

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088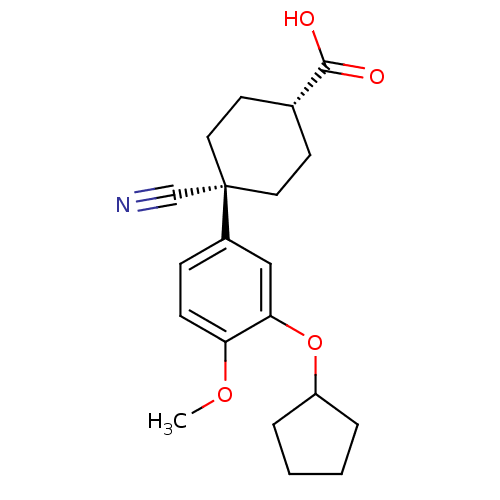 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Inhibitory concentration against human PDE4A isoform using a construct representing the common region of spliced variants expressed as GST-fusion pro... | Bioorg Med Chem Lett 12: 1457-61 (2002) BindingDB Entry DOI: 10.7270/Q2KP82PM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Inhibition of human Phosphodiesterase 4A isoform using construct representing the common region of spliced variants expressed as GST-fusion proteins ... | Bioorg Med Chem Lett 12: 3009-13 (2002) BindingDB Entry DOI: 10.7270/Q2V125B1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Inhibition of human recombinant PDE4A | J Med Chem 50: 344-9 (2007) Article DOI: 10.1021/jm060904g BindingDB Entry DOI: 10.7270/Q2K0753Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of PDE4A (unknown origin) | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112492 BindingDB Entry DOI: 10.7270/Q2R21511 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 398 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG Curated by ChEMBL | Assay Description Inhibition of human Phosphodiesterase 4A from peripheral blood mononuclear cells | Bioorg Med Chem Lett 12: 233-5 (2001) BindingDB Entry DOI: 10.7270/Q2MG7Q1T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG Curated by ChEMBL | Assay Description Inhibitory activity against Phosphodiesterase 4A (PDE4A) from human source expressed in Saccharomyces cerevisiae | J Med Chem 43: 675-82 (2000) BindingDB Entry DOI: 10.7270/Q2N58N3G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| cAMP-specific 3',5'-cyclic phosphodiesterase 4A (Homo sapiens (Human)) | BDBM50346088 ((1r,4r)-4-cyano-4-(3-(cyclopentyloxy)-4-methoxyphe...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research Curated by ChEMBL | Assay Description Inhibitory concentration of compound against LPS- induced TNF-alpha production in whole human blood assay | Bioorg Med Chem Lett 12: 1457-61 (2002) BindingDB Entry DOI: 10.7270/Q2KP82PM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||