

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DNA gyrase subunit A/B (Staphylococcus aureus) | BDBM50393079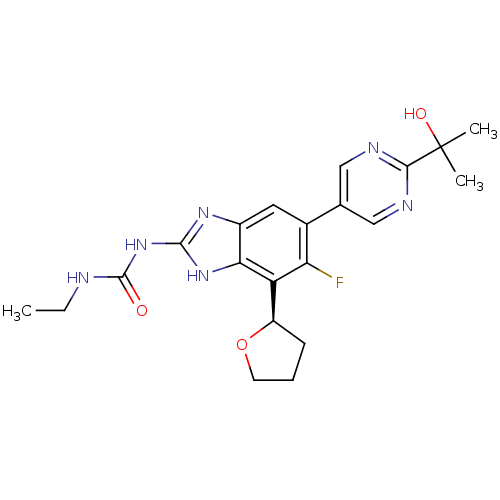 (CHEMBL2152855 | US9040542, 23) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc Curated by ChEMBL | Assay Description Inhibition of Staphylococcus aureus DNA gyrase using pBR322 plasmid DNA as substrate by coupled enzyme reaction assay | J Med Chem 57: 8792-816 (2014) Article DOI: 10.1021/jm500563g BindingDB Entry DOI: 10.7270/Q2TF01BF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA gyrase subunit B (Staphylococcus aureus) | BDBM50393079 (CHEMBL2152855 | US9040542, 23) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of Staphylococcus aureus DNA gyrase Gyr B subunit by ATPase assay | ACS Med Chem Lett 3: 783-784 (2012) Article DOI: 10.1021/ml300234y BindingDB Entry DOI: 10.7270/Q2N29Z1N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA gyrase subunit A/B (Staphylococcus aureus) | BDBM50393079 (CHEMBL2152855 | US9040542, 23) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | US Patent | 9 | n/a | n/a | n/a | n/a | n/a | n/a | 7.6 | n/a |
VERTEX PHARMACEUTICALS INCORPORATED US Patent | Assay Description The ATP hydrolysis activity of S. aureus DNA gyrase is measured by coupling the production of ADP through pyruvate kinase/lactate dehydrogenase to th... | US Patent US9040542 (2015) BindingDB Entry DOI: 10.7270/Q2F18XHX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA topoisomerase 4 subunit B (Staphylococcus aureus) | BDBM50393079 (CHEMBL2152855 | US9040542, 23) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of Staphylococcus aureus DNA topoisomerase 4 Par E subunit by ATPase assay | ACS Med Chem Lett 3: 783-784 (2012) Article DOI: 10.1021/ml300234y BindingDB Entry DOI: 10.7270/Q2N29Z1N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA topoisomerase 4 subunit A/B (Staphylococcus aureus) | BDBM50393079 (CHEMBL2152855 | US9040542, 23) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | US Patent | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
VERTEX PHARMACEUTICALS INCORPORATED US Patent | Assay Description The conversion of ATP to ADP by S. aureus TopoIV enzyme is coupled to the conversion of NADH to NAD+, and the progress of the reaction is measured by... | US Patent US9040542 (2015) BindingDB Entry DOI: 10.7270/Q2F18XHX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA gyrase subunit A/B (Staphylococcus aureus) | BDBM50393079 (CHEMBL2152855 | US9040542, 23) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | <21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc Curated by ChEMBL | Assay Description Inhibition of Staphylococcus aureus DNA gyrase | ACS Med Chem Lett 6: 822-6 (2015) Article DOI: 10.1021/acsmedchemlett.5b00196 BindingDB Entry DOI: 10.7270/Q2J67JPW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| DNA topoisomerase 4 subunit A/B (Staphylococcus aureus) | BDBM50393079 (CHEMBL2152855 | US9040542, 23) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc Curated by ChEMBL | Assay Description Inhibition of Staphylococcus aureus DNA topoisomerase 4 | ACS Med Chem Lett 6: 822-6 (2015) Article DOI: 10.1021/acsmedchemlett.5b00196 BindingDB Entry DOI: 10.7270/Q2J67JPW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||