

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50068666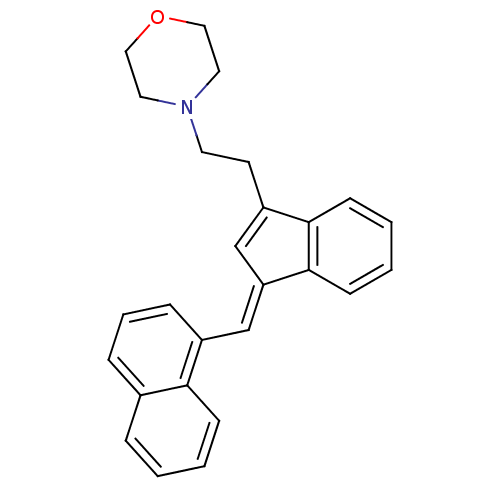 (4-(2-{3-[1-Naphthalen-1-yl-meth-(E)-ylidene]-3H-in...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kennesaw State University Curated by ChEMBL | Assay Description Compound was evaluated for its binding affinity against Cannabinoid receptor 1 in Guinea pig ileum (GPI) using [3H]CP-55940 ligand | J Med Chem 41: 5177-87 (1999) Article DOI: 10.1021/jm9801197 BindingDB Entry DOI: 10.7270/Q2ST7QJJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50068666 (4-(2-{3-[1-Naphthalen-1-yl-meth-(E)-ylidene]-3H-in...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Displacement of [3H]-CP55940 from human CB1 receptor | J Med Chem 59: 7525-43 (2016) Article DOI: 10.1021/acs.jmedchem.6b00516 BindingDB Entry DOI: 10.7270/Q2862KX2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50068666 (4-(2-{3-[1-Naphthalen-1-yl-meth-(E)-ylidene]-3H-in...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kennesaw State University Curated by ChEMBL | Assay Description Compound was evaluated for its binding affinity against Cannabinoid receptor 1 in Guinea pig ileum (GPI) using [3H]SR-141,716A ligand at site 1 | J Med Chem 41: 5177-87 (1999) Article DOI: 10.1021/jm9801197 BindingDB Entry DOI: 10.7270/Q2ST7QJJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50068666 (4-(2-{3-[1-Naphthalen-1-yl-meth-(E)-ylidene]-3H-in...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 611 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kennesaw State University Curated by ChEMBL | Assay Description Compound was evaluated for its binding affinity against Cannabinoid receptor 1 in Guinea pig ileum (GPI) using [3H]SR-141,716A ligand at site 2 | J Med Chem 41: 5177-87 (1999) Article DOI: 10.1021/jm9801197 BindingDB Entry DOI: 10.7270/Q2ST7QJJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50068666 (4-(2-{3-[1-Naphthalen-1-yl-meth-(E)-ylidene]-3H-in...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 257 | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Agonist activity at human CB1R expressed in CHO cells by Ca2+ flux assay | J Med Chem 59: 7525-43 (2016) Article DOI: 10.1021/acs.jmedchem.6b00516 BindingDB Entry DOI: 10.7270/Q2862KX2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||