

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Thioredoxin reductase 1, cytoplasmic (Rattus norvegicus) | BDBM50236319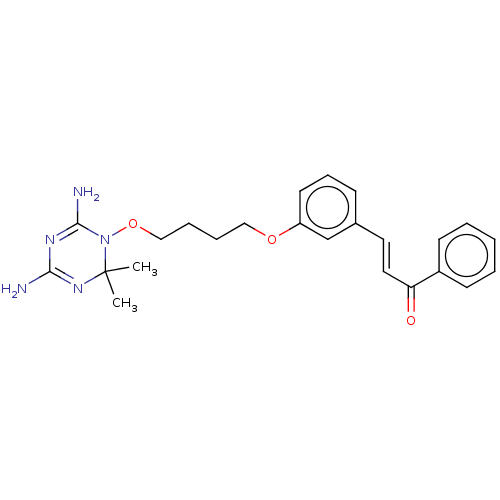 (CHEMBL4101407) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National University of Singapore Curated by ChEMBL | Assay Description Inhibitory concentration was evaluated against Farnesyltransferase in the farnesylation of H-ras protein | J Med Chem 60: 1734-1745 (2017) Article DOI: 10.1021/acs.jmedchem.6b01253 BindingDB Entry DOI: 10.7270/Q2FJ2K2V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thioredoxin reductase 1, cytoplasmic (Rattus norvegicus) | BDBM50236319 (CHEMBL4101407) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National University of Singapore Curated by ChEMBL | Assay Description Inhibition of recombinant rat liver thioredoxin reductase after 30 mins by DTNB reduction assay | J Med Chem 60: 1734-1745 (2017) Article DOI: 10.1021/acs.jmedchem.6b01253 BindingDB Entry DOI: 10.7270/Q2FJ2K2V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||