

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50229191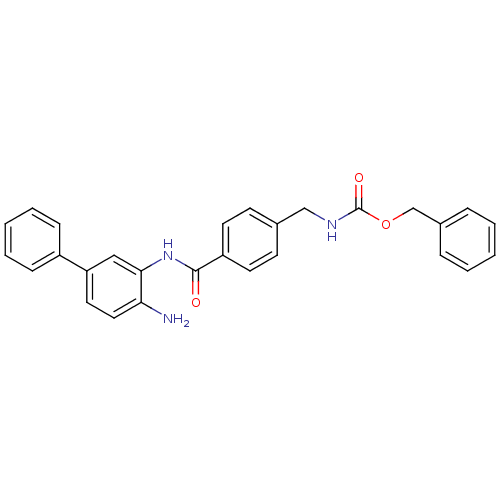 (CHEMBL252409 | US9096559, 15 | [4-(4-amino-bipheny...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Inhibition of HDAC1 | Bioorg Med Chem Lett 18: 726-31 (2008) Article DOI: 10.1016/j.bmcl.2007.11.047 BindingDB Entry DOI: 10.7270/Q28W3D1M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50229191 (CHEMBL252409 | US9096559, 15 | [4-(4-amino-bipheny...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 58 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp. US Patent | Assay Description Novel compounds were tested for their ability to inhibit histone deacetylase, subtype 1 (HDAC1) using an in vitro deacetylation assay. The enzyme sou... | US Patent US9096559 (2015) BindingDB Entry DOI: 10.7270/Q2K07317 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50229191 (CHEMBL252409 | US9096559, 15 | [4-(4-amino-bipheny...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.88E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Inhibition of human recombinant HDAC1 using Boc-L-Lys (Ac)-AMC as substrate preincubated with compound for 3 hrs measured after 35 mins by spectroflu... | Bioorg Med Chem Lett 22: 5025-30 (2012) Article DOI: 10.1016/j.bmcl.2012.06.017 BindingDB Entry DOI: 10.7270/Q2F76DNT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone deacetylase 1 (Homo sapiens (Human)) | BDBM50229191 (CHEMBL252409 | US9096559, 15 | [4-(4-amino-bipheny...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.45E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Inhibition of human recombinant HDAC1 using Boc-L-Lys (Ac)-AMC as substrate preincubated with compound for 5 mins measured after 35 mins by spectrofl... | Bioorg Med Chem Lett 22: 5025-30 (2012) Article DOI: 10.1016/j.bmcl.2012.06.017 BindingDB Entry DOI: 10.7270/Q2F76DNT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||