 Found 20 hits of ic50 for monomerid = 50347388
Found 20 hits of ic50 for monomerid = 50347388 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM50347388
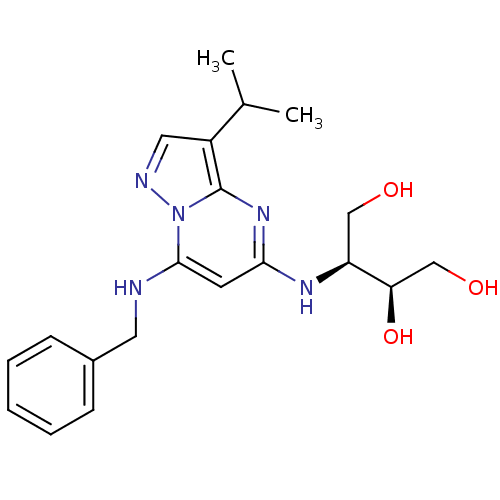
(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CDK2 expressed in baculovirus infected Sf9 cells using Histone H1 as substrate incubated for 10 mins by scintillation counting me... |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113389
BindingDB Entry DOI: 10.7270/Q26W9FVF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Competitive reversible inhibition of CDK2 (unknown origin) |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.126637
BindingDB Entry DOI: 10.7270/Q23F4T3T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/cyclinE assessed as amount of ATP released by luciferase activity based PKLight assay |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 5
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Competitive reversible inhibition of CDK5 (unknown origin) |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.126637
BindingDB Entry DOI: 10.7270/Q23F4T3T |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/cyclinB1 assessed as amount of ATP released by luciferase activity based PKLight assay |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Cyclin-T1/Cyclin-dependent kinase 9
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CDK9/cyclinT assessed as amount of ATP released by luciferase activity based PKLight assay |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 8
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Competitive reversible inhibition of CDK8 (unknown origin) |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.126637
BindingDB Entry DOI: 10.7270/Q23F4T3T |
More data for this
Ligand-Target Pair | |
Cyclin-H/Cyclin-dependent kinase 7
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CDK7/cyclinH/MAT1 assessed as amount of ATP released by luciferase activity based PKLight assay |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 7
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Competitive reversible inhibition of CDK7 (unknown origin) |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.126637
BindingDB Entry DOI: 10.7270/Q23F4T3T |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 15
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of ERK8 |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform alpha
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CK1 |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Insulin receptor-related protein
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of IRR |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Dual specificity tyrosine-phosphorylation-regulated kinase 1A
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of DYRK1A |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Calcium/calmodulin-dependent protein kinase kinase 2
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CAMKKbeta |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 1
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.11E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of ERK2 |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 3
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.73E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of ERK1 |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 4
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Competitive reversible inhibition of CDK4 (unknown origin) |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.126637
BindingDB Entry DOI: 10.7270/Q23F4T3T |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 4/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CDK4/cyclinD1 assessed as amount of ATP released by luciferase activity based PKLight assay |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 6
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.55E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Competitive reversible inhibition of CDK6 (unknown origin) |
Bioorg Med Chem Lett 29: (2019)
Article DOI: 10.1016/j.bmcl.2019.126637
BindingDB Entry DOI: 10.7270/Q23F4T3T |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 6/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM50347388

(CHEMBL1234833)Show SMILES CC(C)c1cnn2c(NCc3ccccc3)cc(N[C@@H](CO)[C@H](O)CO)nc12 |r| Show InChI InChI=1S/C20H27N5O3/c1-13(2)15-10-22-25-19(21-9-14-6-4-3-5-7-14)8-18(24-20(15)25)23-16(11-26)17(28)12-27/h3-8,10,13,16-17,21,26-28H,9,11-12H2,1-2H3,(H,23,24)/t16-,17+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.55E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Imperial College London
Curated by ChEMBL
| Assay Description
Inhibition of CDK6/cyclinD1 assessed as amount of ATP released by luciferase activity based PKLight assay |
J Med Chem 53: 8508-22 (2010)
Article DOI: 10.1021/jm100732t
BindingDB Entry DOI: 10.7270/Q2B858GZ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data