

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A disintegrin and metalloproteinase with thrombospondin motifs 5 (Homo sapiens (Human)) | BDBM194646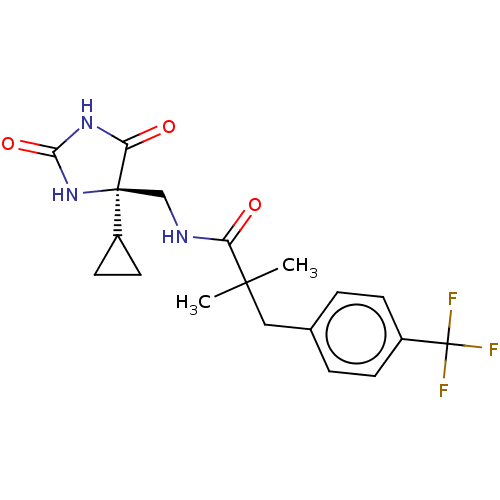 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 4 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Eli Lilly and Company US Patent | Assay Description The compounds of the present invention can be evaluated by using an aggrecanase ADAMTS-4 and ADAMTS-5 AlphaScreen assay (Miller J. A., et al. Anal. B... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| A disintegrin and metalloproteinase with thrombospondin motifs 5 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human ADAMTS5 using VQTVTWPDMELPLPRNITEGEARGSVILTVKPIFEVSPSPLKG peptide as substrate after 3 hrs by Alphascreen assay | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| A disintegrin and metalloproteinase with thrombospondin motifs 4 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 7 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Eli Lilly and Company US Patent | Assay Description The compounds of the present invention can be evaluated by using an aggrecanase ADAMTS-4 and ADAMTS-5 AlphaScreen assay (Miller J. A., et al. Anal. B... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| A disintegrin and metalloproteinase with thrombospondin motifs 4 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human ADAMTS4 using VQTVTWPDMELPLPRNITEGEARGSVILTVKPIFEVSPSPLKG peptide as substrate after 3 hrs by Alphascreen assay | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 54 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADAM metallopeptidase with thrombospondin type 1 motif, 5 (Rattus norvegicus (Rat)) | BDBM194646 (US9206139, 5) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company US Patent | Assay Description The AlphaScreen Assay is modified to include the testing of inhibitors against ADAMTS-5 in the presence of 50% Lewis rat plasma in order to determine... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| A disintegrin and metalloproteinase with thrombospondin motifs 5 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human ADAMTS5 using VQTVTWPDMELPLPRNITEGEARGSVILTVKPIFEVSPSPLKG peptide as substrate after 3 hrs in the presence of 50% Lewis rat plasm... | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Competitive inhibition against rat cytoplasmic Thymidine kinase | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 71 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 4.55E+3 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 4.65E+3 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human MMP2 using Mca-PQGL-(3-[2, 4-dinitrophenyl]-L-2, 3-diaminopropionyl)-AR-OH as substrate after 2 to 4 hrs by fluorescence assay | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human MMP13 using Mca-PQGL-(3-[2, 4-dinitrophenyl]-L-2, 3-diaminopropionyl)-AR-OH as substrate after 2 to 4 hrs by fluorescence assay | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 4.81E+3 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 7.59E+3 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Stromelysin-1 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human MMP3 using Mca-PQGL-(3-[2, 4-dinitrophenyl]-L-2, 3-diaminopropionyl)-AR-OH as substrate after 2 to 4 hrs by fluorescence assay | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Stromelysin-1 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.32E+4 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.49E+4 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description In vitro inhibitory activity against S-adenosyl-L-methionine decarboxylase using liver from rat in absence of putrescine | J Med Chem 60: 5933-5939 (2017) Article DOI: 10.1021/acs.jmedchem.7b00650 BindingDB Entry DOI: 10.7270/Q2Z321XM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrilysin (Homo sapiens (Human)) | BDBM194646 (US9206139, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | 7.5 | n/a |
Eli Lilly and Company US Patent | Assay Description A continuous assay is used in which the substrate is a synthetic peptide containing a fluorescent group (7-methoxycoumarin, Mca), which is quenched b... | US Patent US9206139 (2015) BindingDB Entry DOI: 10.7270/Q25X27R0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||