

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50339081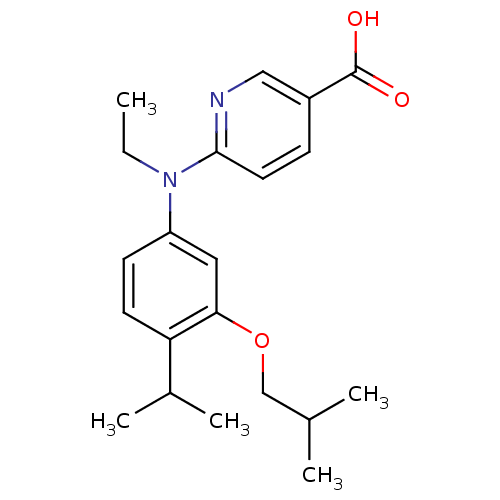 (6-[Ethyl-(3-isobutoxy-4-isopropylphenyl)amino]nico...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 182 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Okayama University Graduate School of Medicine Curated by ChEMBL | Assay Description Displacement of CU-6PMN from human RXRalpha LBD incubated for 2 hrs by fluorescence based assay | J Med Chem 62: 8809-8818 (2019) Article DOI: 10.1021/acs.jmedchem.9b00995 BindingDB Entry DOI: 10.7270/Q2G44TRN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50339081 (6-[Ethyl-(3-isobutoxy-4-isopropylphenyl)amino]nico...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 182 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Okayama University Graduate School of Medicine Curated by ChEMBL | Assay Description Displacement of CU-6PMN from human RXRalpha LBD incubated for 2 hrs by fluorescence based assay | J Med Chem 62: 8809-8818 (2019) Article DOI: 10.1021/acs.jmedchem.9b00995 BindingDB Entry DOI: 10.7270/Q2G44TRN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50339081 (6-[Ethyl-(3-isobutoxy-4-isopropylphenyl)amino]nico...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 19 | n/a | n/a | n/a | n/a |
Okayama University Graduate School of Medicine Curated by ChEMBL | Assay Description Agonist activity at RXRalpha (unknown origin) expressed in COS1 cells incubated for 18 hrs by luciferase reporter gene assay | ACS Med Chem Lett 6: 334-8 (2015) Article DOI: 10.1021/ml500511m BindingDB Entry DOI: 10.7270/Q25T3N7K | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Retinoic acid receptor RXR-alpha (Homo sapiens (Human)) | BDBM50339081 (6-[Ethyl-(3-isobutoxy-4-isopropylphenyl)amino]nico...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | n/a | n/a | 19 | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Agonist activity at RXRalpha transfected in human COS1 cells after 18 hrs by luciferase reporter gene transactivation assay | ACS Med Chem Lett 1: 521-525 (2010) Article DOI: 10.1021/ml100184k BindingDB Entry DOI: 10.7270/Q28S4Q6X | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||