

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P2Y purinoceptor 1 (Meleagris gallopavo) | BDBM50085829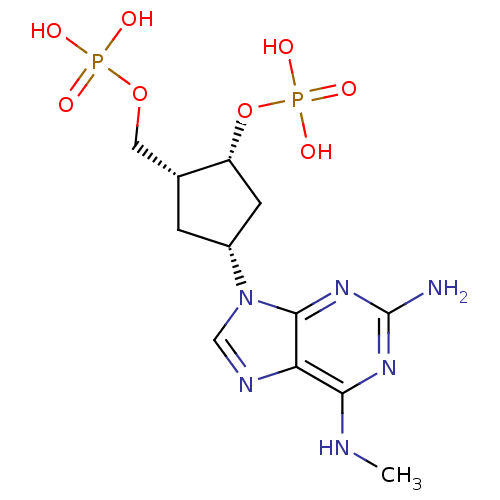 (CHEMBL355406 | Phosphoric acid mono-[4-(2-amino-6-...) | MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.42E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Inhibition of 30 nM 2-MeSADP stimulation of 2PY1-mediated phospholipase C (PLC) activity in Turkey erythocyte ghosts | J Med Chem 43: 829-42 (2000) BindingDB Entry DOI: 10.7270/Q2474BJ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| P2Y purinoceptor 1 (Meleagris gallopavo) | BDBM50085829 (CHEMBL355406 | Phosphoric acid mono-[4-(2-amino-6-...) | MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 6.51E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description In vitro stimulation of 2PY1 purinoceptor mediated phospholipase C (PLC) activity in Turkey Erythrocyte Ghosts | J Med Chem 43: 829-42 (2000) BindingDB Entry DOI: 10.7270/Q2474BJ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||