

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cholinesterase (Homo sapiens (Human)) | BDBM8970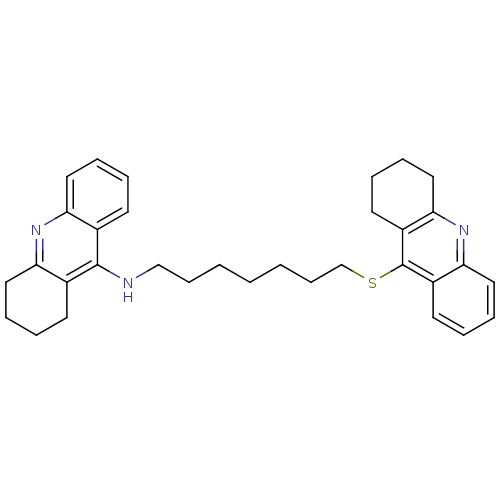 (CHEMBL51197 | N-[7-(1,2,3,4-tetrahydroacridin-9-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena Curated by ChEMBL | Assay Description Inhibition of Equine Butyrylcholinesterase | J Med Chem 46: 1-4 (2002) Article DOI: 10.1021/jm0255668 BindingDB Entry DOI: 10.7270/Q2GF0V7X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM8970 (CHEMBL51197 | N-[7-(1,2,3,4-tetrahydroacridin-9-yl...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate | Eur J Med Chem 132: 294-309 (2017) Article DOI: 10.1016/j.ejmech.2017.03.062 BindingDB Entry DOI: 10.7270/Q2HX1G33 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||