

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50442881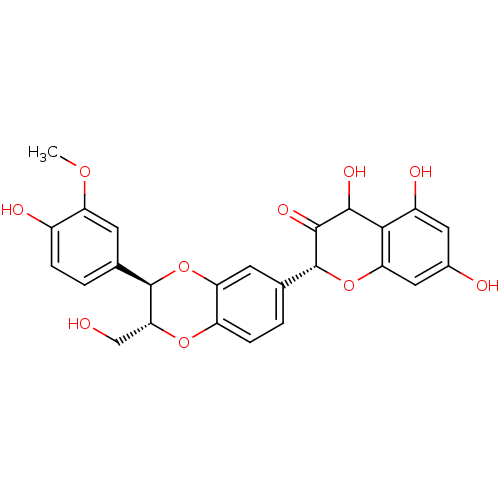 (SILYBIN A) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of monophenolase activity of mushroom tyrosinase assessed as reduction in dopachrome formation using L-Tyrosine substrate by UV-Vis spectr... | Bioorg Med Chem 27: 2499-2507 (2019) Article DOI: 10.1016/j.bmc.2019.03.013 BindingDB Entry DOI: 10.7270/Q2PV6PTR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier organic anion transporter family member 1B3 (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill Curated by ChEMBL | Assay Description Inhibition of OATP1B3 (unknown origin) expressed in HEK293 cells assessed as reduction of [3H]estradiol-17beta-glucuronide uptake after 3 mins by bet... | Drug Metab Dispos 41: 958-65 (2013) Article DOI: 10.1124/dmd.112.048272 BindingDB Entry DOI: 10.7270/Q2PK0HWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier organic anion transporter family member 1B3 (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
KU Leuven Curated by ChEMBL | Assay Description pIC50 values for sodium fluorescein (10 uM) uptake in OATP1B3-transfected CHO cells | Mol Pharmacol 83: 1257-67 (2013) Article DOI: 10.1124/mol.112.084152 BindingDB Entry DOI: 10.7270/Q2TB18BP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier organic anion transporter family member 2B1 (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill Curated by ChEMBL | Assay Description Inhibition of OATP2B1 (unknown origin) expressed in MDCK2 cells assessed as reduction of [3H]estrone-3-sulfate uptake after 3 mins by beta-counting | Drug Metab Dispos 41: 958-65 (2013) Article DOI: 10.1124/dmd.112.048272 BindingDB Entry DOI: 10.7270/Q2PK0HWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier organic anion transporter family member 1B1 (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
KU Leuven Curated by ChEMBL | Assay Description pIC50 values for sodium fluorescein (10 uM) uptake in OATP1B1-transfected CHO cells | Mol Pharmacol 83: 1257-67 (2013) Article DOI: 10.1124/mol.112.084152 BindingDB Entry DOI: 10.7270/Q2TB18BP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier organic anion transporter family member 1B1 (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill Curated by ChEMBL | Assay Description Inhibition of OATP1B1 (unknown origin) expressed in HEK293 cells assessed as reduction of [3H]estradiol-17beta-glucuronide uptake after 3 mins by bet... | Drug Metab Dispos 41: 958-65 (2013) Article DOI: 10.1124/dmd.112.048272 BindingDB Entry DOI: 10.7270/Q2PK0HWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50442881 (SILYBIN A) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.21E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of diphenolase activity of mushroom tyrosinase assessed as reduction in dopachrome formation using L-DOPA substrate by UV-Vis spectrophoto... | Bioorg Med Chem 27: 2499-2507 (2019) Article DOI: 10.1016/j.bmc.2019.03.013 BindingDB Entry DOI: 10.7270/Q2PV6PTR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2C9 (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tasly Pharmaceutical Group Co., Ltd. Curated by ChEMBL | Assay Description Inhibition of CYP2C9 in human liver microsomes using (S)-warfarin as substrate at after 30 mins by LC-MS/MS analysis | Eur J Med Chem 123: 577-595 (2016) Article DOI: 10.1016/j.ejmech.2016.07.070 BindingDB Entry DOI: 10.7270/Q2736SW8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.09E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Lodz Curated by ChEMBL | Assay Description Inhibition of thrombin (unknown origin) assessed as hydrolysis of N-benzoyl-phenylalanylvalyl-arginine-paranitroanilide | Med Chem Res 23: 2324-2337 (2014) Article DOI: 10.1007/s00044-013-0829-4 BindingDB Entry DOI: 10.7270/Q2377B7W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urokinase-type plasminogen activator (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Lodz Curated by ChEMBL | Assay Description Inhibition of urokinase amidolytic activity (unknown origin) | Med Chem Res 23: 2324-2337 (2014) Article DOI: 10.1007/s00044-013-0829-4 BindingDB Entry DOI: 10.7270/Q2377B7W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50442881 (SILYBIN A) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Lodz Curated by ChEMBL | Assay Description Inhibition of human thrombin amidolytic activity using D-Phe-Pip-Arg-pNA as substrate preincubated for 10 mins followed by substrate addition measure... | Med Chem Res 23: 2324-2337 (2014) Article DOI: 10.1007/s00044-013-0829-4 BindingDB Entry DOI: 10.7270/Q2377B7W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||