 Found 11 hits Enz. Inhib. hit(s) with Target = 'Cannabinoid receptor 2' and Ligand = 'BDBM50399157'
Found 11 hits Enz. Inhib. hit(s) with Target = 'Cannabinoid receptor 2' and Ligand = 'BDBM50399157' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157
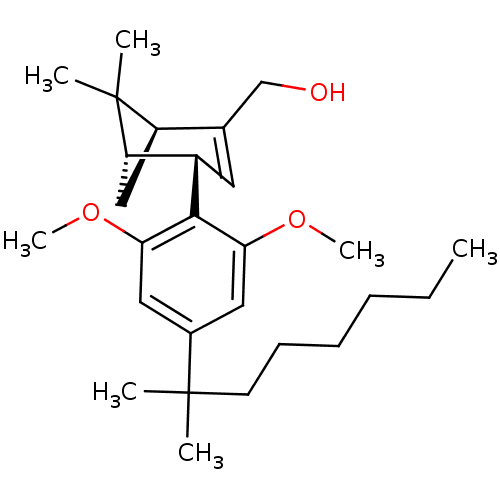
(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]CP55940 from human CB2R transfected in HEK293EBNA cell membrane incubated for 90 mins by Microbeta TriLux based luminescence anal... |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115672
BindingDB Entry DOI: 10.7270/Q2TT4VNQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Sassari
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human CB2 receptor expressed in HEK293 EBNA cell membranes after 90 mins by liquid scintillation and luminescence co... |
Eur J Med Chem 127: 398-412 (2017)
Article DOI: 10.1016/j.ejmech.2017.01.002
BindingDB Entry DOI: 10.7270/Q26W9D9V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Arena Pharmaceuticals
Curated by ChEMBL
| Assay Description
Displacement of [3H]HU-243 from human CB2 receptor |
Cell Chem Biol 56: 8224-56 (2013)
Article DOI: 10.1021/jm4005626
BindingDB Entry DOI: 10.7270/Q2B859M5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Massachusetts Medical School
Curated by ChEMBL
| Assay Description
Displacement of [3H]HU-243 from CB2 receptor (unknown origin) transfected in human COS7 cells |
Bioorg Med Chem 23: 1377-85 (2015)
Article DOI: 10.1016/j.bmc.2015.01.059
BindingDB Entry DOI: 10.7270/Q2Z60QRX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
In vitro antagonism of the 5-HT-3 receptor determined by inhibition of 5-HT-induced depolarization of the isolated rat vagus nerve. |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 44 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113878
BindingDB Entry DOI: 10.7270/Q2PK0M3G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 76 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113878
BindingDB Entry DOI: 10.7270/Q2PK0M3G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 85 | n/a | n/a | n/a | n/a |
University of Pittsburgh
Curated by ChEMBL
| Assay Description
Agonist activity at human CB2 receptor expressed in CHO cell membranes assessed as increase in forskolin-stimulated cAMP production after 45 mins by ... |
ACS Med Chem Lett 4: 387-392 (2013)
Article DOI: 10.1021/ml3004236
BindingDB Entry DOI: 10.7270/Q2MP54MQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 84 | n/a | n/a | n/a | n/a |
University of Pittsburgh
Curated by ChEMBL
| Assay Description
Inverse agonist activity at human CB2 receptor expressed in CHO cells assessed as increase in forskolin-induced cAMP production after 45 mins in pres... |
J Med Chem 56: 2045-58 (2013)
Article DOI: 10.1021/jm3017464
BindingDB Entry DOI: 10.7270/Q2BK1DPW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 150 | n/a | n/a | n/a | n/a |
University of Oxford
Curated by ChEMBL
| Assay Description
Agonist activity at human CB2R expressed in CHO cells assessed as reduction in forskolin-induced cAMP accumulation by beta-galactosidase based comple... |
Bioorg Med Chem 23: 241-63 (2014)
Article DOI: 10.1016/j.bmc.2014.11.002
BindingDB Entry DOI: 10.7270/Q2930VS3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50399157

(CHEMBL2179724)Show SMILES CCCCCCC(C)(C)c1cc(OC)c([C@H]2C=C(CO)[C@H]3C[C@@H]2C3(C)C)c(OC)c1 |r,t:16| Show InChI InChI=1S/C27H42O3/c1-8-9-10-11-12-26(2,3)19-14-23(29-6)25(24(15-19)30-7)20-13-18(17-28)21-16-22(20)27(21,4)5/h13-15,20-22,28H,8-12,16-17H2,1-7H3/t20-,21+,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 83.8 | n/a | n/a | n/a | n/a |
University of Pittsburgh
Curated by ChEMBL
| Assay Description
Agonist activity at human CB2 receptor expressed in CHO cells assessed as reduction in forskolin-induced cAMP accumulation incubated for 45 mins by T... |
J Med Chem 55: 9973-87 (2012)
Article DOI: 10.1021/jm301212u
BindingDB Entry DOI: 10.7270/Q22808R5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data