

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50262341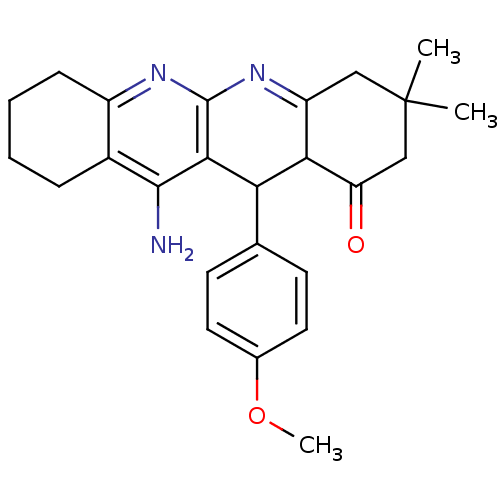 (11-Amino-12-(4-methoxy-phenyl)-3,3-dimethyl-3,4,5,...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 41.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CSIC) Curated by ChEMBL | Assay Description Inhibition of Electrophorus electricus AChE by Ellman's method | Bioorg Med Chem 16: 7759-69 (2008) Article DOI: 10.1016/j.bmc.2008.07.005 BindingDB Entry DOI: 10.7270/Q2DZ096D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50262341 (11-Amino-12-(4-methoxy-phenyl)-3,3-dimethyl-3,4,5,...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIC) Curated by ChEMBL | Assay Description Inhibition of Electrophorus electricus AChE by spectrophotometry | Bioorg Med Chem 16: 7759-69 (2008) Article DOI: 10.1016/j.bmc.2008.07.005 BindingDB Entry DOI: 10.7270/Q2DZ096D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50262341 (11-Amino-12-(4-methoxy-phenyl)-3,3-dimethyl-3,4,5,...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIC) Curated by ChEMBL | Assay Description Inhibition of human erythrocyte AChE by Ellman's method | Bioorg Med Chem 16: 7759-69 (2008) Article DOI: 10.1016/j.bmc.2008.07.005 BindingDB Entry DOI: 10.7270/Q2DZ096D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||