

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250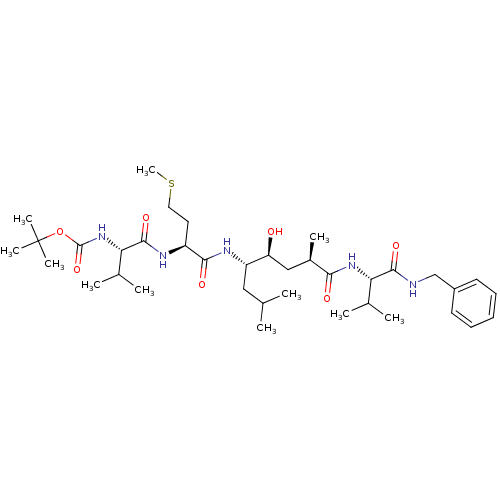 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.5 | -51.1 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
University of Illinois at Chicago | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Med Chem 44: 2865-8 (2001) Article DOI: 10.1021/jm0101803 BindingDB Entry DOI: 10.7270/Q2BG2M7W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.5 | -51.1 | n/a | n/a | n/a | n/a | n/a | 4.5 | 37 |
Purdue University | Assay Description Enzyme activities were assayed by monitoring the hydrolysis of substrate in the presence or absence of inhibitor compounds. The hydrolysis was record... | J Am Chem Soc 128: 5310-1 (2006) Article DOI: 10.1021/ja058636j BindingDB Entry DOI: 10.7270/Q26W98BN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description In vitro inhibition of Beta-secretase-1 in HEK293 (Human Embryonic Kidney) cell line. | Bioorg Med Chem Lett 14: 239-43 (2003) BindingDB Entry DOI: 10.7270/Q2MW2HP3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Santiago de Compostela Curated by ChEMBL | Assay Description Inhibition of human recombinant BACE1 ectodomain (1 to 460 amino acids) assessed as inhibition of proteolytic cleavage of Rhodamine-EVNLDAEFK-Quenche... | J Med Chem 54: 3081-5 (2011) Article DOI: 10.1021/jm101568y BindingDB Entry DOI: 10.7270/Q20C4W7T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Elan Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested for its inhibitory activity against human beta-Secretase (BACE) | J Med Chem 46: 4625-30 (2003) Article DOI: 10.1021/jm030247h BindingDB Entry DOI: 10.7270/Q2PR7VCB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16250 (CHEMBL290001 | N-(tert-butoxycarbonyl)-L-valyl-N-[...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | 4.8 | 37 |
Elan Pharmaceuticals | Assay Description Beta-cleavage ELISA assays were carried out in reaction buffer containing enzyme, substrate MBP-C125, and test compounds. Generated beta-cleaved prod... | J Med Chem 47: 158-64 (2004) Article DOI: 10.1021/jm0304008 BindingDB Entry DOI: 10.7270/Q22Z13RC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||