 Found 14 hits of ic50 for monomerid = 50126829
Found 14 hits of ic50 for monomerid = 50126829 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Malate dehydrogenase
(Thermus thermophilus) | BDBM50126829
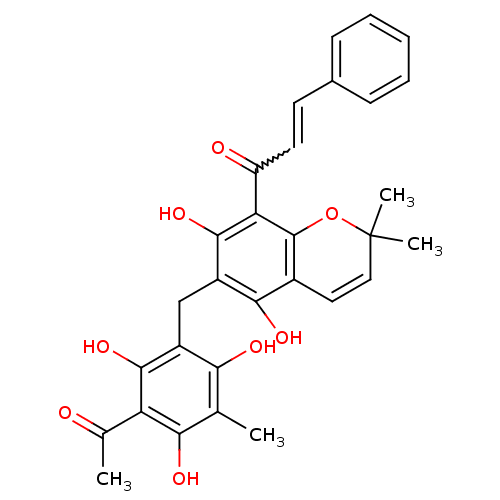
((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibitory activity against malate dehydrogenase (MDH) from Thermus flavus |
J Med Chem 46: 1478-83 (2003)
Article DOI: 10.1021/jm020427b
BindingDB Entry DOI: 10.7270/Q2R78FZT |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Escherichia coli) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibitory activity against Amp C beta-Lactamase |
J Med Chem 46: 4265-72 (2003)
Article DOI: 10.1021/jm030266r
BindingDB Entry DOI: 10.7270/Q29C6Z59 |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 5
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Dundee
Curated by ChEMBL
| Assay Description
Inhibition of His-tagged human PRAK expressed in Sf9 cells |
Biochem J 351: 95-105 (2001)
BindingDB Entry DOI: 10.7270/Q24T6JKN |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 5
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibition of p38-regulated activated kinase (PRAK) |
J Med Chem 46: 1478-83 (2003)
Article DOI: 10.1021/jm020427b
BindingDB Entry DOI: 10.7270/Q2R78FZT |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C delta (PKCdelta) |
J Med Chem 46: 1478-83 (2003)
Article DOI: 10.1021/jm020427b
BindingDB Entry DOI: 10.7270/Q2R78FZT |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01031
BindingDB Entry DOI: 10.7270/Q2FF3XF8 |
More data for this
Ligand-Target Pair | |
Eukaryotic elongation factor 2 kinase
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of eEF2K (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112505
BindingDB Entry DOI: 10.7270/Q2M90D99 |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Dundee
Curated by ChEMBL
| Assay Description
Inhibition of His-tagged human MAPKAPK2 expressed in Escherichia coli at 10 uM |
Biochem J 351: 95-105 (2001)
BindingDB Entry DOI: 10.7270/Q24T6JKN |
More data for this
Ligand-Target Pair | |
WD repeat-containing protein 48
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Connecticut Health Center
Curated by ChEMBL
| Assay Description
Inhibition of human USP1/UAF1 complex using Ub-Rho as substrate by qHTS assay |
J Med Chem 59: 9321-9336 (2016)
BindingDB Entry DOI: 10.7270/Q2ZC84TJ |
More data for this
Ligand-Target Pair | |
RuvB-like 1
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.116726
BindingDB Entry DOI: 10.7270/Q21G0R9W |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type/glutamine gamma-glutamyltransferase 2
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | 37 |
Duke University Medical Center
| Assay Description
In order to eliminated fluorescence interference, chemicals were subjected to a secondary screening using colorimetric BP incorporation assay. TGase... |
Chem Biol 15: 969-78 (2008)
Article DOI: 10.1016/j.chembiol.2008.07.015
BindingDB Entry DOI: 10.7270/Q2DR2SZR |
More data for this
Ligand-Target Pair | |
Chymotrypsinogen B
(Homo sapiens (Human)) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibitory activity against Chymotrypsinogen from Thermus flavus |
J Med Chem 46: 1478-83 (2003)
Article DOI: 10.1021/jm020427b
BindingDB Entry DOI: 10.7270/Q2R78FZT |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Enterobacter cloacae) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyushu University
Curated by ChEMBL
| Assay Description
Inhibition of Enterobacter cloacae beta-lactamase incubated for 10 mins followed by nitrocefin substrate challenge and measured for 5 mins by spectro... |
ACS Med Chem Lett 10: 923-928 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00093
BindingDB Entry DOI: 10.7270/Q2M90CXZ |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Enterobacter cloacae) | BDBM50126829

((E)-1-(6-(3-acetyl-2,4,6-trihydroxy-5-methylbenzyl...)Show SMILES CC(=O)c1c(O)c(C)c(O)c(Cc2c(O)c3C=CC(C)(C)Oc3c(C(=O)C=Cc3ccccc3)c2O)c1O |w:26.26,c:16| Show InChI InChI=1S/C30H28O8/c1-15-24(33)19(27(36)22(16(2)31)25(15)34)14-20-26(35)18-12-13-30(3,4)38-29(18)23(28(20)37)21(32)11-10-17-8-6-5-7-9-17/h5-13,33-37H,14H2,1-4H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.42E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyushu University
Curated by ChEMBL
| Assay Description
Inhibition of Enterobacter cloacae beta-lactamase incubated for 10 mins followed by nitrocefin substrate challenge and measured for 5 mins in presenc... |
ACS Med Chem Lett 10: 923-928 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00093
BindingDB Entry DOI: 10.7270/Q2M90CXZ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data