

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sialidase (Clostridium perfringens) | BDBM50317437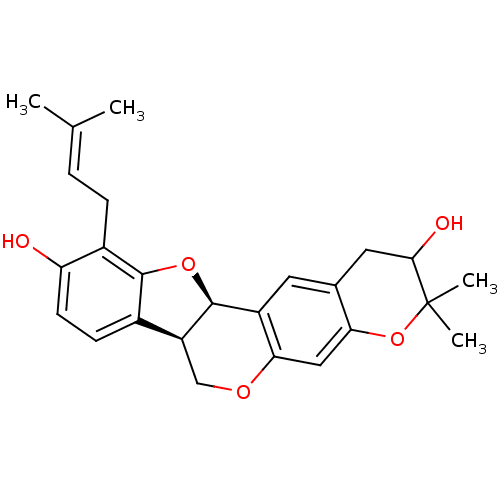 (CHEMBL1096408 | Erythribyssin L) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Chosun University Curated by ChEMBL | Assay Description Noncompetitive inhibition of Clostridium perfringens neuraminidase | Bioorg Med Chem 18: 3335-44 (2010) Article DOI: 10.1016/j.bmc.2010.03.005 BindingDB Entry DOI: 10.7270/Q2MW2HBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50317437 (CHEMBL1096408 | Erythribyssin L) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.79E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chosun University Curated by ChEMBL | Assay Description Inhibition of Clostridium perfringens neuraminidase assessed as inhibition of 4-MU-NANA hydrolysis after 10 mins by spectrofluorometry | Bioorg Med Chem 18: 3335-44 (2010) Article DOI: 10.1016/j.bmc.2010.03.005 BindingDB Entry DOI: 10.7270/Q2MW2HBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Vibrio cholerae) | BDBM50317437 (CHEMBL1096408 | Erythribyssin L) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.71E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Chosun University Curated by ChEMBL | Assay Description Inhibition of Vibrio cholerae neuraminidase assessed as inhibition of 4-MU-NANA hydrolysis after 30 mins by spectrofluorometry | Bioorg Med Chem 18: 3335-44 (2010) Article DOI: 10.1016/j.bmc.2010.03.005 BindingDB Entry DOI: 10.7270/Q2MW2HBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||