

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Proteinase-activated receptor 1 (Homo sapiens (Human)) | BDBM230672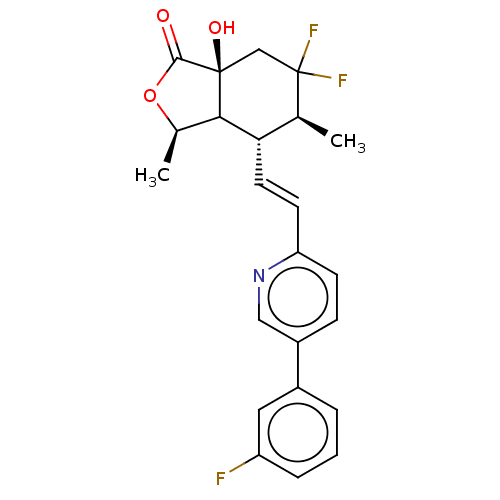 (US9340530, 10) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Merck Sharp & Dohme Corp. US Patent | Assay Description Thrombin receptor antagonists were screened using a modification of the thrombin receptor radioligand binding assay of Ahn et al. (Ahn et al., Mol. P... | US Patent US9340530 (2016) BindingDB Entry DOI: 10.7270/Q2K35SJH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proteinase-activated receptor 1 (Homo sapiens (Human)) | BDBM230672 (US9340530, 10) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 8.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp. US Patent | Assay Description HEK 293 Cells were grown in media containing DMEM, 10% FBS pen/strep/L-Glutamine and non-essential amino acids. The cells were plated in 384-well PDL... | US Patent US9340530 (2016) BindingDB Entry DOI: 10.7270/Q2K35SJH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||