

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM31772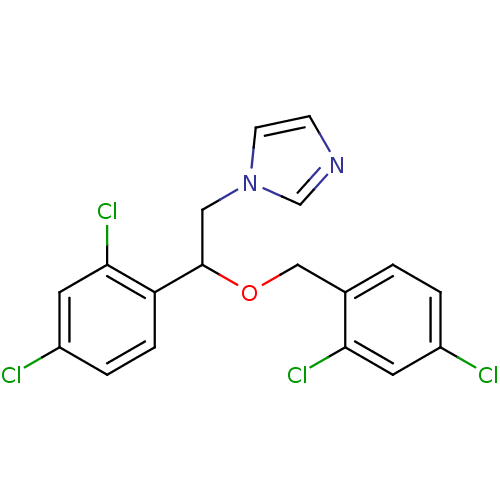 (1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant IDO1 using L-tryptophan as substrate after 30 mins by fluorimetric analysis | Bioorg Med Chem 26: 1665-1674 (2018) Article DOI: 10.1016/j.bmc.2018.02.014 BindingDB Entry DOI: 10.7270/Q2M61NVQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM31772 (1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal IDO1 expressed in Escherichia coli BL21 A1 cells using L-tryptophan as substrate measured after 30 mins by HPLC analys... | Citation and Details Article DOI: 10.1016/j.bmcl.2020.127756 BindingDB Entry DOI: 10.7270/Q2CF9TS5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM31772 (1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Swiss Institute of Bioinformatics Curated by ChEMBL | Assay Description Inhibition of IDO1 (unknown origin) using L-tryptophan substrate incubated for 60 mins in presence of 0.01% Triton-X by HPLC | Eur J Med Chem 84: 284-301 (2014) Article DOI: 10.1016/j.ejmech.2014.06.078 BindingDB Entry DOI: 10.7270/Q21C1ZJV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Homo sapiens (Human)) | BDBM31772 (1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Swiss Institute of Bioinformatics Curated by ChEMBL | Assay Description Inhibition of IDO1 (unknown origin) using L-tryptophan substrate incubated for 60 mins by HPLC | Eur J Med Chem 84: 284-301 (2014) Article DOI: 10.1016/j.ejmech.2014.06.078 BindingDB Entry DOI: 10.7270/Q21C1ZJV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Indoleamine 2,3-dioxygenase 1 (Mus musculus) | BDBM31772 (1-[2-(2,4-dichlorobenzyl)oxy-2-(2,4-dichlorophenyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.78E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Sydney Curated by ChEMBL | Assay Description Inhibition of mouse Ido1 transfected in HEK293T cells using L-tryptophan as substrate assessed as kynurenine formation after 45 mins by spectrophotom... | Bioorg Med Chem Lett 22: 7641-6 (2012) Article DOI: 10.1016/j.bmcl.2012.10.010 BindingDB Entry DOI: 10.7270/Q20G3M92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||