 Found 17 hits of ic50 for monomerid = 50242173
Found 17 hits of ic50 for monomerid = 50242173 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50242173
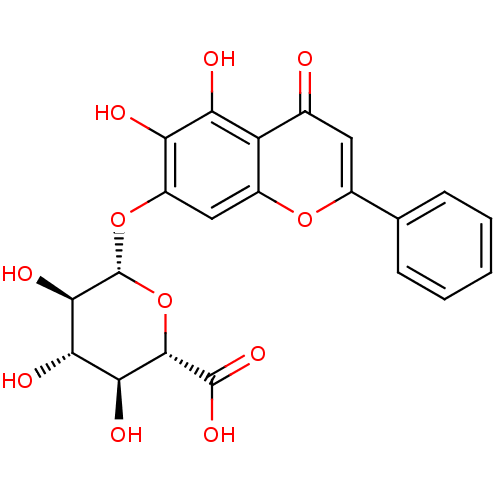
((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of LSD1 (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113501
BindingDB Entry DOI: 10.7270/Q2T72N77 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of LSD1 (unknown origin) expressed in human MGC-803 cells incubated for 5 days by DAPI staining based immunofluorescence method |
Citation and Details
Article DOI: 10.1039/d0md00141d
BindingDB Entry DOI: 10.7270/Q23R0XGK |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| | n/a | n/a | 3.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mean functional activity against human H3 receptor |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.55E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of LSD1 (unknown origin) |
Bioorg Med Chem 27: 370-374 (2019)
Article DOI: 10.1016/j.bmc.2018.12.013
BindingDB Entry DOI: 10.7270/Q2JW8J40 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK1
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.61E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00614
BindingDB Entry DOI: 10.7270/Q2Z89HD2 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
This is a review article. Please point to the original journal. |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00409
BindingDB Entry DOI: 10.7270/Q2J1069F |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115860
BindingDB Entry DOI: 10.7270/Q2154N01 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115860
BindingDB Entry DOI: 10.7270/Q2154N01 |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01140
BindingDB Entry DOI: 10.7270/Q2445RJD |
More data for this
Ligand-Target Pair | |
Replicase polyprotein 1ab
(2019-nCoV) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115860
BindingDB Entry DOI: 10.7270/Q2154N01 |
More data for this
Ligand-Target Pair | |
Prolyl endopeptidase
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Recerca Biomèdica de Barcelona
Curated by ChEMBL
| Assay Description
Inhibition of human brain prolyl oligopeptidase expressed in Escherichia coli |
Bioorg Med Chem 16: 7516-24 (2008)
Article DOI: 10.1016/j.bmc.2008.04.067
BindingDB Entry DOI: 10.7270/Q2ZC82NG |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.22E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Konkuk University
| Assay Description
The kinase assay was performed using the EMD Millipore KinaseProfiler service assay protocol. Aurora B kinase was supplied by EMD Millipore Corp. The... |
Chem Biol Drug Des 85: 574-85 (2015)
Article DOI: 10.1111/cbdd.12445
BindingDB Entry DOI: 10.7270/Q2M61J08 |
More data for this
Ligand-Target Pair | |
Prolyl endopeptidase
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.23E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
McGill University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant POP |
J Med Chem 53: 3423-38 (2010)
Article DOI: 10.1021/jm901104g
BindingDB Entry DOI: 10.7270/Q261119N |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 1
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.44E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Dalian University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of Cdk1/cyclin B |
Bioorg Med Chem 16: 7128-33 (2008)
Article DOI: 10.1016/j.bmc.2008.06.055
BindingDB Entry DOI: 10.7270/Q2JQ11XQ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Tom's of Maine
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human 5HT7 receptor expressed in CHO cells |
J Nat Prod 66: 535-7 (2003)
Article DOI: 10.1021/np0205102
BindingDB Entry DOI: 10.7270/Q2CZ36WZ |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.25E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Recerca Biomèdica de Barcelona
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 (unknown origin) |
Bioorg Med Chem 16: 7516-24 (2008)
Article DOI: 10.1016/j.bmc.2008.04.067
BindingDB Entry DOI: 10.7270/Q2ZC82NG |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50242173

((2S,3S,4S,5R,6S)-6-(5,6-dihydroxy-4-oxo-2-phenyl-4...)Show SMILES O[C@H]1[C@H](Oc2cc3oc(cc(=O)c3c(O)c2O)-c2ccccc2)O[C@@H]([C@@H](O)[C@@H]1O)C(O)=O |r| Show InChI InChI=1S/C21H18O11/c22-9-6-10(8-4-2-1-3-5-8)30-11-7-12(14(23)15(24)13(9)11)31-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-7,16-19,21,23-27H,(H,28,29)/t16-,17-,18+,19-,21+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Ehime University
Curated by ChEMBL
| Assay Description
Inhibition of trypsin-induced elevation in PAI1 production in HUVEC by ELISA |
J Nat Prod 60: 598-601 (1997)
Article DOI: 10.1021/np970035l
BindingDB Entry DOI: 10.7270/Q20C4ZK4 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data