 Found 10 hits of ic50 for monomerid = 50134315
Found 10 hits of ic50 for monomerid = 50134315 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetolactate synthase catalytic subunit, mitochondrial
(Saccharomyces cerevisiae) | BDBM50134315
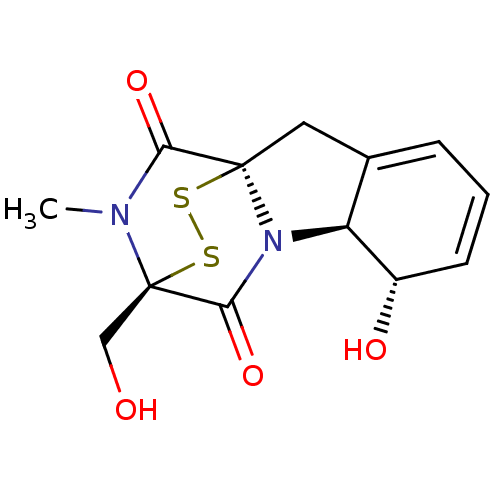
(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of Saccharomyces cerevisiae acetolactate synthase |
Bioorg Med Chem 18: 2566-74 (2010)
Article DOI: 10.1016/j.bmc.2010.02.034
BindingDB Entry DOI: 10.7270/Q22J6C0K |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H1
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H1 |
Eur J Med Chem 56: 179-194 (2012)
Article DOI: 10.1016/j.ejmech.2012.08.010
BindingDB Entry DOI: 10.7270/Q2TQ62NX |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase RNF31/RanBP-type and C3HC4-type zinc finger-containing protein 1/Sharpin
(Homo sapiens) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 510 | n/a | n/a | n/a | n/a | n/a | n/a |
Temple University
Curated by ChEMBL
| Assay Description
Inhibition of petit-LUBAC (unknown origin)-mediated ubiquitylation expressed in Escherichia coli BL21 after 2 hrs in presence of E1, UbcH5c, E3 and u... |
J Med Chem 61: 3239-3252 (2018)
Article DOI: 10.1021/acs.jmedchem.6b01817
BindingDB Entry DOI: 10.7270/Q2542R5N |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EHMT2
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 530 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of G9a |
Eur J Med Chem 56: 179-194 (2012)
Article DOI: 10.1016/j.ejmech.2012.08.010
BindingDB Entry DOI: 10.7270/Q2TQ62NX |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Activity against Farnesyltransferase |
J Med Chem 40: 2971-90 (1997)
Article DOI: 10.1021/jm970226l
BindingDB Entry DOI: 10.7270/Q26M37HF |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EHMT2
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Tokyo
Curated by ChEMBL
| Assay Description
Inhibition of HMT-G9a using S-adenosylmethionine and biotinylated H3 peptide after 1 hr |
J Nat Prod 75: 111-4 (2012)
Article DOI: 10.1021/np200740e
BindingDB Entry DOI: 10.7270/Q29G5N8K |
More data for this
Ligand-Target Pair | |
Geranylgeranyl transferase type-1 subunit beta/Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College of Science
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-GGPP incorporation into recombinant human Ha-Ras-CVLL by Geranylgeranyl transferase type I |
Bioorg Med Chem Lett 13: 3661-3 (2003)
BindingDB Entry DOI: 10.7270/Q2CF9PHX |
More data for this
Ligand-Target Pair | |
Pyruvate kinase PKM
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00981
BindingDB Entry DOI: 10.7270/Q2M330SP |
More data for this
Ligand-Target Pair | |
Mycothiol S-conjugate amidase
(Mycobacterium tuberculosis) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis mycothiol-S-conjugate amidase |
Bioorg Med Chem 18: 2566-74 (2010)
Article DOI: 10.1016/j.bmc.2010.02.034
BindingDB Entry DOI: 10.7270/Q22J6C0K |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50134315

(7-hydroxy-11-hydroxymethyl-12-methyl-14,15-dithia-...)Show SMILES CN1C(=O)[C@]23CC4=CC=C[C@H](O)[C@H]4N2C(=O)[C@@]1(CO)SS3 |r,c:8,t:6,THB:12:13:1.2:19.20,15:14:1.2:19.20| Show InChI InChI=1S/C13H14N2O4S2/c1-14-10(18)12-5-7-3-2-4-8(17)9(7)15(12)11(19)13(14,6-16)21-20-12/h2-4,8-9,16-17H,5-6H2,1H3/t8-,9-,12+,13+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College of Science
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into recombinant human Ha-Ras-CVLS by farnesyl transferase |
Bioorg Med Chem Lett 13: 3661-3 (2003)
BindingDB Entry DOI: 10.7270/Q2CF9PHX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data