 Found 12 hits of ic50 for monomerid = 136570
Found 12 hits of ic50 for monomerid = 136570 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Beta-secretase 1
(Homo sapiens (Human)) | BDBM136570
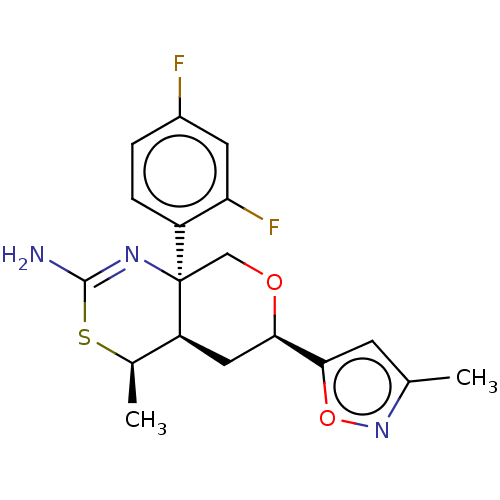
(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 (unknown origin) using Biotin-GLTNIKTEEISEISYEVEFR-C[oregon green]KK-OH substrate assessed as fluorescence polarization by cell f... |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 in human H4 cells overexpressing wild type human APP695 assessed as colorimetric reaction by Whole cell assay |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 75.9 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
US Patent
| Assay Description
Beta-secretase (BACE) is one of the enzymes involved in the generation of the amyloid beta peptide found in the amyloid plaques of Alzheimer's Diseas... |
US Patent US8865706 (2014)
BindingDB Entry DOI: 10.7270/Q2FX7855 |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co. Inc.
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D (unknown origin) by cell based assay |
J Med Chem 61: 1767-1773 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00762
BindingDB Entry DOI: 10.7270/Q2C53P79 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human ERG expressed in CHO cells |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co. Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant cathepsin D (unknown origin) |
J Med Chem 61: 1767-1773 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00762
BindingDB Entry DOI: 10.7270/Q2C53P79 |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.42E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of CatD (unknown origin) assessed as fluorescence polarization by cell free assay |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.42E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C8 (unknown origin) |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM136570

(US8865706, 9)Show SMILES C[C@H]1SC(N)=N[C@]2(CO[C@H](C[C@@H]12)c1cc(C)no1)c1ccc(F)cc1F |c:4| Show InChI InChI=1S/C18H19F2N3O2S/c1-9-5-16(25-23-9)15-7-13-10(2)26-17(21)22-18(13,8-24-15)12-4-3-11(19)6-14(12)20/h3-6,10,13,15H,7-8H2,1-2H3,(H2,21,22)/t10-,13+,15-,18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 58: 3223-52 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00191
BindingDB Entry DOI: 10.7270/Q29W0H68 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data