

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50387127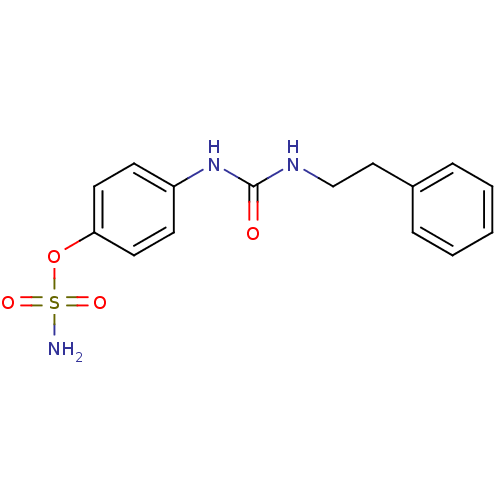 (4-ureidophenyl sulfamate ring derivative 3l | CHEM...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Romanian Academy Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase-9 | Bioorg Med Chem 21: 1404-9 (2013) Article DOI: 10.1016/j.bmc.2012.11.004 BindingDB Entry DOI: 10.7270/Q2XW4M4Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50387127 (4-ureidophenyl sulfamate ring derivative 3l | CHEM...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Manchester Curated by ChEMBL | Assay Description Inhibition of human recombinant transmembrane CA9 preincubated for 15 mins by stopped-flow CO2 hydration method | J Med Chem 55: 5591-600 (2012) Article DOI: 10.1021/jm300529u BindingDB Entry DOI: 10.7270/Q23R0TZM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50387127 (4-ureidophenyl sulfamate ring derivative 3l | CHEM...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 9 preincubated for 15 mins measured for 10 to 100 sec using phenol red indicator-based stopped flow assay | Bioorg Med Chem Lett 22: 4681-5 (2012) Article DOI: 10.1016/j.bmcl.2012.05.083 BindingDB Entry DOI: 10.7270/Q20866C2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50387127 (4-ureidophenyl sulfamate ring derivative 3l | CHEM...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Birla Institute of Technology | J Enzyme Inhib Med Chem 29: 571-81 (2014) Article DOI: 10.3109/14756366.2013.827677 BindingDB Entry DOI: 10.7270/Q2NC5ZCQ | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||