

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50184785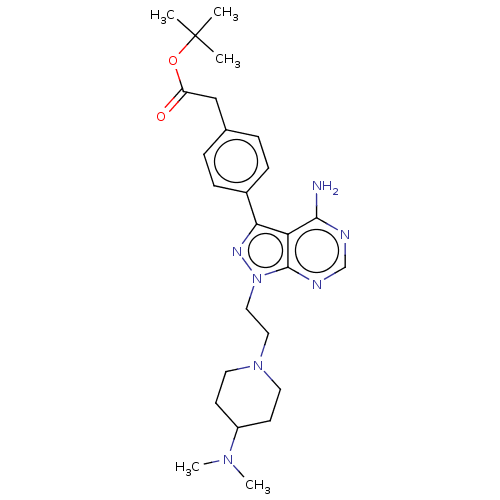 (CHEMBL3823620 | US10294227, Code 553) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim Pharmaceuticals Inc. | Assay Description Compound IC50 values were determined from 10-point, 1:3 dilution curves starting at either 100 μM or 10 μM with 10 μM ATP, by Reaction... | Bioorg Med Chem Lett 19: 773-7 (2009) BindingDB Entry DOI: 10.7270/Q27D2XFP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50184785 (CHEMBL3823620 | US10294227, Code 553) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Boehringer Ingelheim Pharmaceuticals Inc. | Assay Description Compound IC50 values were determined from 10-point, 1:3 dilution curves starting at either 100 μM or 10 μM with 10 μM ATP, by Reaction... | Bioorg Med Chem Lett 19: 773-7 (2009) BindingDB Entry DOI: 10.7270/Q27D2XFP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase ABL1 (Homo sapiens (Human)) | BDBM50184785 (CHEMBL3823620 | US10294227, Code 553) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.95E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Edinburgh Curated by ChEMBL | Assay Description Inhibition of human ABL using EAIYAAPFAKKK as substrate in presence of [gamma-33P]ATP | J Med Chem 59: 4697-710 (2016) Article DOI: 10.1021/acs.jmedchem.6b00065 BindingDB Entry DOI: 10.7270/Q2B85B2D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||