

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiotensin-converting enzyme (Homo sapiens (Human)) | BDBM50038066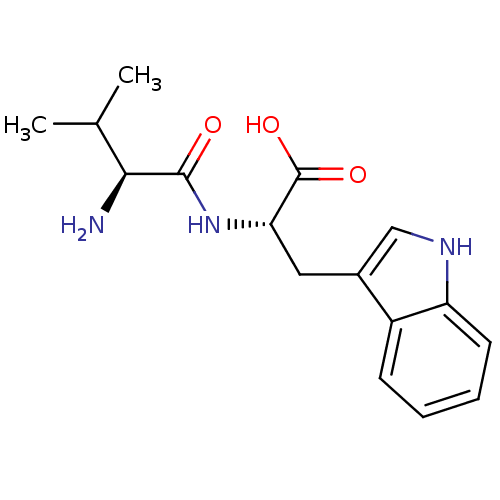 ((S)-2-((S)-2-Amino-3-methyl-butyrylamino)-3-(1H-in...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.58E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Biological activity was measured against Angiotensin I converting enzyme | J Med Chem 38: 2705-13 (1995) BindingDB Entry DOI: 10.7270/Q2GX4CSN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Homo sapiens (Human)) | BDBM50038066 ((S)-2-((S)-2-Amino-3-methyl-butyrylamino)-3-(1H-in...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Tanabe Seiyaku Co., Ltd. Curated by ChEMBL | Assay Description Inhibitory activity on Angiotensin I converting enzyme (ACE) obtained from pig renal cortex and hippuryl-histidyl-leucine as substrate | J Med Chem 32: 289-97 (1989) BindingDB Entry DOI: 10.7270/Q2XP75JS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Homo sapiens (Human)) | BDBM50038066 ((S)-2-((S)-2-Amino-3-methyl-butyrylamino)-3-(1H-in...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Proteus Molecular Design Ltd. Curated by ChEMBL | Assay Description Inhibition against ACE. | J Med Chem 37: 3994-4002 (1994) BindingDB Entry DOI: 10.7270/Q2XW4HWB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Homo sapiens (Human)) | BDBM50038066 ((S)-2-((S)-2-Amino-3-methyl-butyrylamino)-3-(1H-in...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.58E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Tianjin University Curated by ChEMBL | Assay Description Inhibition of ACE (unknown origin) | Eur J Med Chem 84: 100-6 (2014) Article DOI: 10.1016/j.ejmech.2014.07.015 BindingDB Entry DOI: 10.7270/Q29025F2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||