

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bromodomain-containing protein 9 (Homo sapiens (Human)) | BDBM50183448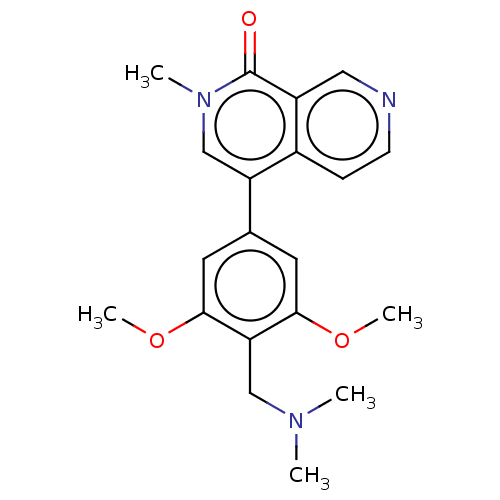 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human N-terminal GST-tagged BRD9 isoform 1 expressed in Escherichia coli using tetra-acetylated histone H4 measured after 60 mins by Al... | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Bromodomain-containing protein 9 [133-239] (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bromodomain-containing protein 7 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 117 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human N-terminal GST-tagged BRD7 expressed in Escherichia coli using tetra-acetylated histone H4 measured after 60 mins by AlphaScreen ... | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Bromodomain-containing protein 7 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 117 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human BRD7 measured after mins by AlphaScreen assay | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| TGF-beta receptor type-1 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 3.85E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of TGFBR1 (unknown origin) | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Activin receptor type-1 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 5.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of ACVR1 (unknown origin) | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Activin receptor type-2B (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of ACVR2B (unknown origin) | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C8 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of CYP2C8 in human liver microsomes in presence of NADPH using amodiaquine as substrate measured within 2.5 mins by LC-MS/MS analysis | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of CYP2D6 in human liver microsomes in presence of NADPH using dextromethorphan as substrate measured within 2.5 mins by LC-MS/MS analysis | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of CYP3A4 in human liver microsomes in presence of NADPH using midazolam as substrate measured within 2.5 mins by LC-MS/MS analysis | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C19 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of CYP2C19 in human liver microsomes in presence of NADPH using mephenytoin as substrate measured within 2.5 mins by LC-MS/MS analysis | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of CYP2C9 in human liver microsomes in presence of NADPH using diclofenac as substrate measured within 2.5 mins by LC-MS/MS analysis | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bromodomain-containing protein 4 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human BRD4-BD1 by alpha assay | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bromodomain-containing protein 4 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human N-terminal GST-tagged BRD4-BD2 expressed in Escherichia coli using tetra-acetylated histone H4 measured after 60 mins by AlphaScr... | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bromodomain-containing protein 2 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human N-terminal GST-tagged BRD2-BD1 expressed in Escherichia coli using tetra-acetylated histone H4 measured after 60 mins by AlphaScr... | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Bromodomain-containing protein 4 (Homo sapiens (Human)) | BDBM50183448 (CHEMBL3823478 | US11773085, Compound B2) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim RCV GmbH& Co KG Curated by ChEMBL | Assay Description Inhibition of human N-terminal GST-tagged BRD4-BD1 expressed in Escherichia coli using tetra-acetylated histone H4 measured after 60 mins by AlphaScr... | J Med Chem 59: 4462-75 (2016) Article DOI: 10.1021/acs.jmedchem.5b01865 BindingDB Entry DOI: 10.7270/Q27H1MJZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||