

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Multidrug resistance-associated protein 1 (Homo sapiens (Human)) | BDBM50140849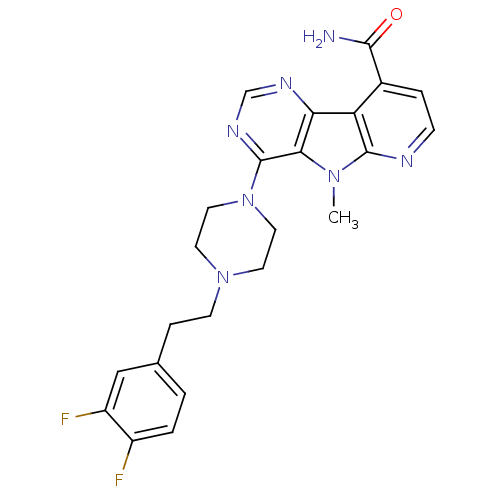 (8-{4-[2-(3,4-Difluoro-phenyl)-ethyl]-piperazin-1-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Xenova Ltd. Curated by ChEMBL | Assay Description Inhibitory activity against MRP1 (Multidrug resistance associated protein 1) expressed in COR.L23/R cell line in single-dose potentiation assay | J Med Chem 47: 1339-50 (2004) Article DOI: 10.1021/jm0310129 BindingDB Entry DOI: 10.7270/Q21Z43T3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Multidrug resistance-associated protein 1 (Homo sapiens (Human)) | BDBM50140849 (8-{4-[2-(3,4-Difluoro-phenyl)-ethyl]-piperazin-1-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Xenova Ltd. Curated by ChEMBL | Assay Description In vitro inhibitory activity against MRP1 (Multidrug resistance associated protein 1) expressed in COR.L23/R cell line in accumulation assay | J Med Chem 47: 1339-50 (2004) Article DOI: 10.1021/jm0310129 BindingDB Entry DOI: 10.7270/Q21Z43T3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Multidrug resistance-associated protein 1 (Homo sapiens (Human)) | BDBM50140849 (8-{4-[2-(3,4-Difluoro-phenyl)-ethyl]-piperazin-1-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | <250 | n/a | n/a | n/a | n/a | n/a | n/a |
M.D. University Curated by ChEMBL | Assay Description Inhibitory concentration against multidrug resistance associated protein 1 | Bioorg Med Chem Lett 15: 4967-72 (2005) Article DOI: 10.1016/j.bmcl.2005.08.011 BindingDB Entry DOI: 10.7270/Q2X34X15 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50140849 (8-{4-[2-(3,4-Difluoro-phenyl)-ethyl]-piperazin-1-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.23E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Xenova Ltd. Curated by ChEMBL | Assay Description Inhibitory activity against cytochrome P450 3A4 | J Med Chem 47: 1339-50 (2004) Article DOI: 10.1021/jm0310129 BindingDB Entry DOI: 10.7270/Q21Z43T3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ATP-dependent translocase ABCB1 (Mus musculus (Mouse)) | BDBM50140849 (8-{4-[2-(3,4-Difluoro-phenyl)-ethyl]-piperazin-1-y...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Xenova Ltd. Curated by ChEMBL | Assay Description Inhibitory activity against P-glycoprotein expressed in murine mammary carcinoma (EMT6/AR1.0 cell line) in single-dose potentiation assay | J Med Chem 47: 1339-50 (2004) Article DOI: 10.1021/jm0310129 BindingDB Entry DOI: 10.7270/Q21Z43T3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||