as calcium mobilization by FLIPR | J Med Chem 53: 2695-708 (2010) Article DOI: 10.1021/jm901294u BindingDB Entry DOI: 10.7270/Q20G3K9N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urotensin-2 receptor (Homo sapiens (Human)) | BDBM50320463 (CHEMBL218994 | D[CFWKYC]V | H-Asp-Cys-Phe-Trp-Lys-...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a |
University of Naples "Federico II" Curated by ChEMBL | Assay Description Agonist activity at urotensin 2 receptor (unknown origin) expressed in HEK293 cells co-expressing Gq-polycistronic BRET biosensor assessed as inducti... | J Med Chem 62: 1455-1467 (2019) Article DOI: 10.1021/acs.jmedchem.8b01601 BindingDB Entry DOI: 10.7270/Q2PV6PRV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urotensin-2 receptor (RAT) | BDBM50320463 (CHEMBL218994 | D[CFWKYC]V | H-Asp-Cys-Phe-Trp-Lys-...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a |
University of Naples"Federico II" Curated by ChEMBL | Assay Description Agonist activity at UT receptor in rat aorta assessed as contraction | J Med Chem 57: 5965-74 (2014) Article DOI: 10.1021/jm500218x BindingDB Entry DOI: 10.7270/Q2MC91NB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urotensin-2 receptor (RAT) | BDBM50320463 (CHEMBL218994 | D[CFWKYC]V | H-Asp-Cys-Phe-Trp-Lys-...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 5.20 | n/a | n/a | n/a | n/a |
University of Naples "Federico II" Curated by ChEMBL | Assay Description Agonist activity at urotensin 2 receptor in Sprague-Dawley rat thoracic aortic ring assessed as induction of contraction relative to KCl-induced cont... | J Med Chem 62: 1455-1467 (2019) Article DOI: 10.1021/acs.jmedchem.8b01601 BindingDB Entry DOI: 10.7270/Q2PV6PRV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||