

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM50342885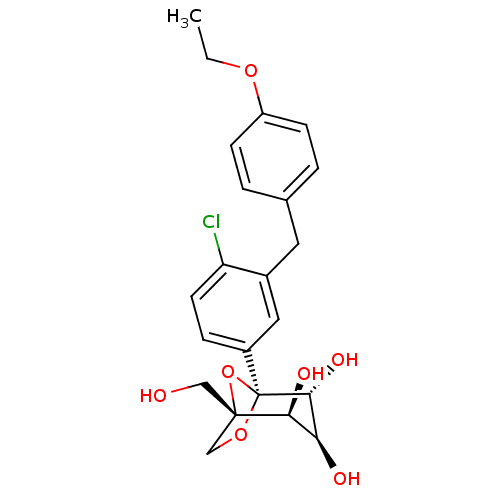 ((1S,2S,3S,4R,5S)-5-[4-Chloro-3-(4-ethoxybenzyl)phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.877 | n/a | n/a | n/a | n/a | n/a | n/a |
Jamia Hamdard Curated by ChEMBL | Assay Description Inhibition of human SGLT2 | Eur J Med Chem 184: (2019) Article DOI: 10.1016/j.ejmech.2019.111773 BindingDB Entry DOI: 10.7270/Q2TT4V97 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Rattus norvegicus) | BDBM50342885 ((1S,2S,3S,4R,5S)-5-[4-Chloro-3-(4-ethoxybenzyl)phe...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.15 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of rat SGLT2 | J Med Chem 54: 2952-60 (2011) Article DOI: 10.1021/jm200049r BindingDB Entry DOI: 10.7270/Q2NZ880H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium/glucose cotransporter 2 (Homo sapiens (Human)) | BDBM50342885 ((1S,2S,3S,4R,5S)-5-[4-Chloro-3-(4-ethoxybenzyl)phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Haisco Pharmaceuticals Group Co. Ltd. Curated by ChEMBL | Assay Description Inhibition of human SGLT2 expressed in CHO cells assessed as decrease in uptake of [14C]AMG after 120 mins by TopCount method | J Med Chem 60: 4173-4184 (2017) Article DOI: 10.1021/acs.jmedchem.6b01818 BindingDB Entry DOI: 10.7270/Q2W37ZFS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||