

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM102944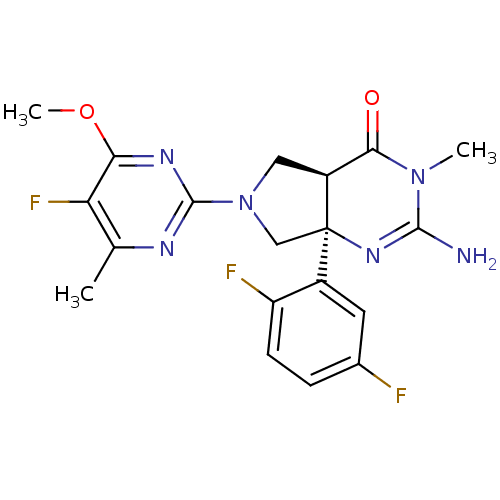 (US8541427, 15 | US8541427, 206 | US8541427, 44 | U...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | 6.10 | -47.7 | 25 | n/a | n/a | n/a | n/a | n/a | 30 |
Merck, Sharp & Dohme, Corp. US Patent | Assay Description A homogeneous time-resolved FRET assay was used to determine IC50 values for inhibitors of the soluble human BACE1 catalytic domain. The assay monit... | US Patent US8541427 (2013) BindingDB Entry DOI: 10.7270/Q26D5RN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM102944 (US8541427, 15 | US8541427, 206 | US8541427, 44 | U...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | 7.30 | -47.2 | 23 | n/a | n/a | n/a | n/a | n/a | 30 |
Merck, Sharp & Dohme, Corp. US Patent | Assay Description A homogeneous time-resolved FRET assay was used to determine IC50 values for inhibitors of the soluble human BACE1 catalytic domain. The assay monit... | US Patent US8541427 (2013) BindingDB Entry DOI: 10.7270/Q26D5RN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM102944 (US8541427, 15 | US8541427, 206 | US8541427, 44 | U...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | 8.5 | -46.8 | 28 | n/a | n/a | n/a | n/a | n/a | 30 |
Merck, Sharp & Dohme, Corp. US Patent | Assay Description A homogeneous time-resolved FRET assay was used to determine IC50 values for inhibitors of the soluble human BACE1 catalytic domain. The assay monit... | US Patent US8541427 (2013) BindingDB Entry DOI: 10.7270/Q26D5RN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM102944 (US8541427, 15 | US8541427, 206 | US8541427, 44 | U...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 8.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research and Development Curated by ChEMBL | Assay Description Inhibition of soluble human BACE1 (1 to 454) expressed in baculovirus infected insect Sf9 cells using EuK-KTEEISEVNLDAEFRHDKC-biotin as substrate pre... | Bioorg Med Chem Lett 24: 2033-45 (2014) Article DOI: 10.1016/j.bmcl.2014.03.025 BindingDB Entry DOI: 10.7270/Q2H41T0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM102944 (US8541427, 15 | US8541427, 206 | US8541427, 44 | U...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of recombinant human BACE1 preincubated for 30 mins followed QSY7EISEVNLDAEFC-Eu-amide substrate addition measured after 90 mins by FRET a... | J Med Chem 59: 3231-48 (2016) Article DOI: 10.1021/acs.jmedchem.5b01995 BindingDB Entry DOI: 10.7270/Q2CR5W8V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM102944 (US8541427, 15 | US8541427, 206 | US8541427, 44 | U...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of BACE1 in human HEK293 cells transfected with human APP Swe/Lon mutations assessed as amyloid beta 40 level after 4 hrs by electrochemil... | J Med Chem 59: 3231-48 (2016) Article DOI: 10.1021/acs.jmedchem.5b01995 BindingDB Entry DOI: 10.7270/Q2CR5W8V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||