 Found 2607 hits of ec50 data for polymerid = 2213
Found 2607 hits of ec50 data for polymerid = 2213 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430798
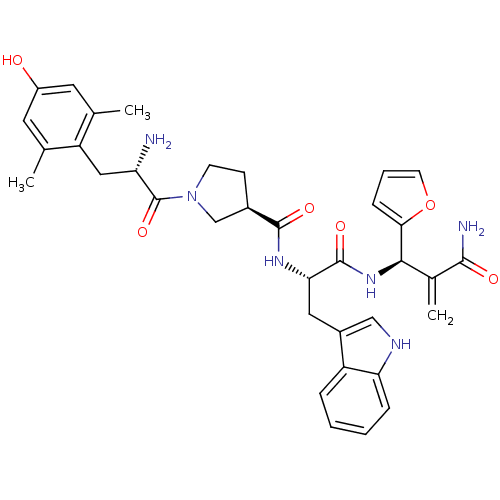
(CHEMBL2335120)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CC[C@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C35H40N6O6/c1-19-13-24(42)14-20(2)26(19)16-27(36)35(46)41-11-10-22(18-41)33(44)39-29(15-23-17-38-28-8-5-4-7-25(23)28)34(45)40-31(21(3)32(37)43)30-9-6-12-47-30/h4-9,12-14,17,22,27,29,31,38,42H,3,10-11,15-16,18,36H2,1-2H3,(H2,37,43)(H,39,44)(H,40,45)/t22-,27+,29+,31-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0000420 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells after 60 mins by liquid scintillation c... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430801
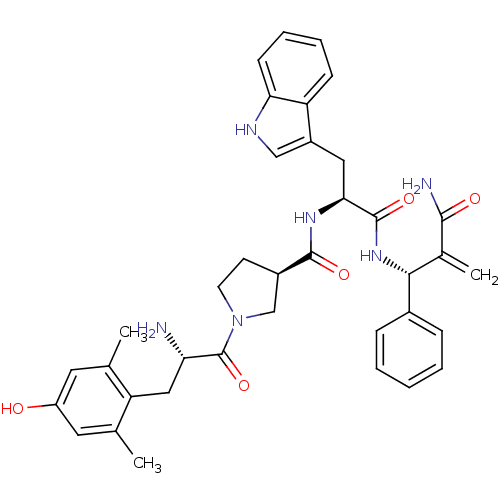
(CHEMBL2334776)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CC[C@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccccc1 |r| Show InChI InChI=1S/C37H42N6O5/c1-21-15-27(44)16-22(2)29(21)18-30(38)37(48)43-14-13-25(20-43)35(46)41-32(17-26-19-40-31-12-8-7-11-28(26)31)36(47)42-33(23(3)34(39)45)24-9-5-4-6-10-24/h4-12,15-16,19,25,30,32-33,40,44H,3,13-14,17-18,20,38H2,1-2H3,(H2,39,45)(H,41,46)(H,42,47)/t25-,30+,32+,33-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0000910 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430803
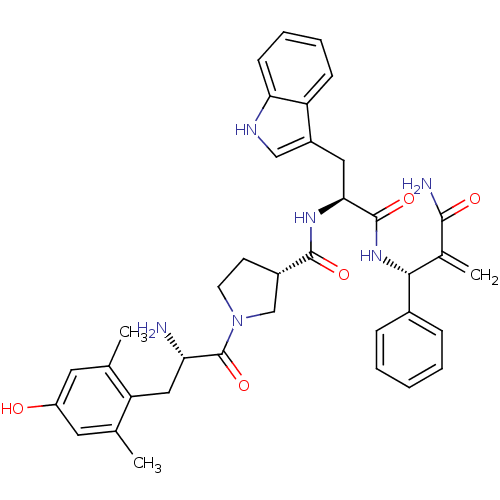
(CHEMBL2334774)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CC[C@@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccccc1 |r| Show InChI InChI=1S/C37H42N6O5/c1-21-15-27(44)16-22(2)29(21)18-30(38)37(48)43-14-13-25(20-43)35(46)41-32(17-26-19-40-31-12-8-7-11-28(26)31)36(47)42-33(23(3)34(39)45)24-9-5-4-6-10-24/h4-12,15-16,19,25,30,32-33,40,44H,3,13-14,17-18,20,38H2,1-2H3,(H2,39,45)(H,41,46)(H,42,47)/t25-,30-,32-,33+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.000107 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430802
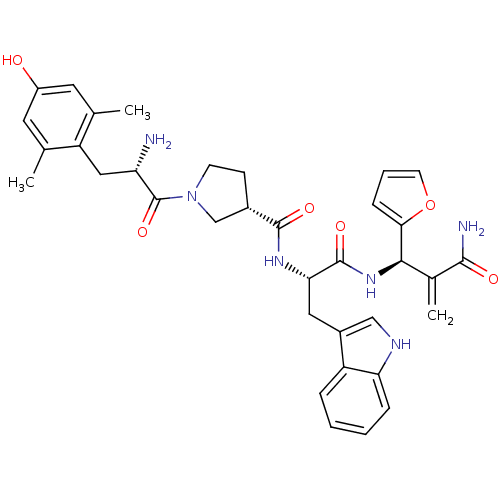
(CHEMBL2334775)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CC[C@@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C35H40N6O6/c1-19-13-24(42)14-20(2)26(19)16-27(36)35(46)41-11-10-22(18-41)33(44)39-29(15-23-17-38-28-8-5-4-7-25(23)28)34(45)40-31(21(3)32(37)43)30-9-6-12-47-30/h4-9,12-14,17,22,27,29,31,38,42H,3,10-11,15-16,18,36H2,1-2H3,(H2,37,43)(H,39,44)(H,40,45)/t22-,27-,29-,31+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.000109 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430799
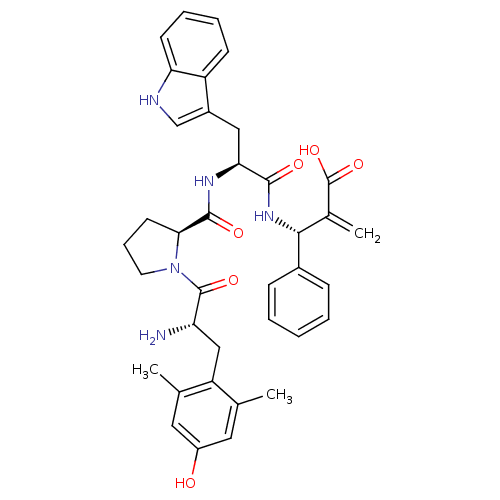
(CHEMBL2334772)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(O)=O)c1ccccc1 |r| Show InChI InChI=1S/C37H41N5O6/c1-21-16-26(43)17-22(2)28(21)19-29(38)36(46)42-15-9-14-32(42)35(45)40-31(18-25-20-39-30-13-8-7-12-27(25)30)34(44)41-33(23(3)37(47)48)24-10-5-4-6-11-24/h4-8,10-13,16-17,20,29,31-33,39,43H,3,9,14-15,18-19,38H2,1-2H3,(H,40,45)(H,41,44)(H,47,48)/t29-,31-,32-,33+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.000130 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50594825
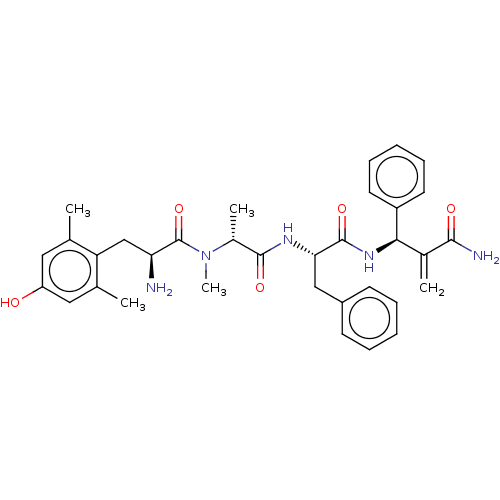
(CHEMBL5198856)Show SMILES C[C@@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@H](C(=C)C(N)=O)c1ccccc1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.000515 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01915
BindingDB Entry DOI: 10.7270/Q22B931S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430800
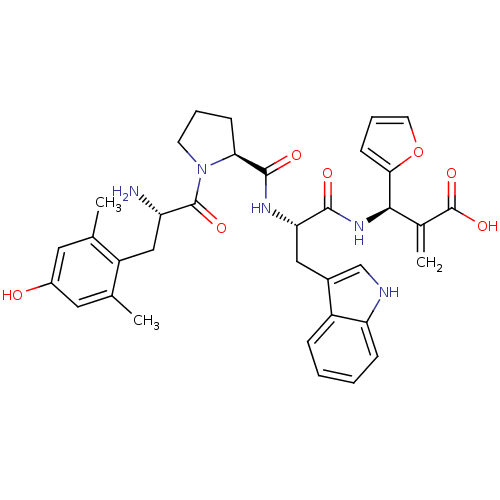
(CHEMBL2334773)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(O)=O)c1ccco1 |r| Show InChI InChI=1S/C35H39N5O7/c1-19-14-23(41)15-20(2)25(19)17-26(36)34(44)40-12-6-10-29(40)33(43)38-28(16-22-18-37-27-9-5-4-8-24(22)27)32(42)39-31(21(3)35(45)46)30-11-7-13-47-30/h4-5,7-9,11,13-15,18,26,28-29,31,37,41H,3,6,10,12,16-17,36H2,1-2H3,(H,38,43)(H,39,42)(H,45,46)/t26-,28-,29-,31+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.000516 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50095155
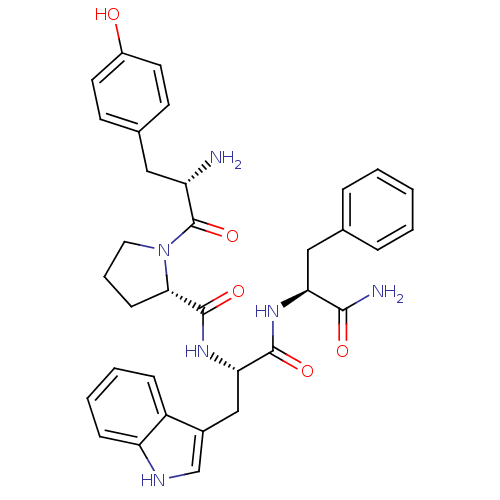
((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00100 | n/a | n/a | n/a | n/a |
Medical University
Curated by ChEMBL
| Assay Description
Activity at mu opioid receptor assessed as increase in calcium level in CHO cells by aequorin luminescence based calcium assay |
J Med Chem 50: 512-20 (2007)
Article DOI: 10.1021/jm060998u
BindingDB Entry DOI: 10.7270/Q28915JT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50393257
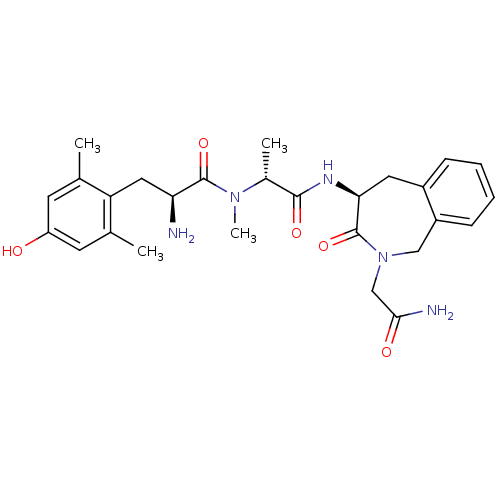
(CHEMBL2151735)Show SMILES C[C@@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@H]1Cc2ccccc2CN(CC(N)=O)C1=O |r| Show InChI InChI=1S/C27H35N5O5/c1-15-9-20(33)10-16(2)21(15)12-22(28)26(36)31(4)17(3)25(35)30-23-11-18-7-5-6-8-19(18)13-32(27(23)37)14-24(29)34/h5-10,17,22-23,33H,11-14,28H2,1-4H3,(H2,29,34)(H,30,35)/t17-,22+,23+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00174 | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in HEK293 cell membrane assessed as inhibition of forskolin-induced cAMP accumulation by by liquid scintillat... |
J Med Chem 54: 7848-59 (2011)
Article DOI: 10.1021/jm200894e
BindingDB Entry DOI: 10.7270/Q25X2B1T |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50528966
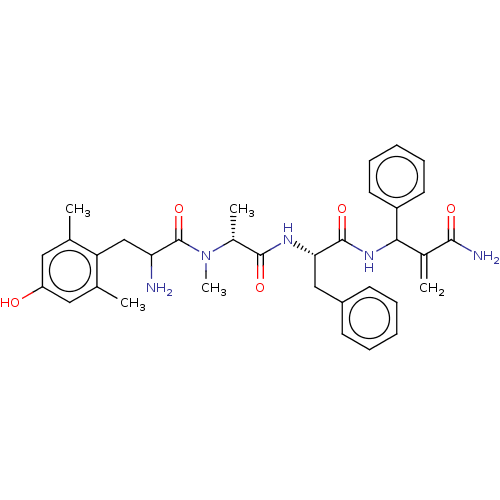
(CHEMBL4521879)Show SMILES C[C@@H](N(C)C(=O)C(N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1ccccc1)C(=O)NC(C(=C)C(N)=O)c1ccccc1 |r| Show InChI InChI=1S/C34H41N5O5/c1-20-16-26(40)17-21(2)27(20)19-28(35)34(44)39(5)23(4)32(42)37-29(18-24-12-8-6-9-13-24)33(43)38-30(22(3)31(36)41)25-14-10-7-11-15-25/h6-17,23,28-30,40H,3,18-19,35H2,1-2,4-5H3,(H2,36,41)(H,37,42)(H,38,43)/t23-,28?,29+,30?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00177 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation counting |
Eur J Med Chem 183: (2019)
Article DOI: 10.1016/j.ejmech.2019.111701
BindingDB Entry DOI: 10.7270/Q2668HN9 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50528967
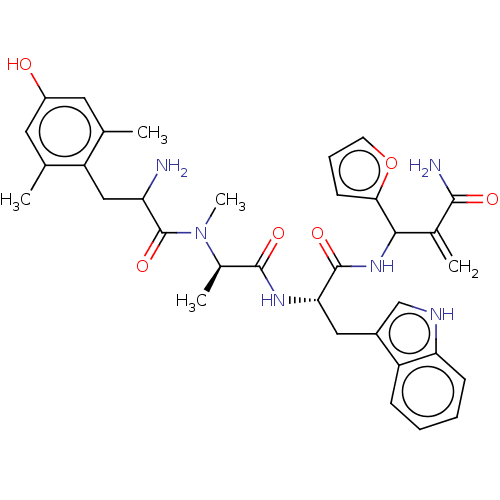
(CHEMBL4439415)Show SMILES C[C@@H](N(C)C(=O)C(N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NC(C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C34H40N6O6/c1-18-13-23(41)14-19(2)25(18)16-26(35)34(45)40(5)21(4)32(43)38-28(15-22-17-37-27-10-7-6-9-24(22)27)33(44)39-30(20(3)31(36)42)29-11-8-12-46-29/h6-14,17,21,26,28,30,37,41H,3,15-16,35H2,1-2,4-5H3,(H2,36,42)(H,38,43)(H,39,44)/t21-,26?,28+,30?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00177 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation counting |
Eur J Med Chem 183: (2019)
Article DOI: 10.1016/j.ejmech.2019.111701
BindingDB Entry DOI: 10.7270/Q2668HN9 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50528965
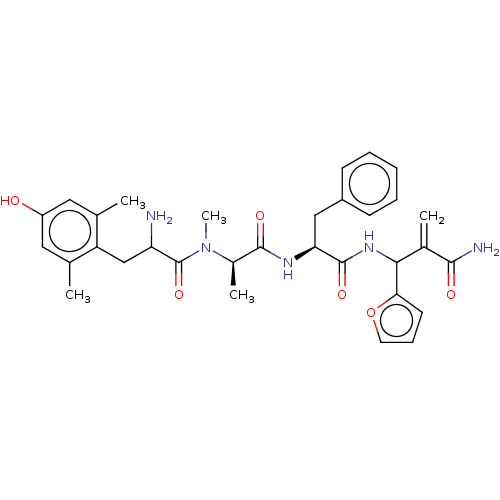
(CHEMBL4550234)Show SMILES C[C@@H](N(C)C(=O)C(N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1ccccc1)C(=O)NC(C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C32H39N5O6/c1-18-14-23(38)15-19(2)24(18)17-25(33)32(42)37(5)21(4)30(40)35-26(16-22-10-7-6-8-11-22)31(41)36-28(20(3)29(34)39)27-12-9-13-43-27/h6-15,21,25-26,28,38H,3,16-17,33H2,1-2,4-5H3,(H2,34,39)(H,35,40)(H,36,41)/t21-,25?,26+,28?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00177 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation counting |
Eur J Med Chem 183: (2019)
Article DOI: 10.1016/j.ejmech.2019.111701
BindingDB Entry DOI: 10.7270/Q2668HN9 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50528960
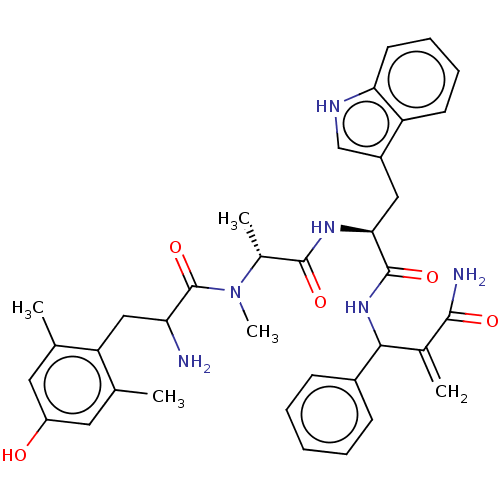
(CHEMBL4450250)Show SMILES C[C@@H](N(C)C(=O)C(N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NC(C(=C)C(N)=O)c1ccccc1 |r| Show InChI InChI=1S/C36H42N6O5/c1-20-15-26(43)16-21(2)28(20)18-29(37)36(47)42(5)23(4)34(45)40-31(17-25-19-39-30-14-10-9-13-27(25)30)35(46)41-32(22(3)33(38)44)24-11-7-6-8-12-24/h6-16,19,23,29,31-32,39,43H,3,17-18,37H2,1-2,4-5H3,(H2,38,44)(H,40,45)(H,41,46)/t23-,29?,31+,32?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00177 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation counting |
Eur J Med Chem 183: (2019)
Article DOI: 10.1016/j.ejmech.2019.111701
BindingDB Entry DOI: 10.7270/Q2668HN9 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50594821
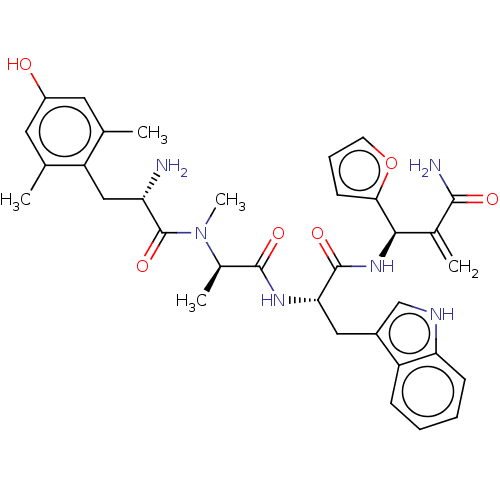
(CHEMBL5175179)Show SMILES C[C@@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00180 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01915
BindingDB Entry DOI: 10.7270/Q22B931S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50594822
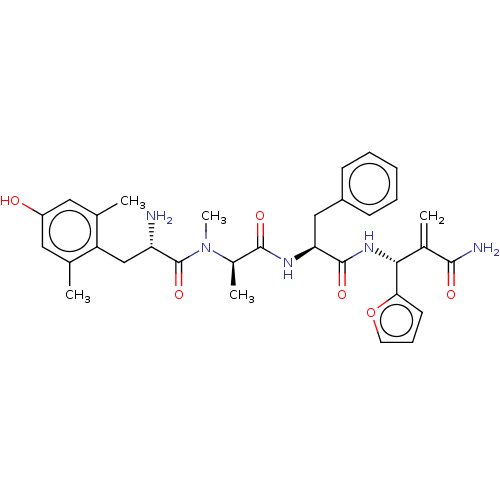
(CHEMBL5182849)Show SMILES C[C@@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00201 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01915
BindingDB Entry DOI: 10.7270/Q22B931S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529413
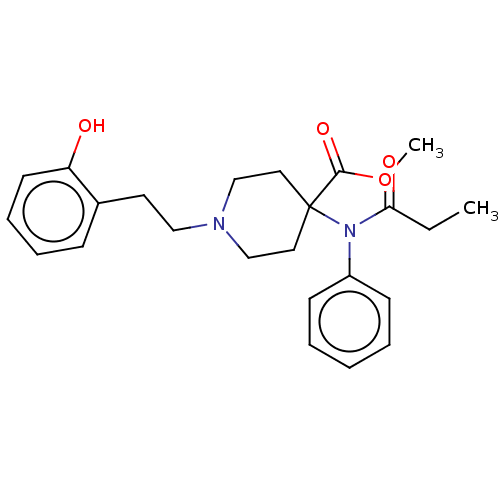
(CHEMBL4563672)Show SMILES CCC(=O)N(c1ccccc1)C1(CCN(CCc2ccccc2O)CC1)C(=O)OC Show InChI InChI=1S/C24H30N2O4/c1-3-22(28)26(20-10-5-4-6-11-20)24(23(29)30-2)14-17-25(18-15-24)16-13-19-9-7-8-12-21(19)27/h4-12,27H,3,13-18H2,1-2H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00240 | n/a | n/a | n/a | n/a |
United States Army CCDC Chemical Biological Center
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO-K1 cells assessed as cAMP accumulation incubated for 30 mins and measured after 1 hr by Eu-cAMP tracer... |
ACS Med Chem Lett 10: 1568-1572 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00404
BindingDB Entry DOI: 10.7270/Q2JD5175 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50594820
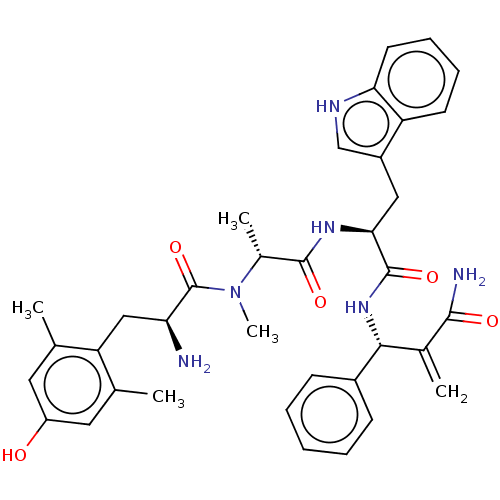
(CHEMBL5176887)Show SMILES C[C@@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccccc1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00400 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01915
BindingDB Entry DOI: 10.7270/Q22B931S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50012477
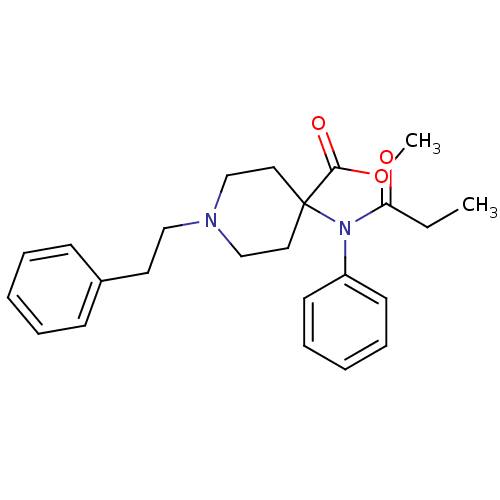
(1-Phenethyl-4-(phenyl-propionyl-amino)-piperidine-...)Show SMILES CCC(=O)N(c1ccccc1)C1(CCN(CCc2ccccc2)CC1)C(=O)OC Show InChI InChI=1S/C24H30N2O3/c1-3-22(27)26(21-12-8-5-9-13-21)24(23(28)29-2)15-18-25(19-16-24)17-14-20-10-6-4-7-11-20/h4-13H,3,14-19H2,1-2H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00490 | n/a | n/a | n/a | n/a |
United States Army CCDC Chemical Biological Center
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO-K1 cells assessed as cAMP accumulation incubated for 30 mins and measured after 1 hr by Eu-cAMP tracer... |
ACS Med Chem Lett 10: 1568-1572 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00404
BindingDB Entry DOI: 10.7270/Q2JD5175 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529410
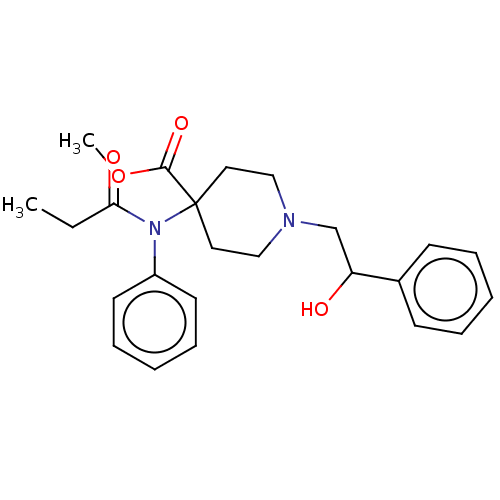
(CHEMBL4463749)Show SMILES CCC(=O)N(c1ccccc1)C1(CCN(CC(O)c2ccccc2)CC1)C(=O)OC Show InChI InChI=1S/C24H30N2O4/c1-3-22(28)26(20-12-8-5-9-13-20)24(23(29)30-2)14-16-25(17-15-24)18-21(27)19-10-6-4-7-11-19/h4-13,21,27H,3,14-18H2,1-2H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00510 | n/a | n/a | n/a | n/a |
United States Army CCDC Chemical Biological Center
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO-K1 cells assessed as cAMP accumulation incubated for 30 mins and measured after 1 hr by Eu-cAMP tracer... |
ACS Med Chem Lett 10: 1568-1572 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00404
BindingDB Entry DOI: 10.7270/Q2JD5175 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430806
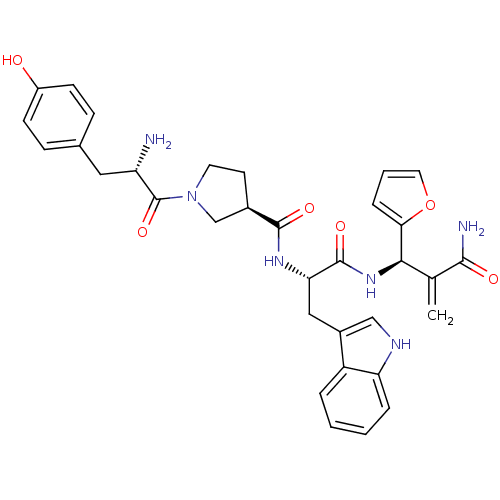
(CHEMBL2334771)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CC[C@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C33H36N6O6/c1-19(30(35)41)29(28-7-4-14-45-28)38-32(43)27(16-22-17-36-26-6-3-2-5-24(22)26)37-31(42)21-12-13-39(18-21)33(44)25(34)15-20-8-10-23(40)11-9-20/h2-11,14,17,21,25,27,29,36,40H,1,12-13,15-16,18,34H2,(H2,35,41)(H,37,42)(H,38,43)/t21-,25+,27+,29-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.00853 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529415
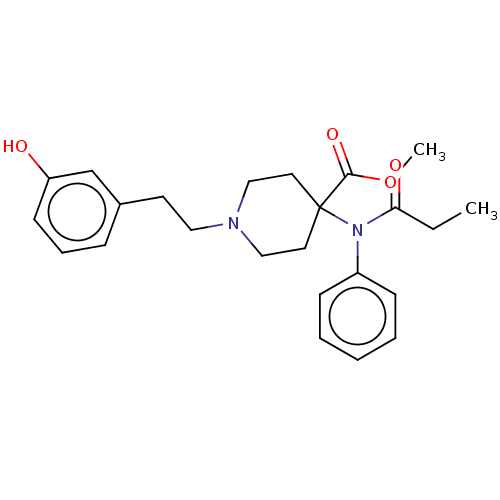
(CHEMBL4588535)Show SMILES CCC(=O)N(c1ccccc1)C1(CCN(CCc2cccc(O)c2)CC1)C(=O)OC Show InChI InChI=1S/C24H30N2O4/c1-3-22(28)26(20-9-5-4-6-10-20)24(23(29)30-2)13-16-25(17-14-24)15-12-19-8-7-11-21(27)18-19/h4-11,18,27H,3,12-17H2,1-2H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0140 | n/a | n/a | n/a | n/a |
United States Army CCDC Chemical Biological Center
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO-K1 cells assessed as cAMP accumulation incubated for 30 mins and measured after 1 hr by Eu-cAMP tracer... |
ACS Med Chem Lett 10: 1568-1572 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00404
BindingDB Entry DOI: 10.7270/Q2JD5175 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50550066
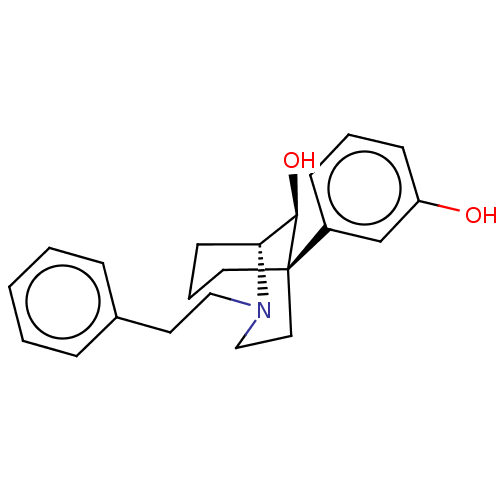
(CHEMBL4798954)Show SMILES [H][C@]12CCC[C@@](CCN1CCc1ccccc1)([C@@H]2O)c1cccc(O)c1 |r,THB:9:8:4.3.2:17| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0170 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Agonist activity at human MOR expressed in CHOK1 cells assessed as inhibition of forskolin-stimulated cAMP accumulation incubated for 30 mins by lumi... |
Citation and Details
Article DOI: 10.1039/d0md00104j
BindingDB Entry DOI: 10.7270/Q2K64NQR |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529414
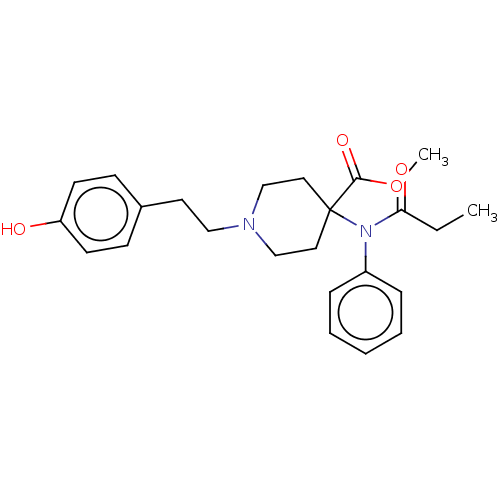
(CHEMBL4578287)Show SMILES CCC(=O)N(c1ccccc1)C1(CCN(CCc2ccc(O)cc2)CC1)C(=O)OC Show InChI InChI=1S/C24H30N2O4/c1-3-22(28)26(20-7-5-4-6-8-20)24(23(29)30-2)14-17-25(18-15-24)16-13-19-9-11-21(27)12-10-19/h4-12,27H,3,13-18H2,1-2H3 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0280 | n/a | n/a | n/a | n/a |
United States Army CCDC Chemical Biological Center
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO-K1 cells assessed as cAMP accumulation incubated for 30 mins and measured after 1 hr by Eu-cAMP tracer... |
ACS Med Chem Lett 10: 1568-1572 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00404
BindingDB Entry DOI: 10.7270/Q2JD5175 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50594865
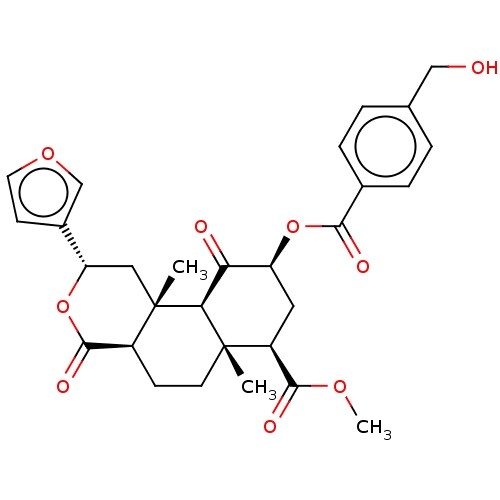
(CHEMBL5186887)Show SMILES [H][C@@]12CC[C@@]3(C)[C@@H](C[C@H](OC(=O)c4ccc(CO)cc4)C(=O)[C@]3([H])[C@@]1(C)C[C@H](OC2=O)c1ccoc1)C(=O)OC |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01915
BindingDB Entry DOI: 10.7270/Q22B931S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50395060
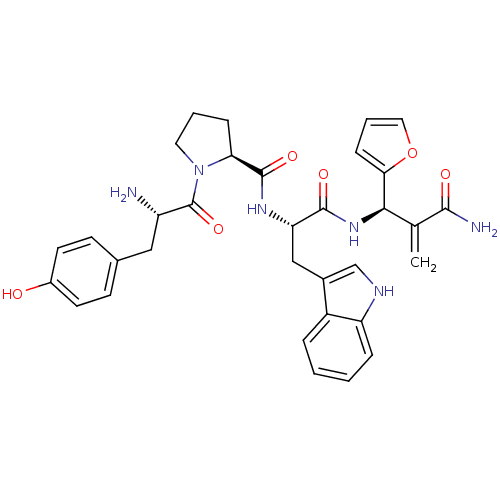
(CHEMBL2163916)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C33H36N6O6/c1-19(30(35)41)29(28-9-5-15-45-28)38-31(42)26(17-21-18-36-25-7-3-2-6-23(21)25)37-32(43)27-8-4-14-39(27)33(44)24(34)16-20-10-12-22(40)13-11-20/h2-3,5-7,9-13,15,18,24,26-27,29,36,40H,1,4,8,14,16-17,34H2,(H2,35,41)(H,37,43)(H,38,42)/t24-,26-,27-,29+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0334 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at mu opioid receptor expressed in HEK293 cells assessed as inhibition of forskolin-induced cAMP accumulation after 30 mins by liqui... |
J Med Chem 55: 6224-36 (2012)
Article DOI: 10.1021/jm300664y
BindingDB Entry DOI: 10.7270/Q2T154S8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50395059
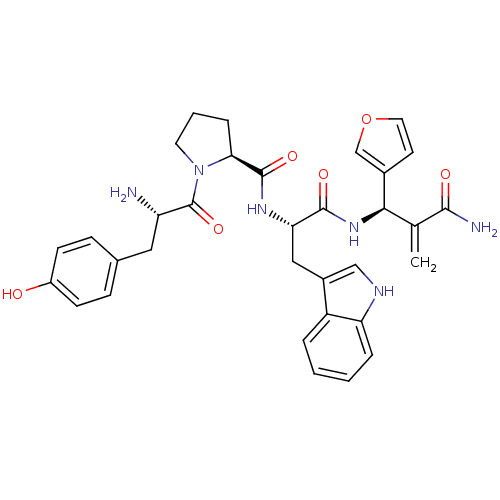
(CHEMBL2163917)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccoc1 |r| Show InChI InChI=1S/C33H36N6O6/c1-19(30(35)41)29(21-12-14-45-18-21)38-31(42)27(16-22-17-36-26-6-3-2-5-24(22)26)37-32(43)28-7-4-13-39(28)33(44)25(34)15-20-8-10-23(40)11-9-20/h2-3,5-6,8-12,14,17-18,25,27-29,36,40H,1,4,7,13,15-16,34H2,(H2,35,41)(H,37,43)(H,38,42)/t25-,27-,28-,29+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0342 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at mu opioid receptor expressed in HEK293 cells assessed as inhibition of forskolin-induced cAMP accumulation after 30 mins by liqui... |
J Med Chem 55: 6224-36 (2012)
Article DOI: 10.1021/jm300664y
BindingDB Entry DOI: 10.7270/Q2T154S8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430807
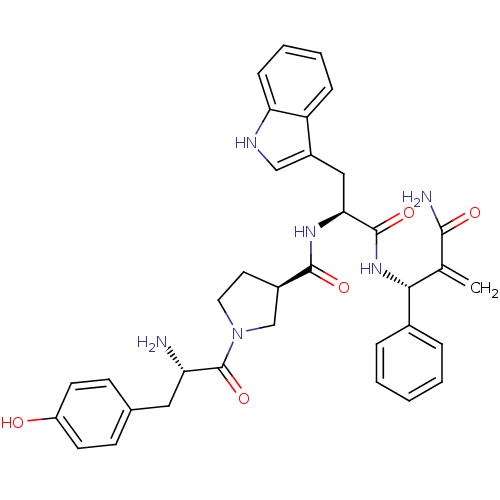
(CHEMBL2334770)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CC[C@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccccc1 |r| Show InChI InChI=1S/C35H38N6O5/c1-21(32(37)43)31(23-7-3-2-4-8-23)40-34(45)30(18-25-19-38-29-10-6-5-9-27(25)29)39-33(44)24-15-16-41(20-24)35(46)28(36)17-22-11-13-26(42)14-12-22/h2-14,19,24,28,30-31,38,42H,1,15-18,20,36H2,(H2,37,43)(H,39,44)(H,40,45)/t24-,28+,30+,31-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0397 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM518593
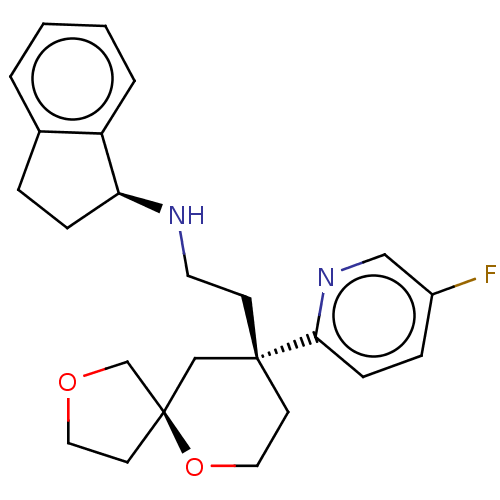
(US11124523, Example (+)-28b)Show SMILES Fc1ccc(nc1)[C@]1(CCN[C@H]2CCc3ccccc23)CCO[C@]2(CCOC2)C1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The experiment was performed using a cAMP detection kit from Cisbio (Cisbio #62AM4PEJ). |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2M90CTM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50027230
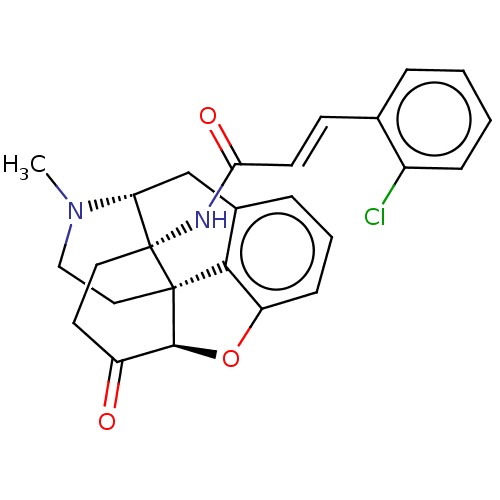
(CHEMBL2113666)Show SMILES [H][C@@]12Oc3cccc4C[C@@]5([H])N(C)CC[C@@]1(c34)[C@]5(CCC2=O)NC(=O)\C=C\c1ccccc1Cl |r,THB:12:11:17:8.7.16| Show InChI InChI=1S/C26H25ClN2O3/c1-29-14-13-25-23-17-6-4-8-20(23)32-24(25)19(30)11-12-26(25,21(29)15-17)28-22(31)10-9-16-5-2-3-7-18(16)27/h2-10,21,24H,11-15H2,1H3,(H,28,31)/b10-9+/t21-,24+,25+,26-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Stimulation of [35S]GTPgammaS binding to human recombinant MOR |
J Med Chem 49: 5333-8 (2006)
Article DOI: 10.1021/jm0604777
BindingDB Entry DOI: 10.7270/Q2WH2QSM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50193546
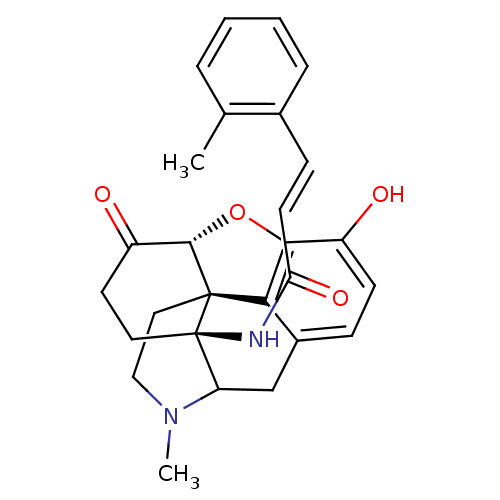
(14beta-(2'-methylcinnamoylamino)-7,8-dihydromorphi...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(CC1[C@@]3(CCC4=O)NC(=O)\C=C\c1ccccc1C)ccc5O |TLB:29:9:12:1.3.2,7:8:12:1.3.2| Show InChI InChI=1S/C27H28N2O4/c1-16-5-3-4-6-17(16)8-10-22(32)28-27-12-11-20(31)25-26(27)13-14-29(2)21(27)15-18-7-9-19(30)24(33-25)23(18)26/h3-10,21,25,30H,11-15H2,1-2H3,(H,28,32)/b10-8+/t21?,25-,26-,27+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
University of Bristol
Curated by ChEMBL
| Assay Description
Stimulation of [35S]GTPgammaS binding to human recombinant MOR |
J Med Chem 49: 5333-8 (2006)
Article DOI: 10.1021/jm0604777
BindingDB Entry DOI: 10.7270/Q2WH2QSM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50139013
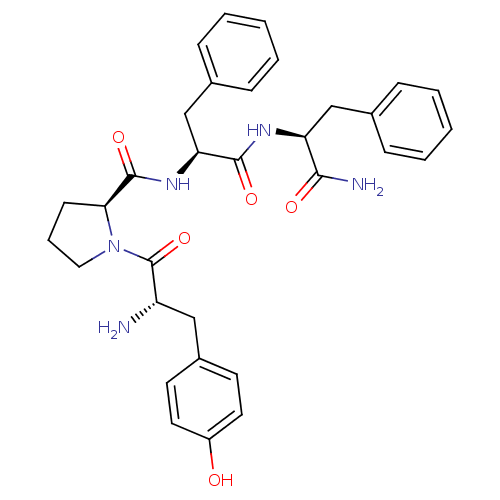
((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26-,27-,28-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
Medical University of Lodz
Curated by ChEMBL
| Assay Description
Antagonist activity at mu opioid receptor expressed in CHO cells assessed as release of intracellular calcium ions by aequorin luminescence-based cal... |
Bioorg Med Chem Lett 18: 1350-3 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.009
BindingDB Entry DOI: 10.7270/Q2QR4WWN |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50139013

((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26-,27-,28-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
Medical University
Curated by ChEMBL
| Assay Description
Activity at mu opioid receptor assessed as increase in calcium level in CHO cells by aequorin luminescence based calcium assay |
J Med Chem 50: 512-20 (2007)
Article DOI: 10.1021/jm060998u
BindingDB Entry DOI: 10.7270/Q28915JT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430805
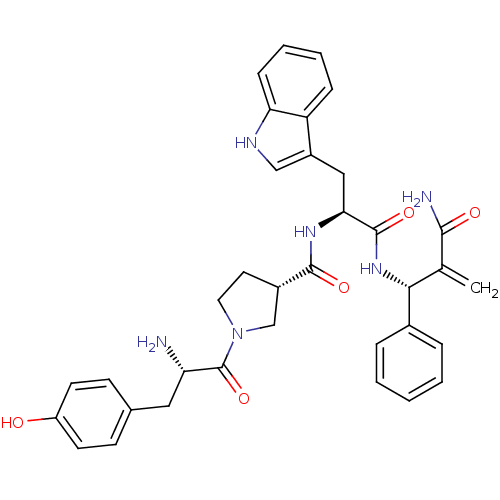
(CHEMBL2334768)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CC[C@@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccccc1 |r| Show InChI InChI=1S/C35H38N6O5/c1-21(32(37)43)31(23-7-3-2-4-8-23)40-34(45)30(18-25-19-38-29-10-6-5-9-27(25)29)39-33(44)24-15-16-41(20-24)35(46)28(36)17-22-11-13-26(42)14-12-22/h2-14,19,24,28,30-31,38,42H,1,15-18,20,36H2,(H2,37,43)(H,39,44)(H,40,45)/t24-,28-,30-,31+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0570 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50127700
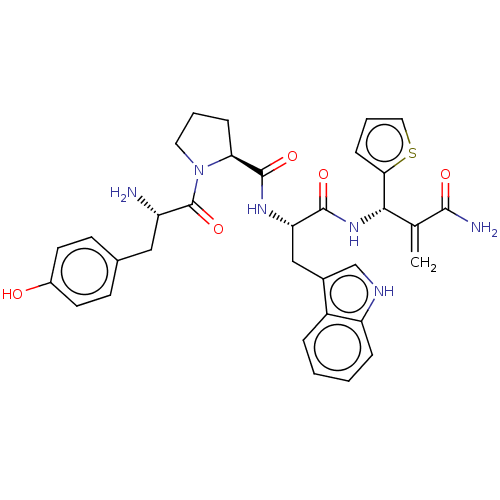
(CHEMBL3629337)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](C(=C)C(N)=O)c1cccs1 |r| Show InChI InChI=1S/C33H36N6O5S/c1-19(30(35)41)29(28-9-5-15-45-28)38-31(42)26(17-21-18-36-25-7-3-2-6-23(21)25)37-32(43)27-8-4-14-39(27)33(44)24(34)16-20-10-12-22(40)13-11-20/h2-3,5-7,9-13,15,18,24,26-27,29,36,40H,1,4,8,14,16-17,34H2,(H2,35,41)(H,37,43)(H,38,42)/t24-,26-,27-,29-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0629 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at mu opioid receptor (unknown origin) transfected in HEK293 cells assessed as inhibition of forskolin-stimulated cAMP production |
Bioorg Med Chem Lett 25: 5393-7 (2015)
Article DOI: 10.1016/j.bmcl.2015.09.025
BindingDB Entry DOI: 10.7270/Q24M96CW |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM397187
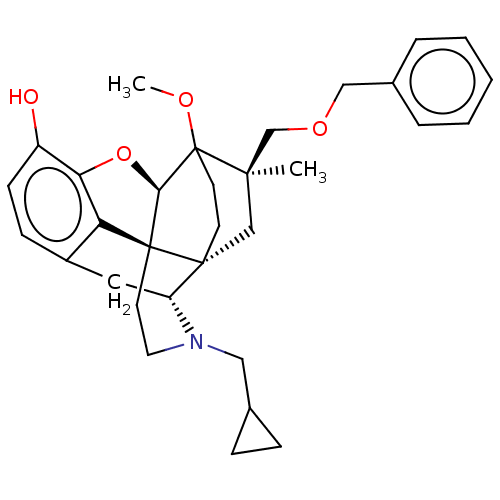
(US9988392, Compound 7)Show SMILES COC12CC[C@@]3(C[C@]1(C)COCc1ccccc1)[C@H]1Cc4ccc(O)c5O[C@@H]2[C@]3(CCN1CC1CC1)c45 |r,TLB:8:7:28.27:4.3,9:7:28.27:4.3| Show InChI InChI=1S/C32H39NO4/c1-29(20-36-18-22-6-4-3-5-7-22)19-30-12-13-32(29,35-2)28-31(30)14-15-33(17-21-8-9-21)25(30)16-23-10-11-24(34)27(37-28)26(23)31/h3-7,10-11,21,25,28,34H,8-9,12-20H2,1-2H3/t25-,28-,29-,30-,31+,32?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 0.0640 | n/a | n/a | n/a | n/a |
BioCryst Pharmaceuticals
| Assay Description
μ-Opioid:Radioligand dose-displacement binding assays for μ-opioid receptors used 0.3 nM [3H]-diprenorphine (Perkin Elmer, Shelton, Conn.),... |
Bioorg Med Chem 17: 3934-58 (2009)
BindingDB Entry DOI: 10.7270/Q28K7CF0 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50127701
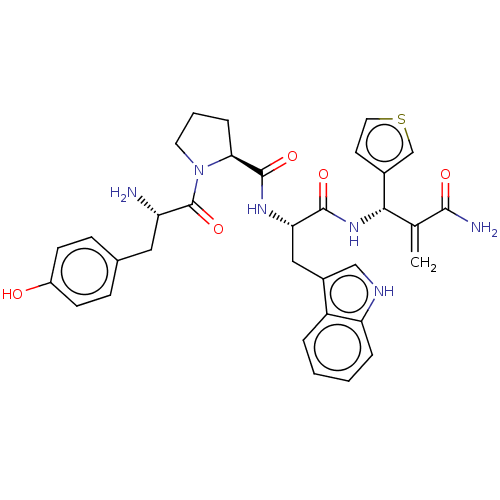
(CHEMBL3627737)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](C(=C)C(N)=O)c1ccsc1 |r| Show InChI InChI=1S/C33H36N6O5S/c1-19(30(35)41)29(21-12-14-45-18-21)38-31(42)27(16-22-17-36-26-6-3-2-5-24(22)26)37-32(43)28-7-4-13-39(28)33(44)25(34)15-20-8-10-23(40)11-9-20/h2-3,5-6,8-12,14,17-18,25,27-29,36,40H,1,4,7,13,15-16,34H2,(H2,35,41)(H,37,43)(H,38,42)/t25-,27-,28-,29-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0643 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at mu opioid receptor (unknown origin) transfected in HEK293 cells assessed as inhibition of forskolin-stimulated cAMP production |
Bioorg Med Chem Lett 25: 5393-7 (2015)
Article DOI: 10.1016/j.bmcl.2015.09.025
BindingDB Entry DOI: 10.7270/Q24M96CW |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM518565
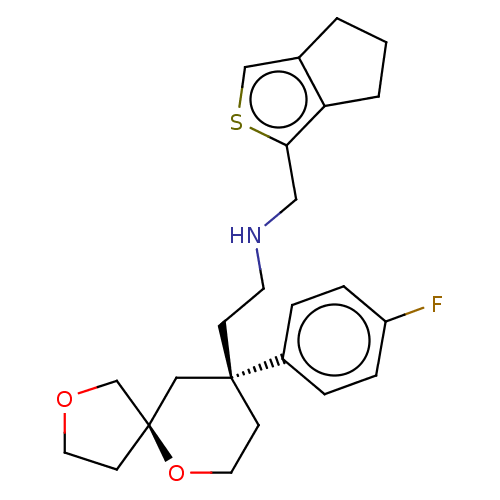
(US11124523, Example (+)-4)Show SMILES Fc1ccc(cc1)[C@]1(CCNCc2scc3CCCc23)CCO[C@]2(CCOC2)C1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The experiment was performed using a cAMP detection kit from Cisbio (Cisbio #62AM4PEJ). |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2M90CTM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50583967
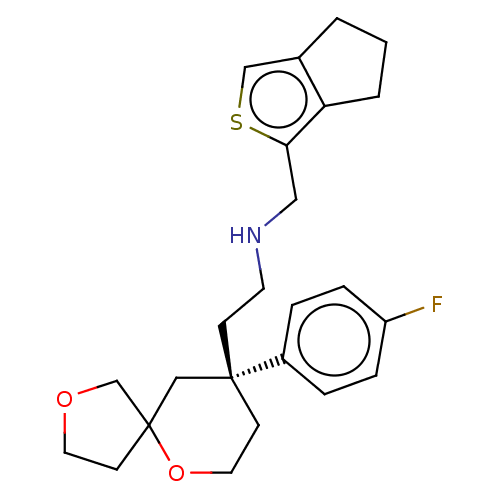
(CHEMBL5074742)Show SMILES Fc1ccc(cc1)[C@]1(CCNCc2scc3CCCc23)CCOC2(CCOC2)C1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Agonist activity at mu opioid receptor (unknown origin) assessed as increase in cAMP level incubated for 40 mins by spectrophotometry |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113986
BindingDB Entry DOI: 10.7270/Q2TX3K84 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50249017
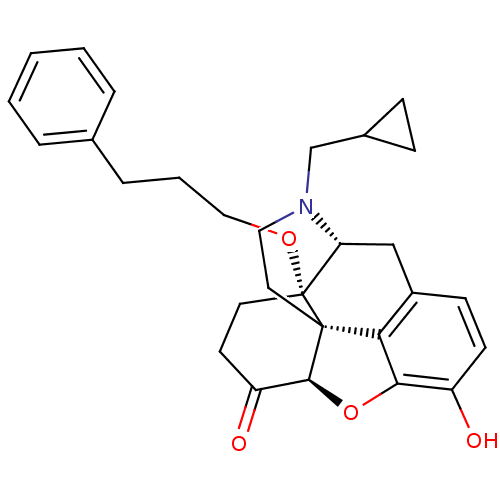
(14-O-phenylpropylnaltrexone | 4-cyclopropylmethyl-...)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35OCCCc1ccccc1 |r| Show InChI InChI=1S/C29H33NO4/c31-22-11-10-21-17-24-29(33-16-4-7-19-5-2-1-3-6-19)13-12-23(32)27-28(29,25(21)26(22)34-27)14-15-30(24)18-20-8-9-20/h1-3,5-6,10-11,20,24,27,31H,4,7-9,12-18H2/t24-,27+,28+,29-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | n/a | n/a | 0.0750 | n/a | n/a | n/a | n/a |
ALKERMES PHARMA IRELAND LIMITED
US Patent
| Assay Description
The Ki (binding affinity) for opioid receptors was determined using a competitive displacement assay as previously described in Neumeyer (Journal of ... |
US Patent US9656961 (2017)
BindingDB Entry DOI: 10.7270/Q2PR7Z13 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50249017

(14-O-phenylpropylnaltrexone | 4-cyclopropylmethyl-...)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35OCCCc1ccccc1 |r| Show InChI InChI=1S/C29H33NO4/c31-22-11-10-21-17-24-29(33-16-4-7-19-5-2-1-3-6-19)13-12-23(32)27-28(29,25(21)26(22)34-27)14-15-30(24)18-20-8-9-20/h1-3,5-6,10-11,20,24,27,31H,4,7-9,12-18H2/t24-,27+,28+,29-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | n/a | n/a | 0.0750 | n/a | n/a | n/a | n/a |
Universita degli Studi di Bari
| Assay Description
The Ki (binding affinity) for opioid receptors was determined using a competitive displacement assay as previously described in Neumeyer (Journal of ... |
J Med Chem 50: 4214-21 (2007)
BindingDB Entry DOI: 10.7270/Q2W66P26 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50393258
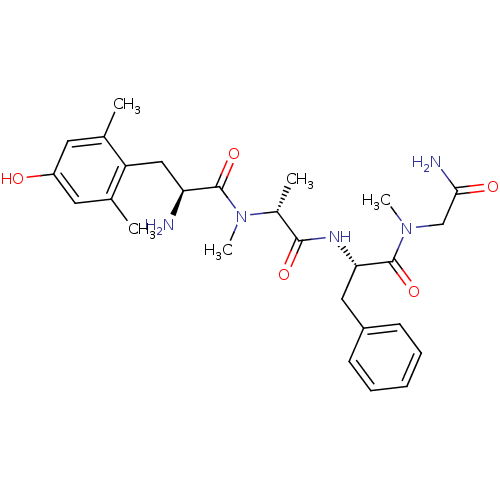
(CHEMBL2151734)Show SMILES C[C@@H](N(C)C(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N(C)CC(N)=O |r| Show InChI InChI=1S/C27H37N5O5/c1-16-11-20(33)12-17(2)21(16)14-22(28)26(36)32(5)18(3)25(35)30-23(13-19-9-7-6-8-10-19)27(37)31(4)15-24(29)34/h6-12,18,22-23,33H,13-15,28H2,1-5H3,(H2,29,34)(H,30,35)/t18-,22+,23+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0790 | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in HEK293 cell membrane assessed as inhibition of forskolin-induced cAMP accumulation by by liquid scintillat... |
J Med Chem 54: 7848-59 (2011)
Article DOI: 10.1021/jm200894e
BindingDB Entry DOI: 10.7270/Q25X2B1T |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50354650
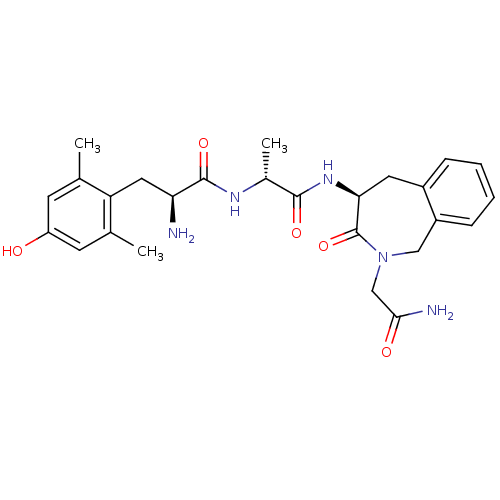
(CHEMBL1834247)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@H]1Cc2ccccc2CN(CC(N)=O)C1=O |r| Show InChI InChI=1S/C26H33N5O5/c1-14-8-19(32)9-15(2)20(14)11-21(27)25(35)29-16(3)24(34)30-22-10-17-6-4-5-7-18(17)12-31(26(22)36)13-23(28)33/h4-9,16,21-22,32H,10-13,27H2,1-3H3,(H2,28,33)(H,29,35)(H,30,34)/t16-,21+,22+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0832 | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in HEK293 cell membrane assessed as inhibition of forskolin-induced cAMP accumulation by by liquid scintillat... |
J Med Chem 54: 7848-59 (2011)
Article DOI: 10.1021/jm200894e
BindingDB Entry DOI: 10.7270/Q25X2B1T |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50292917
(17-Cyclopropylmethyl-3,14beta-dihydroxy-4,5r-epoxy...)Show SMILES Oc1ccc2C[C@H]3N(CC4CC4)CC[C@@]45[C@@H](Oc1c24)[C@H](CC[C@@]35O)NC(=O)c1ccc2ccccc2n1 |r| Show InChI InChI=1S/C30H31N3O4/c34-23-10-8-19-15-24-30(36)12-11-21(32-28(35)22-9-7-18-3-1-2-4-20(18)31-22)27-29(30,25(19)26(23)37-27)13-14-33(24)16-17-5-6-17/h1-4,7-10,17,21,24,27,34,36H,5-6,11-16H2,(H,32,35)/t21-,24+,27-,29-,30+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a |
Virginia Commonwealth University
Curated by ChEMBL
| Assay Description
Activity at monocloned mu opioid receptor expressed in CHO cells by [35S]GTPgammaS binding assay |
J Med Chem 52: 1416-27 (2010)
Article DOI: 10.1021/jm801272c
BindingDB Entry DOI: 10.7270/Q2416XZ6 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50430804
(CHEMBL2334769)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CC[C@@H](C1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C(=C)C(N)=O)c1ccco1 |r| Show InChI InChI=1S/C33H36N6O6/c1-19(30(35)41)29(28-7-4-14-45-28)38-32(43)27(16-22-17-36-26-6-3-2-5-24(22)26)37-31(42)21-12-13-39(18-21)33(44)25(34)15-20-8-10-23(40)11-9-20/h2-11,14,17,21,25,27,29,36,40H,1,12-13,15-16,18,34H2,(H2,35,41)(H,37,42)(H,38,43)/t21-,25-,27-,29+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0933 | n/a | n/a | n/a | n/a |
Lanzhou University
Curated by ChEMBL
| Assay Description
Agonist activity at FLAG-tagged mu-type opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-stimulated cAM... |
J Med Chem 56: 3102-14 (2013)
Article DOI: 10.1021/jm400195y
BindingDB Entry DOI: 10.7270/Q2KS6SW8 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50070386
(CHEMBL3408737)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(N)=O)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C30H42N8O5/c1-17-12-21(39)13-18(2)22(17)15-23(31)27(41)36-24(8-5-10-35-30(33)34)28(42)37-25-14-19-6-3-4-7-20(19)16-38(29(25)43)11-9-26(32)40/h3-4,6-7,12-13,23-25,39H,5,8-11,14-16,31H2,1-2H3,(H2,32,40)(H,36,41)(H,37,42)(H4,33,34,35)/t23-,24+,25-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Agonist activity at human MOR stably expressed in CHO cell membranes assessed as stimulation of [35S]-GTPgammaS binding incubated for 1 hr by liquid ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01376
BindingDB Entry DOI: 10.7270/Q2H41W2P |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM518566
(US11124523, Example (+)-5)Show SMILES Fc1ccc(nc1)[C@]1(CCNCc2scc3CCCc23)CCO[C@]2(CCOC2)C1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The experiment was performed using a cAMP detection kit from Cisbio (Cisbio #62AM4PEJ). |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2M90CTM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM518586

(US11124523, Example (+)-22)Show SMILES Cc1cccc(CNCC[C@]2(CCO[C@]3(CCOC3)C2)c2ccc(F)cn2)c1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The experiment was performed using a cAMP detection kit from Cisbio (Cisbio #62AM4PEJ). |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2M90CTM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50149384
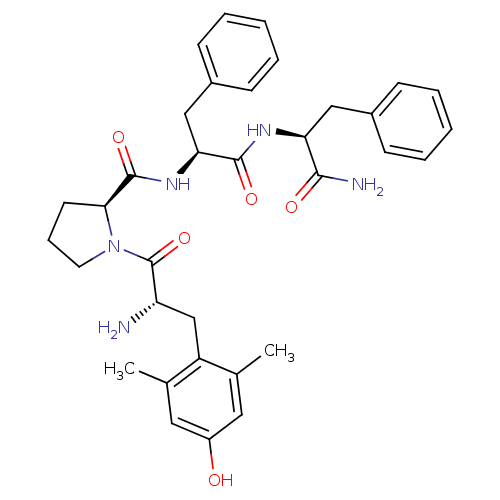
((S)-1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-pheny...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H41N5O5/c1-21-16-25(40)17-22(2)26(21)20-27(35)34(44)39-15-9-14-30(39)33(43)38-29(19-24-12-7-4-8-13-24)32(42)37-28(31(36)41)18-23-10-5-3-6-11-23/h3-8,10-13,16-17,27-30,40H,9,14-15,18-20,35H2,1-2H3,(H2,36,41)(H,37,42)(H,38,43)/t27-,28-,29-,30-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO cell membranes after 60 mins by [35S]GTPgammaS binding assay |
J Med Chem 61: 9784-9789 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01282
BindingDB Entry DOI: 10.7270/Q2F76G8Q |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50070386

(CHEMBL3408737)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(N)=O)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C30H42N8O5/c1-17-12-21(39)13-18(2)22(17)15-23(31)27(41)36-24(8-5-10-35-30(33)34)28(42)37-25-14-19-6-3-4-7-20(19)16-38(29(25)43)11-9-26(32)40/h3-4,6-7,12-13,23-25,39H,5,8-11,14-16,31H2,1-2H3,(H2,32,40)(H,36,41)(H,37,42)(H4,33,34,35)/t23-,24+,25-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO cell membranes after 60 mins by [35S]GTPgammaS binding assay |
J Med Chem 61: 9784-9789 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01282
BindingDB Entry DOI: 10.7270/Q2F76G8Q |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50199865
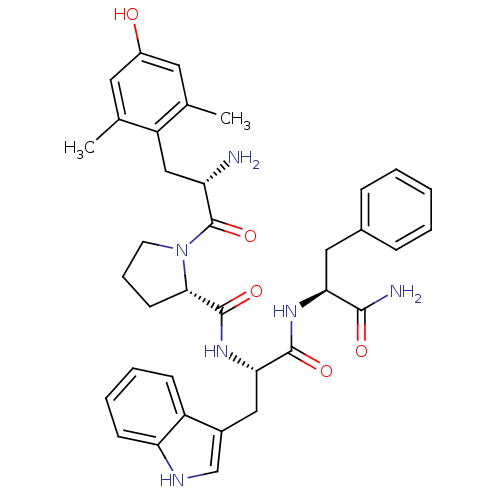
((S)-N-((S)-1-((S)-1-amino-1-oxo-3-phenylpropan-2-y...)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C36H42N6O5/c1-21-15-25(43)16-22(2)27(21)19-28(37)36(47)42-14-8-13-32(42)35(46)41-31(18-24-20-39-29-12-7-6-11-26(24)29)34(45)40-30(33(38)44)17-23-9-4-3-5-10-23/h3-7,9-12,15-16,20,28,30-32,39,43H,8,13-14,17-19,37H2,1-2H3,(H2,38,44)(H,40,45)(H,41,46)/t28-,30-,31-,32-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Agonist activity at human MOR expressed in CHO cell membranes after 60 mins by [35S]GTPgammaS binding assay |
J Med Chem 61: 9784-9789 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01282
BindingDB Entry DOI: 10.7270/Q2F76G8Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data