

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial (Homo sapiens (Human)) | BDBM50386310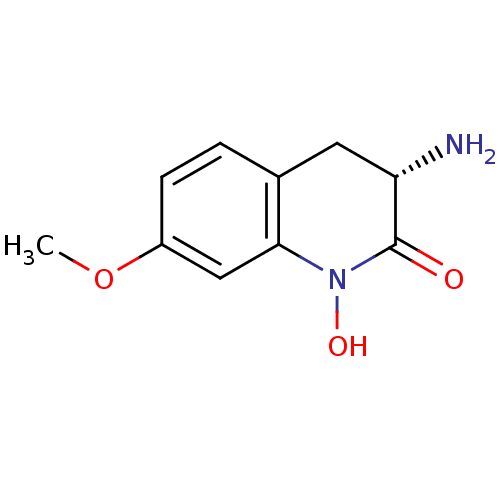 (CHEMBL2049092) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Irreversible inhibition of human KAT2 using L-kynurenine as substrate measured every 5 mins over 16 hrs by SpectraMax plate reader analysis | ACS Med Chem Lett 4: 37-40 (2013) Article DOI: 10.1021/ml300237v BindingDB Entry DOI: 10.7270/Q2KH0PNV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial (Homo sapiens (Human)) | BDBM50386310 (CHEMBL2049092) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of human recombinant KAT2 assessed as conversion of L-kynurenine into kynurenic acid after 15 to 20 hrs | ACS Med Chem Lett 3: 187-192 (2012) Article DOI: 10.1021/ml200204m BindingDB Entry DOI: 10.7270/Q2MC9135 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial (Homo sapiens (Human)) | BDBM50386310 (CHEMBL2049092) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of human KAT2 using L-kynurenine as substrate measured after 15-20 hrs by SpectraMax plate reader analysis | ACS Med Chem Lett 4: 37-40 (2013) Article DOI: 10.1021/ml300237v BindingDB Entry DOI: 10.7270/Q2KH0PNV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial (Rattus norvegicus) | BDBM50386310 (CHEMBL2049092) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 137 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of rat recombinant KAT2 assessed as conversion of L-kynurenine into kynurenic acid after 15 to 20 hrs | ACS Med Chem Lett 3: 187-192 (2012) Article DOI: 10.1021/ml200204m BindingDB Entry DOI: 10.7270/Q2MC9135 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||