

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50017698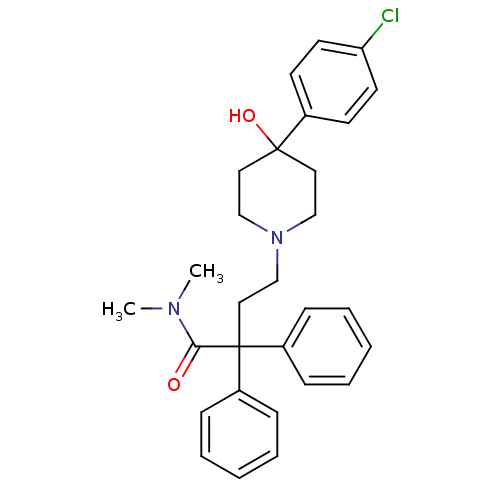 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
F-59000 Lille Curated by ChEMBL | Assay Description Binding affinity against mu-opiate receptor (human) using [3H]DAMGO radioligand | J Med Chem 44: 3378-90 (2001) BindingDB Entry DOI: 10.7270/Q23X87B8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human ERG expressed in HEK293 cells at holding potential of -80 mV by whole cell patch clamp assay | Citation and Details Article DOI: 10.1016/j.bmcl.2017.12.020 BindingDB Entry DOI: 10.7270/Q2HM5C1C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank PubMed | n/a | n/a | 123 | n/a | n/a | n/a | n/a | n/a | n/a |
F-59000 Lille Curated by ChEMBL | Assay Description Binding affinity against delta-opiate receptor (human) using [3H]-DPDPE radioligand | J Med Chem 44: 3378-90 (2001) BindingDB Entry DOI: 10.7270/Q23X87B8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank PubMed | n/a | n/a | 155 | n/a | n/a | n/a | n/a | n/a | n/a |
F-59000 Lille Curated by ChEMBL | Assay Description Binding affinity against opioid receptor kappa 1 using [3H]- U-69,593 radioligand | J Med Chem 44: 3378-90 (2001) BindingDB Entry DOI: 10.7270/Q23X87B8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium channel protein type 5 subunit alpha (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 239 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human Nav1.5 channel expressed in HEK293 cells at holding potential of -70 mV by whole cell patch clamp assay | Citation and Details Article DOI: 10.1016/j.bmcl.2017.12.020 BindingDB Entry DOI: 10.7270/Q2HM5C1C | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium channel protein type 1/2/3 subunit alpha (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of binding of Batrachotoxinin [3H]BTX-B to high affinity sites on voltage dependent sodium channels in a vesicular preparation from guinea... | J Med Chem 28: 381-8 (1985) BindingDB Entry DOI: 10.7270/Q2Z321T8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ATP-dependent translocase ABCB1 (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vanderbilt University Curated by ChEMBL | Assay Description TP_TRANSPORTER: inhibition of Digoxin transepithelial transport (basal to apical) (Digoxin: 5 uM) in Caco-2 cells | Anesthesiology 96: 913-20 (2002) BindingDB Entry DOI: 10.7270/Q27D2WDT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cocaine esterase (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | NCI pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.06E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Medical Sciences and Peking Union Medical College Curated by ChEMBL | Assay Description Inhibition of CE2 in human liver microsomes using fluorescein diacetate as substrate preincubated for 10 mins followed by substrate addition measured... | Eur J Med Chem 145: 717-725 (2018) Article DOI: 10.1016/j.ejmech.2018.01.030 BindingDB Entry DOI: 10.7270/Q2542R46 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cocaine esterase (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | NCI pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Medical Sciences and Peking Union Medical College Curated by ChEMBL | Assay Description Inhibition of human CE2 using fluorescein diacetate as substrate preincubated for 10 mins followed by substrate addition and measured after 10 mins b... | J Nat Prod 83: 489-496 (2020) Article DOI: 10.1021/acs.jnatprod.9b01155 BindingDB Entry DOI: 10.7270/Q2571GJX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cocaine esterase (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | NCI pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of hCES2A in human liver microsome assessed as reduction in fluorescein diacetate hydrolysis preincubated for 10 mins followed by substrat... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112856 BindingDB Entry DOI: 10.7270/Q2NZ8CB7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (2019-nCoV) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut Pasteur Korea | Assay Description Ten-point DRCs were generated for each drug. Vero cells were seeded at 1.2 � 104 cells per well in DMEM, supplemented with 2% FBS and 1� ... | Antimicrob Agents Chemother 64: (2020) Article DOI: 10.1128/AAC.00819-20 BindingDB Entry DOI: 10.7270/Q22N54QR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calmodulin (Bos taurus) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Bari 'Aldo Moro' Curated by ChEMBL | Assay Description Displacement of [3H]trifluoperazine from bovine brain CaM in presence of calcium | Eur J Med Chem 116: 36-45 (2016) Article DOI: 10.1016/j.ejmech.2016.03.045 BindingDB Entry DOI: 10.7270/Q2Q2424Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier family 22 member 1 (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.37E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Uppsala University Curated by ChEMBL | Assay Description Inhibition of 4-(4-(dimethylamino)styryl)-N-methylpyridinium uptake at human OCT1 expressed in HEK293 cells by confocal microscopy | J Med Chem 51: 5932-42 (2008) Article DOI: 10.1021/jm8003152 BindingDB Entry DOI: 10.7270/Q23779MD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide Y receptor type 1 (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | PCBioAssay | n/a | n/a | >3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Molecular Screening Center Curated by PubChem BioAssay | Assay Description Source (MLSCN Center Name): The Scripps Research Institute Molecular Screening Center Affiliation: The Scripps Research Institute, TSRI Assay Provide... | PubChem Bioassay (2008) BindingDB Entry DOI: 10.7270/Q2Q81BGP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bile salt export pump (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human BSEP expressed in baculovirus infected sf9 cell membrane vesicles assessed as reduction in ATP or AMP-dependent [3H]-... | Hepatology 60: 1015-22 (2014) Article DOI: 10.1002/hep.27206 BindingDB Entry DOI: 10.7270/Q2TF00N2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Menin (Homo sapiens (Human)) | BDBM50017698 (4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)-N,N...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of FITC-MBM1 from full length human menin measured after 2 hrs by fluorescence polarization assay | Bioorg Med Chem 26: 356-365 (2018) Article DOI: 10.1016/j.bmc.2017.11.045 BindingDB Entry DOI: 10.7270/Q2SQ92ZS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||