 Found 136 hits of ec50 data for polymerid = 2857
Found 136 hits of ec50 data for polymerid = 2857 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50428286

(DABRAFENIB | GSK2118436A)Show SMILES CC(C)(C)c1nc(c(s1)-c1ccnc(N)n1)-c1cccc(NS(=O)(=O)c2c(F)cccc2F)c1F Show InChI InChI=1S/C23H20F3N5O2S2/c1-23(2,3)21-30-18(19(34-21)16-10-11-28-22(27)29-16)12-6-4-9-15(17(12)26)31-35(32,33)20-13(24)7-5-8-14(20)25/h4-11,31H,1-3H3,(H2,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01354
BindingDB Entry DOI: 10.7270/Q2474FZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50038473
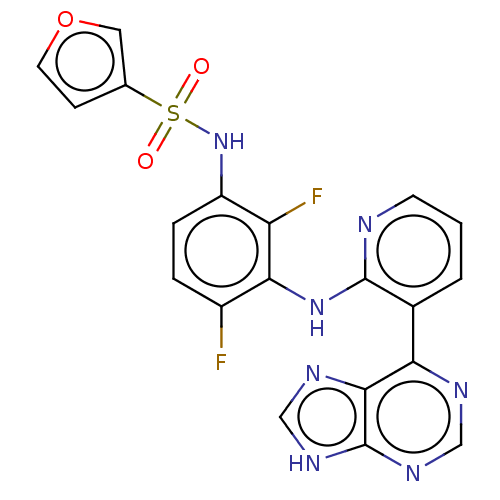
(CHEMBL3354844 | US9216981, 18)Show SMILES Fc1ccc(NS(=O)(=O)c2ccoc2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C20H13F2N7O3S/c21-13-3-4-14(29-33(30,31)11-5-7-32-8-11)15(22)17(13)28-19-12(2-1-6-23-19)16-18-20(26-9-24-16)27-10-25-18/h1-10,29H,(H,23,28)(H,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM16673

(4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamo...)Show SMILES CNC(=O)c1cc(Oc2ccc(NC(=O)Nc3ccc(Cl)c(c3)C(F)(F)F)cc2)ccn1 Show InChI InChI=1S/C21H16ClF3N4O3/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25/h2-11H,1H3,(H,26,30)(H2,28,29,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Inhibition of B-raf by cellular assay |
Proc Natl Acad Sci USA 104: 19936-41 (2007)
Article DOI: 10.1073/pnas.0707498104
BindingDB Entry DOI: 10.7270/Q24X58QS |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197686

(US9216981, 14)Show SMILES Cc1c(Cl)cccc1S(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C23H16ClF2N7O2S/c1-12-14(24)5-2-6-17(12)36(34,35)33-16-8-7-15(25)20(18(16)26)32-22-13(4-3-9-27-22)19-21-23(30-10-28-19)31-11-29-21/h2-11,33H,1H3,(H,27,32)(H,28,29,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 6 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50165861
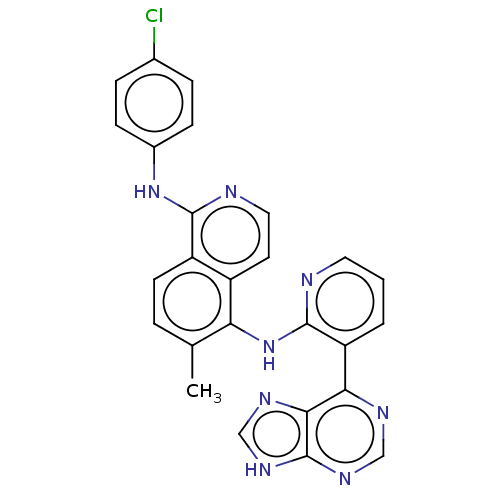
(CHEMBL1197798)Show SMILES Cc1ccc2c(Nc3ccc(Cl)cc3)nccc2c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C26H19ClN8/c1-15-4-9-19-18(10-12-29-24(19)34-17-7-5-16(27)6-8-17)21(15)35-25-20(3-2-11-28-25)22-23-26(32-13-30-22)33-14-31-23/h2-14H,1H3,(H,28,35)(H,29,34)(H,30,31,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 22 | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild type B-Raf in human MIAPaCa2 cells assessed as reduction in ERK phosphorylation preincubated for 1 hr by Western blot method |
Bioorg Med Chem 24: 2215-34 (2016)
Article DOI: 10.1016/j.bmc.2016.03.055
BindingDB Entry DOI: 10.7270/Q21C1ZS0 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50452152
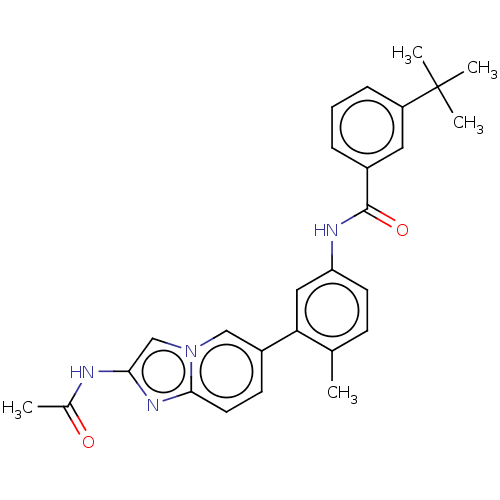
(CHEMBL4217462)Show SMILES CC(=O)Nc1cn2cc(ccc2n1)-c1cc(NC(=O)c2cccc(c2)C(C)(C)C)ccc1C Show InChI InChI=1S/C27H28N4O2/c1-17-9-11-22(29-26(33)19-7-6-8-21(13-19)27(3,4)5)14-23(17)20-10-12-25-30-24(28-18(2)32)16-31(25)15-20/h6-16H,1-5H3,(H,28,32)(H,29,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf V600E mutant in human A375 cells assessed as reduction in ERK phosphorylation |
Bioorg Med Chem Lett 27: 5221-5224 (2017)
Article DOI: 10.1016/j.bmcl.2017.10.047
BindingDB Entry DOI: 10.7270/Q2SB4892 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197676
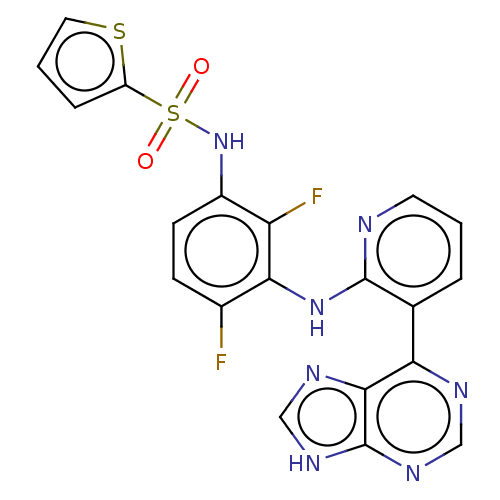
(US9216981, 4)Show SMILES Fc1ccc(NS(=O)(=O)c2cccs2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C20H13F2N7O2S2/c21-12-5-6-13(29-33(30,31)14-4-2-8-32-14)15(22)17(12)28-19-11(3-1-7-23-19)16-18-20(26-9-24-16)27-10-25-18/h1-10,29H,(H,23,28)(H,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197689
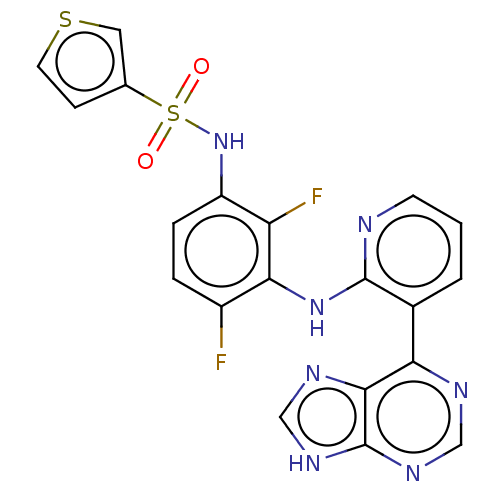
(US9216981, 17 | US9550781, 24)Show SMILES Fc1ccc(NS(=O)(=O)c2ccsc2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C20H13F2N7O2S2/c21-13-3-4-14(29-33(30,31)11-5-7-32-8-11)15(22)17(13)28-19-12(2-1-6-23-19)16-18-20(26-9-24-16)27-10-25-18/h1-10,29H,(H,23,28)(H,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 40 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026679
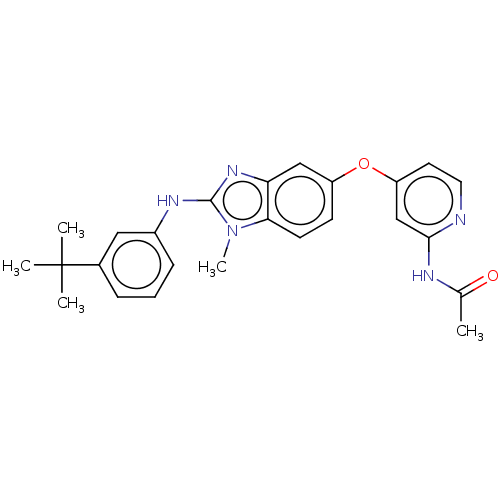
(CHEMBL3335371)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cccc(c4)C(C)(C)C)nc3c2)ccn1 Show InChI InChI=1S/C25H27N5O2/c1-16(31)27-23-15-20(11-12-26-23)32-19-9-10-22-21(14-19)29-24(30(22)5)28-18-8-6-7-17(13-18)25(2,3)4/h6-15H,1-5H3,(H,28,29)(H,26,27,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 40 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026676

(CHEMBL3335380)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4ccc(cc4)C(F)(F)F)nc3c2)ccn1 Show InChI InChI=1S/C22H18F3N5O2/c1-13(31)27-20-12-17(9-10-26-20)32-16-7-8-19-18(11-16)29-21(30(19)2)28-15-5-3-14(4-6-15)22(23,24)25/h3-12H,1-2H3,(H,28,29)(H,26,27,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026685

(CHEMBL3335373)Show SMILES CC(C)c1cccc(Nc2nc3cc(Oc4ccnc(NC(C)=O)c4)ccc3n2C)c1 Show InChI InChI=1S/C24H25N5O2/c1-15(2)17-6-5-7-18(12-17)27-24-28-21-13-19(8-9-22(21)29(24)4)31-20-10-11-25-23(14-20)26-16(3)30/h5-15H,1-4H3,(H,27,28)(H,25,26,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026684
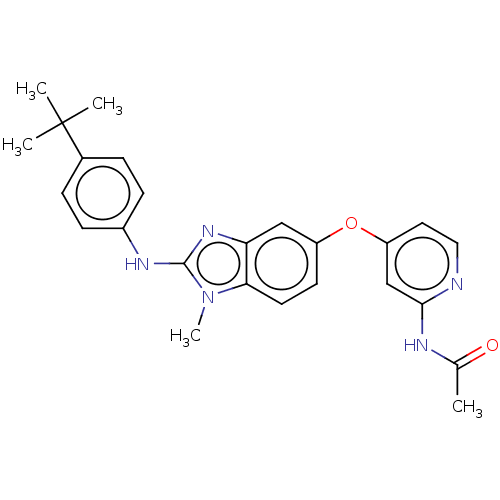
(CHEMBL3335376)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4ccc(cc4)C(C)(C)C)nc3c2)ccn1 Show InChI InChI=1S/C25H27N5O2/c1-16(31)27-23-15-20(12-13-26-23)32-19-10-11-22-21(14-19)29-24(30(22)5)28-18-8-6-17(7-9-18)25(2,3)4/h6-15H,1-5H3,(H,28,29)(H,26,27,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197694

(US9216981, 23)Show SMILES Cc1ccc(o1)S(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C21H15F2N7O3S/c1-11-4-7-15(33-11)34(31,32)30-14-6-5-13(22)18(16(14)23)29-20-12(3-2-8-24-20)17-19-21(27-9-25-17)28-10-26-19/h2-10,30H,1H3,(H,24,29)(H,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 60 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197701
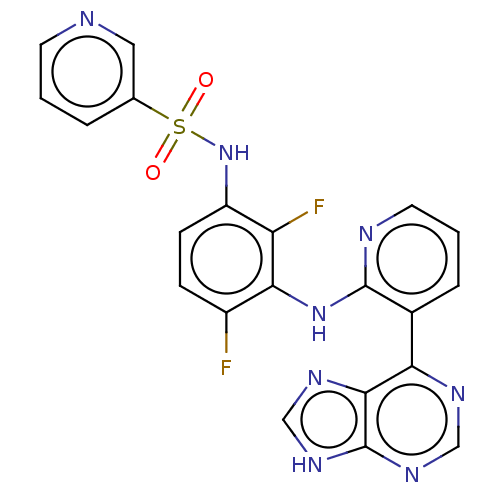
(US9216981, 30)Show SMILES Fc1ccc(NS(=O)(=O)c2cccnc2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C21H14F2N8O2S/c22-14-5-6-15(31-34(32,33)12-3-1-7-24-9-12)16(23)18(14)30-20-13(4-2-8-25-20)17-19-21(28-10-26-17)29-11-27-19/h1-11,31H,(H,25,30)(H,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 70 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026677
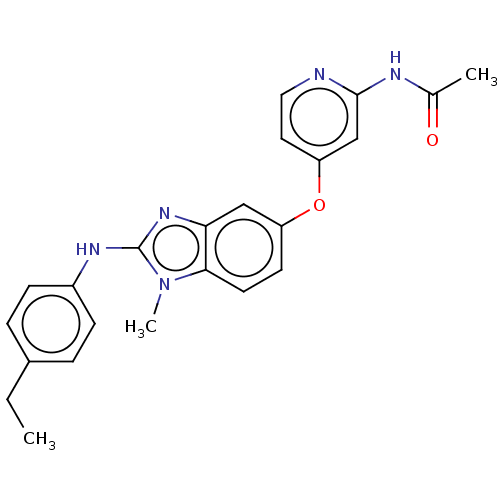
(CHEMBL3335377)Show SMILES CCc1ccc(Nc2nc3cc(Oc4ccnc(NC(C)=O)c4)ccc3n2C)cc1 Show InChI InChI=1S/C23H23N5O2/c1-4-16-5-7-17(8-6-16)26-23-27-20-13-18(9-10-21(20)28(23)3)30-19-11-12-24-22(14-19)25-15(2)29/h5-14H,4H2,1-3H3,(H,26,27)(H,24,25,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 80 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026692
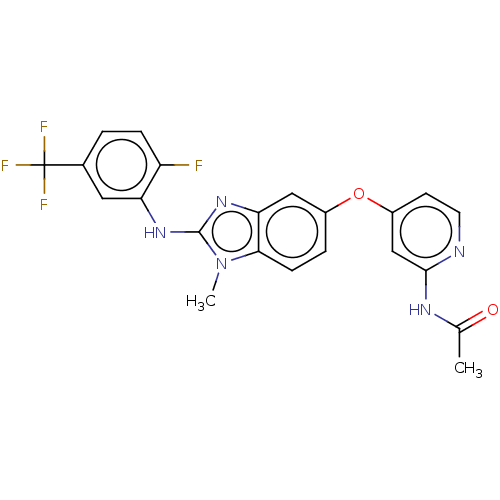
(CHEMBL3335381)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cc(ccc4F)C(F)(F)F)nc3c2)ccn1 Show InChI InChI=1S/C22H17F4N5O2/c1-12(32)28-20-11-15(7-8-27-20)33-14-4-6-19-18(10-14)30-21(31(19)2)29-17-9-13(22(24,25)26)3-5-16(17)23/h3-11H,1-2H3,(H,29,30)(H,27,28,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 90 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026693
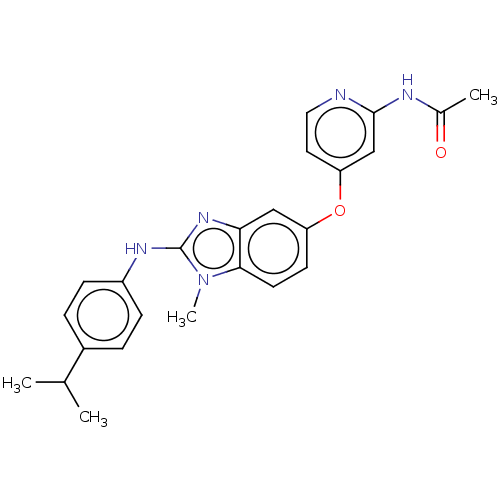
(CHEMBL3335378)Show SMILES CC(C)c1ccc(Nc2nc3cc(Oc4ccnc(NC(C)=O)c4)ccc3n2C)cc1 Show InChI InChI=1S/C24H25N5O2/c1-15(2)17-5-7-18(8-6-17)27-24-28-21-13-19(9-10-22(21)29(24)4)31-20-11-12-25-23(14-20)26-16(3)30/h5-15H,1-4H3,(H,27,28)(H,25,26,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 90 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026695
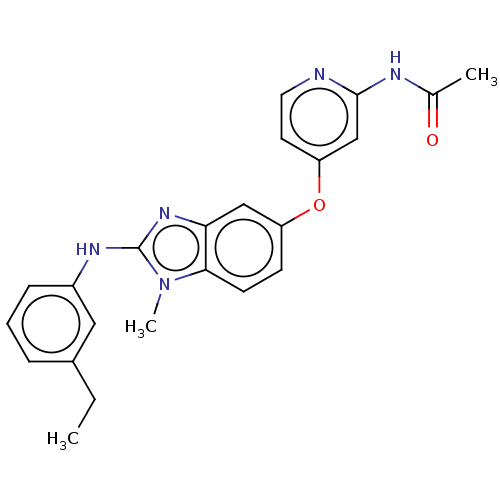
(CHEMBL3335372)Show SMILES CCc1cccc(Nc2nc3cc(Oc4ccnc(NC(C)=O)c4)ccc3n2C)c1 Show InChI InChI=1S/C23H23N5O2/c1-4-16-6-5-7-17(12-16)26-23-27-20-13-18(8-9-21(20)28(23)3)30-19-10-11-24-22(14-19)25-15(2)29/h5-14H,4H2,1-3H3,(H,26,27)(H,24,25,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026686
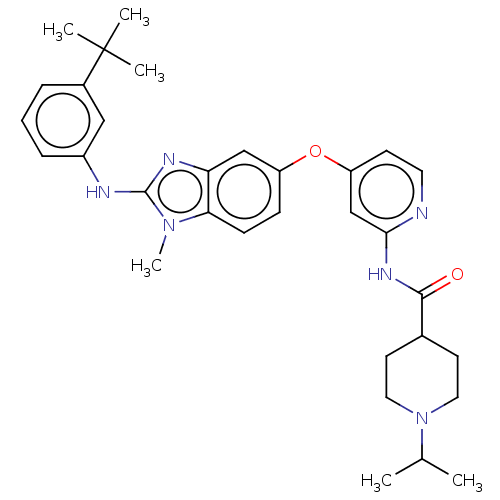
(CHEMBL3335384)Show SMILES CC(C)N1CCC(CC1)C(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cccc(c4)C(C)(C)C)nc3c2)ccn1 Show InChI InChI=1S/C32H40N6O2/c1-21(2)38-16-13-22(14-17-38)30(39)36-29-20-26(12-15-33-29)40-25-10-11-28-27(19-25)35-31(37(28)6)34-24-9-7-8-23(18-24)32(3,4)5/h7-12,15,18-22H,13-14,16-17H2,1-6H3,(H,34,35)(H,33,36,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026673

(CHEMBL3335387)Show SMILES CCN1CCN(CC(=O)Nc2cc(Oc3ccc4n(C)c(Nc5cc(ccc5F)C(F)(F)F)nc4c3)ccn2)CC1 Show InChI InChI=1S/C28H29F4N7O2/c1-3-38-10-12-39(13-11-38)17-26(40)36-25-16-20(8-9-33-25)41-19-5-7-24-23(15-19)35-27(37(24)2)34-22-14-18(28(30,31)32)4-6-21(22)29/h4-9,14-16H,3,10-13,17H2,1-2H3,(H,34,35)(H,33,36,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026683
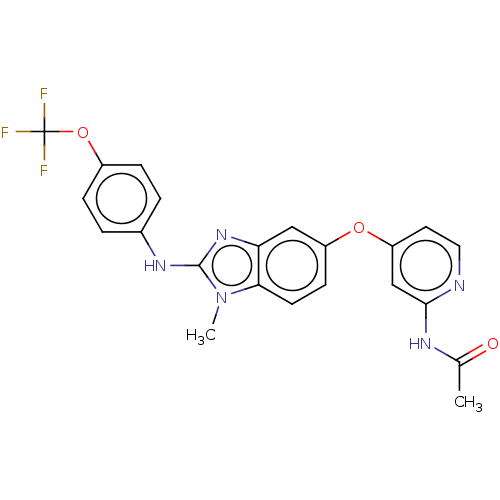
(CHEMBL3335379)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4ccc(OC(F)(F)F)cc4)nc3c2)ccn1 Show InChI InChI=1S/C22H18F3N5O3/c1-13(31)27-20-12-17(9-10-26-20)32-16-7-8-19-18(11-16)29-21(30(19)2)28-14-3-5-15(6-4-14)33-22(23,24)25/h3-12H,1-2H3,(H,28,29)(H,26,27,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026681
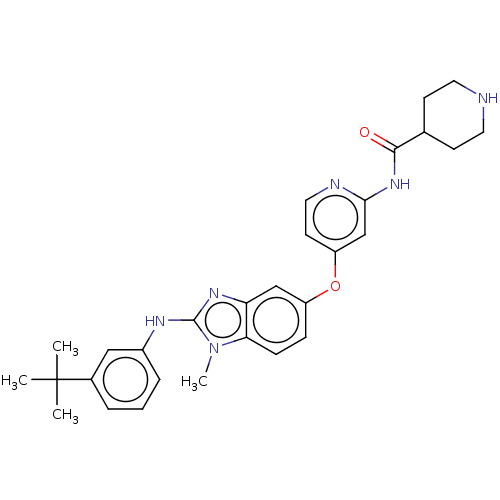
(CHEMBL3335382)Show SMILES Cn1c(Nc2cccc(c2)C(C)(C)C)nc2cc(Oc3ccnc(NC(=O)C4CCNCC4)c3)ccc12 Show InChI InChI=1S/C29H34N6O2/c1-29(2,3)20-6-5-7-21(16-20)32-28-33-24-17-22(8-9-25(24)35(28)4)37-23-12-15-31-26(18-23)34-27(36)19-10-13-30-14-11-19/h5-9,12,15-19,30H,10-11,13-14H2,1-4H3,(H,32,33)(H,31,34,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026680

(CHEMBL3335385)Show SMILES CC(C)N1CCC(CC1)C(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cc(ccc4F)C(F)(F)F)nc3c2)ccn1 Show InChI InChI=1S/C29H30F4N6O2/c1-17(2)39-12-9-18(10-13-39)27(40)37-26-16-21(8-11-34-26)41-20-5-7-25-24(15-20)36-28(38(25)3)35-23-14-19(29(31,32)33)4-6-22(23)30/h4-8,11,14-18H,9-10,12-13H2,1-3H3,(H,35,36)(H,34,37,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026675
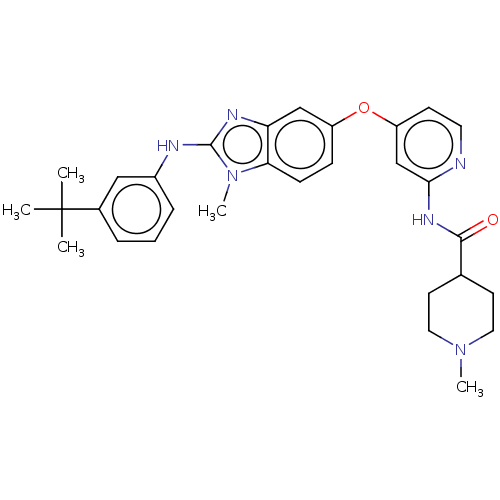
(CHEMBL3335383)Show SMILES CN1CCC(CC1)C(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cccc(c4)C(C)(C)C)nc3c2)ccn1 Show InChI InChI=1S/C30H36N6O2/c1-30(2,3)21-7-6-8-22(17-21)32-29-33-25-18-23(9-10-26(25)36(29)5)38-24-11-14-31-27(19-24)34-28(37)20-12-15-35(4)16-13-20/h6-11,14,17-20H,12-13,15-16H2,1-5H3,(H,32,33)(H,31,34,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026674

(CHEMBL3335386)Show SMILES CC(C)N1CCN(CC(=O)Nc2cc(Oc3ccc4n(C)c(Nc5cc(ccc5F)C(F)(F)F)nc4c3)ccn2)CC1 Show InChI InChI=1S/C29H31F4N7O2/c1-18(2)40-12-10-39(11-13-40)17-27(41)37-26-16-21(8-9-34-26)42-20-5-7-25-24(15-20)36-28(38(25)3)35-23-14-19(29(31,32)33)4-6-22(23)30/h4-9,14-16,18H,10-13,17H2,1-3H3,(H,35,36)(H,34,37,41) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197696

(US9216981, 25)Show SMILES Cc1cc(c(C)o1)S(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C22H17F2N7O3S/c1-11-8-16(12(2)34-11)35(32,33)31-15-6-5-14(23)19(17(15)24)30-21-13(4-3-7-25-21)18-20-22(28-9-26-18)29-10-27-20/h3-10,31H,1-2H3,(H,25,30)(H,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026678
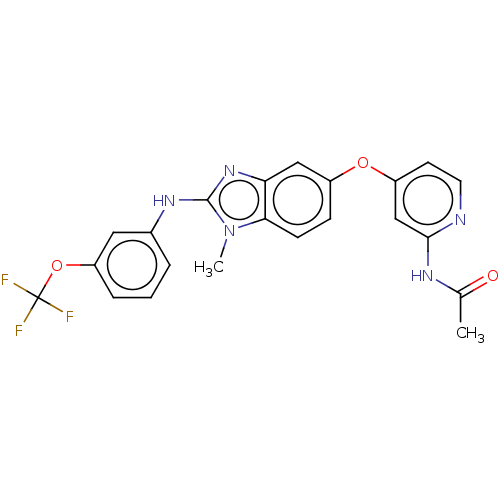
(CHEMBL3335374)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cccc(OC(F)(F)F)c4)nc3c2)ccn1 Show InChI InChI=1S/C22H18F3N5O3/c1-13(31)27-20-12-16(8-9-26-20)32-15-6-7-19-18(11-15)29-21(30(19)2)28-14-4-3-5-17(10-14)33-22(23,24)25/h3-12H,1-2H3,(H,28,29)(H,26,27,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197673
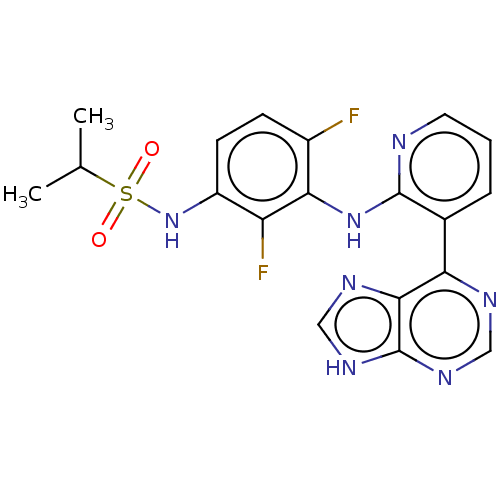
(US9216981, 1)Show SMILES CC(C)S(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C19H17F2N7O2S/c1-10(2)31(29,30)28-13-6-5-12(20)16(14(13)21)27-18-11(4-3-7-22-18)15-17-19(25-8-23-15)26-9-24-17/h3-10,28H,1-2H3,(H,22,27)(H,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 120 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50452154

(CHEMBL4205266)Show SMILES CN1CCN(Cc2ccc(cc2C(F)(F)F)C(=O)Nc2ccc(C)c(c2)-c2ccc3nc(NC(C)=O)cn3c2)CC1 Show InChI InChI=1S/C30H31F3N6O2/c1-19-4-8-24(15-25(19)22-7-9-28-36-27(34-20(2)40)18-39(28)17-22)35-29(41)21-5-6-23(26(14-21)30(31,32)33)16-38-12-10-37(3)11-13-38/h4-9,14-15,17-18H,10-13,16H2,1-3H3,(H,34,40)(H,35,41) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 130 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf V600E mutant in human A375 cells assessed as reduction in ERK phosphorylation |
Bioorg Med Chem Lett 27: 5221-5224 (2017)
Article DOI: 10.1016/j.bmcl.2017.10.047
BindingDB Entry DOI: 10.7270/Q2SB4892 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197685
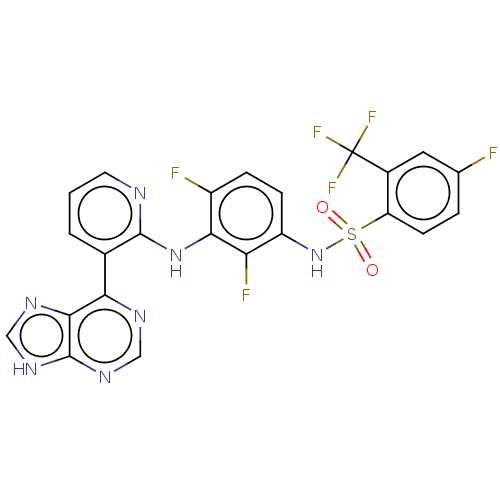
(US9216981, 13)Show SMILES Fc1ccc(c(c1)C(F)(F)F)S(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C23H13F6N7O2S/c24-11-3-6-16(13(8-11)23(27,28)29)39(37,38)36-15-5-4-14(25)19(17(15)26)35-21-12(2-1-7-30-21)18-20-22(33-9-31-18)34-10-32-20/h1-10,36H,(H,30,35)(H,31,32,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 150 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197706

(US9216981, 35 | US9550781, 15)Show SMILES Fc1ccc(NS(=O)(=O)c2ccccc2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C22H15F2N7O2S/c23-15-8-9-16(31-34(32,33)13-5-2-1-3-6-13)17(24)19(15)30-21-14(7-4-10-25-21)18-20-22(28-11-26-18)29-12-27-20/h1-12,31H,(H,25,30)(H,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 160 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50072070
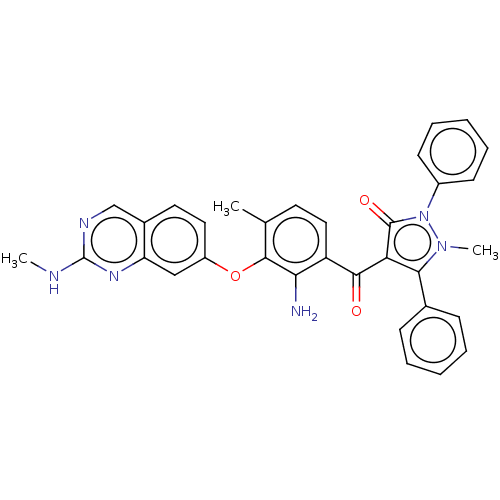
(CHEMBL3407504)Show SMILES CNc1ncc2ccc(Oc3c(C)ccc(C(=O)c4c(-c5ccccc5)n(C)n(-c5ccccc5)c4=O)c3N)cc2n1 Show InChI InChI=1S/C33H28N6O3/c1-20-14-17-25(28(34)31(20)42-24-16-15-22-19-36-33(35-2)37-26(22)18-24)30(40)27-29(21-10-6-4-7-11-21)38(3)39(32(27)41)23-12-8-5-9-13-23/h4-19H,34H2,1-3H3,(H,35,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 190 | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf in human MIAPaCa2 cells assessed as inhibition of MAPK signaling by immunoblot analysis |
J Med Chem 58: 1426-41 (2015)
Article DOI: 10.1021/jm5017494
BindingDB Entry DOI: 10.7270/Q27W6DWN |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197715

(US9216981, 44)Show SMILES Fc1ccc(NS(=O)(=O)c2ccc(Cl)s2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C20H12ClF2N7O2S2/c21-13-5-6-14(33-13)34(31,32)30-12-4-3-11(22)17(15(12)23)29-19-10(2-1-7-24-19)16-18-20(27-8-25-16)28-9-26-18/h1-9,30H,(H,24,29)(H,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50165856
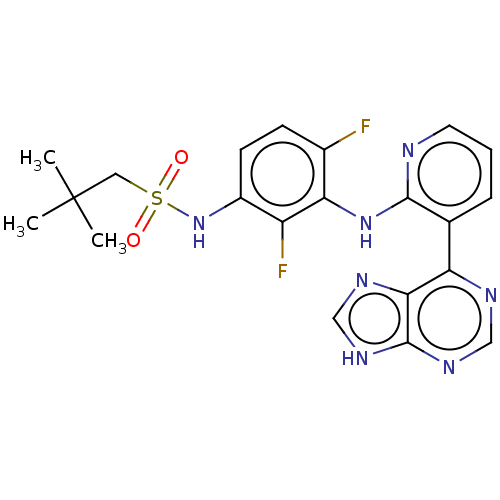
(CHEMBL3799342)Show SMILES CC(C)(C)CS(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C21H21F2N7O2S/c1-21(2,3)9-33(31,32)30-14-7-6-13(22)17(15(14)23)29-19-12(5-4-8-24-19)16-18-20(27-10-25-16)28-11-26-18/h4-8,10-11,30H,9H2,1-3H3,(H,24,29)(H,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 202 | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild type B-Raf in human MIAPaCa2 cells assessed as reduction in ERK phosphorylation preincubated for 1 hr by Western blot method |
Bioorg Med Chem 24: 2215-34 (2016)
Article DOI: 10.1016/j.bmc.2016.03.055
BindingDB Entry DOI: 10.7270/Q21C1ZS0 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197674
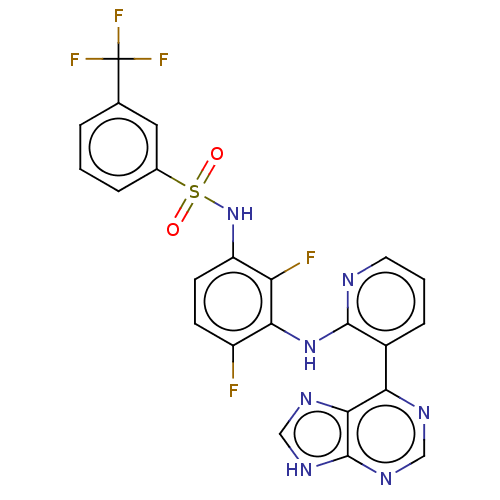
(US9216981, 2)Show SMILES Fc1ccc(NS(=O)(=O)c2cccc(c2)C(F)(F)F)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C23H14F5N7O2S/c24-15-6-7-16(35-38(36,37)13-4-1-3-12(9-13)23(26,27)28)17(25)19(15)34-21-14(5-2-8-29-21)18-20-22(32-10-30-18)33-11-31-20/h1-11,35H,(H,29,34)(H,30,31,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 230 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50452149
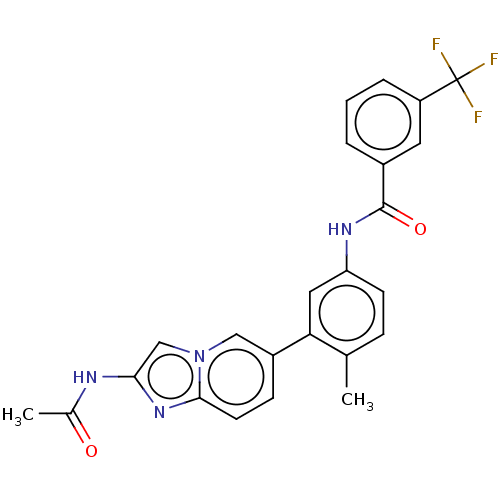
(CHEMBL4216073)Show SMILES CC(=O)Nc1cn2cc(ccc2n1)-c1cc(NC(=O)c2cccc(c2)C(F)(F)F)ccc1C Show InChI InChI=1S/C24H19F3N4O2/c1-14-6-8-19(29-23(33)16-4-3-5-18(10-16)24(25,26)27)11-20(14)17-7-9-22-30-21(28-15(2)32)13-31(22)12-17/h3-13H,1-2H3,(H,28,32)(H,29,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 230 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf V600E mutant in human A375 cells assessed as reduction in ERK phosphorylation |
Bioorg Med Chem Lett 27: 5221-5224 (2017)
Article DOI: 10.1016/j.bmcl.2017.10.047
BindingDB Entry DOI: 10.7270/Q2SB4892 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50452151
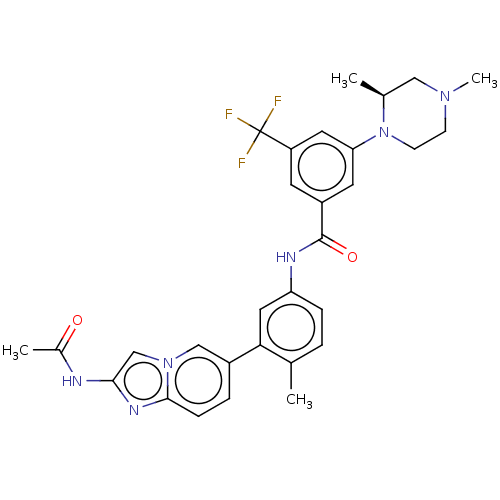
(CHEMBL4208527)Show SMILES C[C@H]1CN(C)CCN1c1cc(cc(c1)C(F)(F)F)C(=O)Nc1ccc(C)c(c1)-c1ccc2nc(NC(C)=O)cn2c1 |r| Show InChI InChI=1S/C30H31F3N6O2/c1-18-5-7-24(14-26(18)21-6-8-28-36-27(34-20(3)40)17-38(28)16-21)35-29(41)22-11-23(30(31,32)33)13-25(12-22)39-10-9-37(4)15-19(39)2/h5-8,11-14,16-17,19H,9-10,15H2,1-4H3,(H,34,40)(H,35,41)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 240 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf V600E mutant in human A375 cells assessed as reduction in ERK phosphorylation |
Bioorg Med Chem Lett 27: 5221-5224 (2017)
Article DOI: 10.1016/j.bmcl.2017.10.047
BindingDB Entry DOI: 10.7270/Q2SB4892 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197690
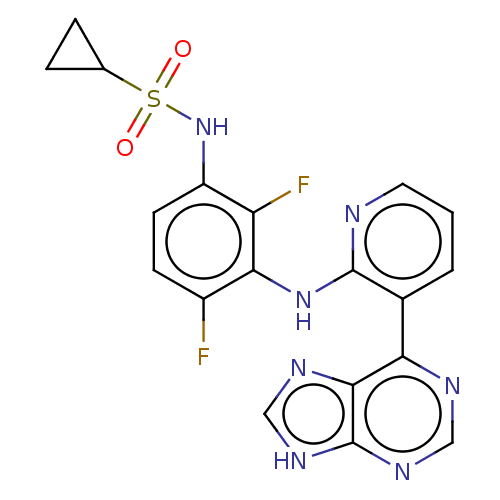
(US9216981, 19)Show SMILES Fc1ccc(NS(=O)(=O)C2CC2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C19H15F2N7O2S/c20-12-5-6-13(28-31(29,30)10-3-4-10)14(21)16(12)27-18-11(2-1-7-22-18)15-17-19(25-8-23-15)26-9-24-17/h1-2,5-10,28H,3-4H2,(H,22,27)(H,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 250 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197677
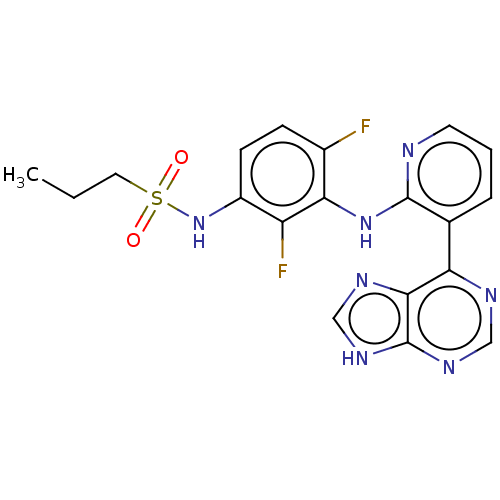
(US9216981, 5 | US9550781, 1)Show SMILES CCCS(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C19H17F2N7O2S/c1-2-8-31(29,30)28-13-6-5-12(20)16(14(13)21)27-18-11(4-3-7-22-18)15-17-19(25-9-23-15)26-10-24-17/h3-7,9-10,28H,2,8H2,1H3,(H,22,27)(H,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 310 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197697
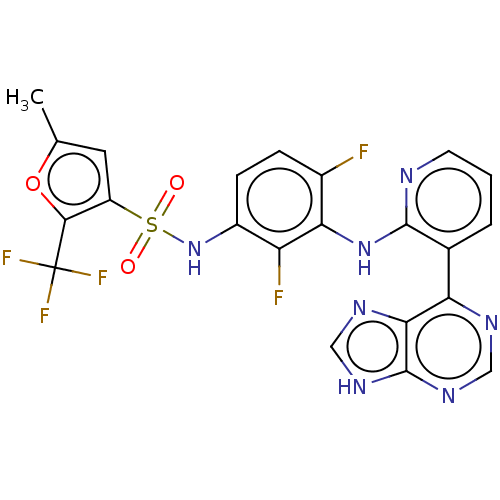
(US9216981, 26)Show SMILES Cc1cc(c(o1)C(F)(F)F)S(=O)(=O)Nc1ccc(F)c(Nc2ncccc2-c2ncnc3[nH]cnc23)c1F Show InChI InChI=1S/C22H14F5N7O3S/c1-10-7-14(19(37-10)22(25,26)27)38(35,36)34-13-5-4-12(23)17(15(13)24)33-20-11(3-2-6-28-20)16-18-21(31-8-29-16)32-9-30-18/h2-9,34H,1H3,(H,28,33)(H,29,30,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 330 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197683
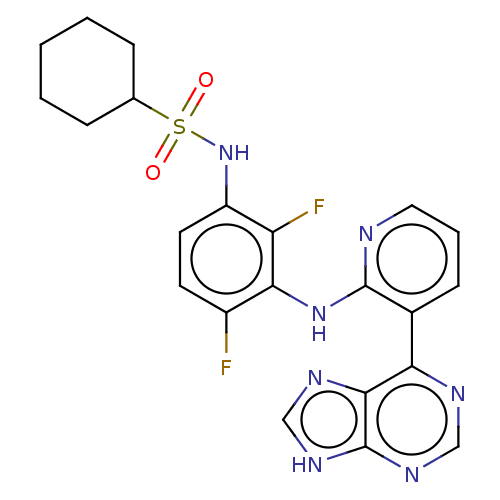
(US9216981, 11)Show SMILES Fc1ccc(NS(=O)(=O)C2CCCCC2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C22H21F2N7O2S/c23-15-8-9-16(31-34(32,33)13-5-2-1-3-6-13)17(24)19(15)30-21-14(7-4-10-25-21)18-20-22(28-11-26-18)29-12-27-20/h4,7-13,31H,1-3,5-6H2,(H,25,30)(H,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 340 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197692
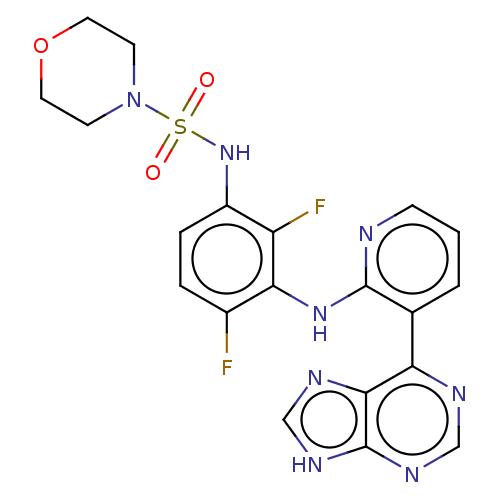
(US9216981, 21)Show SMILES Fc1ccc(NS(=O)(=O)N2CCOCC2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C20H18F2N8O3S/c21-13-3-4-14(29-34(31,32)30-6-8-33-9-7-30)15(22)17(13)28-19-12(2-1-5-23-19)16-18-20(26-10-24-16)27-11-25-18/h1-5,10-11,29H,6-9H2,(H,23,28)(H,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 360 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50026694
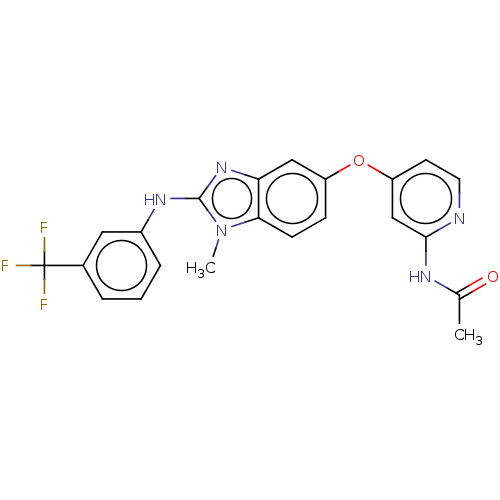
(CHEMBL3335375)Show SMILES CC(=O)Nc1cc(Oc2ccc3n(C)c(Nc4cccc(c4)C(F)(F)F)nc3c2)ccn1 Show InChI InChI=1S/C22H18F3N5O2/c1-13(31)27-20-12-17(8-9-26-20)32-16-6-7-19-18(11-16)29-21(30(19)2)28-15-5-3-4-14(10-15)22(23,24)25/h3-12H,1-2H3,(H,28,29)(H,26,27,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 400 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BRAF (unknown origin)-mediated ERK phosphorylation by cell-based assay |
ACS Med Chem Lett 5: 989-92 (2014)
Article DOI: 10.1021/ml5002272
BindingDB Entry DOI: 10.7270/Q2RN39FP |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197676

(US9216981, 4)Show SMILES Fc1ccc(NS(=O)(=O)c2cccs2)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C20H13F2N7O2S2/c21-12-5-6-13(29-33(30,31)14-4-2-8-32-14)15(22)17(12)28-19-11(3-1-7-23-19)16-18-20(26-9-24-16)27-10-25-18/h1-10,29H,(H,23,28)(H,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 402 | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild type B-Raf in human MIAPaCa2 cells assessed as reduction in ERK phosphorylation preincubated for 1 hr by Western blot method |
Bioorg Med Chem 24: 2215-34 (2016)
Article DOI: 10.1016/j.bmc.2016.03.055
BindingDB Entry DOI: 10.7270/Q21C1ZS0 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197703

(US9216981, 32)Show SMILES Fc1ccc(NS(=O)(=O)c2ccccc2Cl)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C22H14ClF2N7O2S/c23-13-5-1-2-6-16(13)35(33,34)32-15-8-7-14(24)19(17(15)25)31-21-12(4-3-9-26-21)18-20-22(29-10-27-18)30-11-28-20/h1-11,32H,(H,26,31)(H,27,28,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 440 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50452150
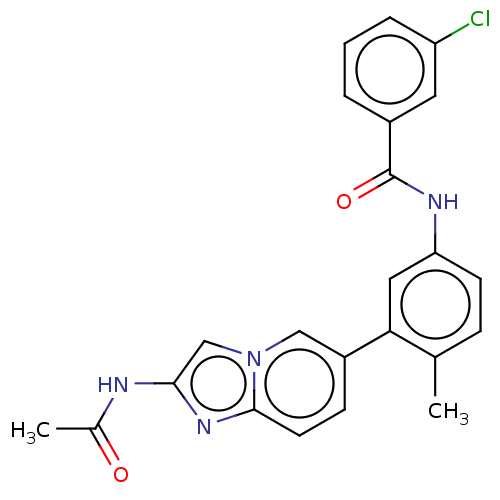
(CHEMBL4216386)Show SMILES CC(=O)Nc1cn2cc(ccc2n1)-c1cc(NC(=O)c2cccc(Cl)c2)ccc1C Show InChI InChI=1S/C23H19ClN4O2/c1-14-6-8-19(26-23(30)16-4-3-5-18(24)10-16)11-20(14)17-7-9-22-27-21(25-15(2)29)13-28(22)12-17/h3-13H,1-2H3,(H,25,29)(H,26,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 480 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf V600E mutant in human A375 cells assessed as reduction in ERK phosphorylation |
Bioorg Med Chem Lett 27: 5221-5224 (2017)
Article DOI: 10.1016/j.bmcl.2017.10.047
BindingDB Entry DOI: 10.7270/Q2SB4892 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50075576
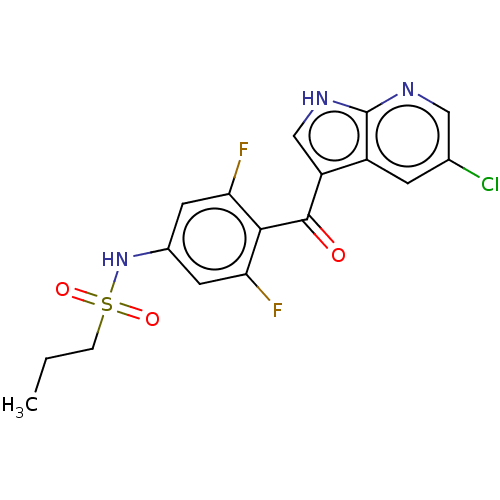
(CHEMBL3415554)Show SMILES CCCS(=O)(=O)Nc1cc(F)c(C(=O)c2c[nH]c3ncc(Cl)cc23)c(F)c1 Show InChI InChI=1S/C17H14ClF2N3O3S/c1-2-3-27(25,26)23-10-5-13(19)15(14(20)6-10)16(24)12-8-22-17-11(12)4-9(18)7-21-17/h4-8,23H,2-3H2,1H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <500 | n/a | n/a | n/a | n/a |
UMR CNRS-Universit£ d'Orl£ans 7311
Curated by ChEMBL
| Assay Description
Activation of tumorigenic wild type B-Raf in human IPC-298 cells with Ras mutation assessed as phosphorylation of ERK |
J Med Chem 58: 1818-31 (2015)
Article DOI: 10.1021/jm501667n
BindingDB Entry DOI: 10.7270/Q27M09N7 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM197705
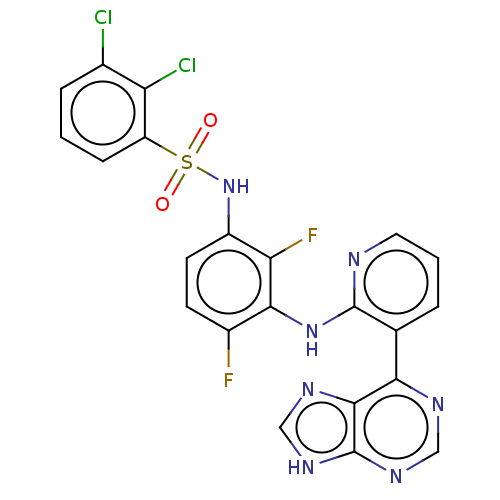
(US9216981, 34)Show SMILES Fc1ccc(NS(=O)(=O)c2cccc(Cl)c2Cl)c(F)c1Nc1ncccc1-c1ncnc2[nH]cnc12 Show InChI InChI=1S/C22H13Cl2F2N7O2S/c23-12-4-1-5-15(16(12)24)36(34,35)33-14-7-6-13(25)19(17(14)26)32-21-11(3-2-8-27-21)18-20-22(30-9-28-18)31-10-29-20/h1-10,33H,(H,27,32)(H,28,29,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | n/a | n/a | 500 | n/a | n/a | n/a | 37 |
Medpacto, Inc.
US Patent
| Assay Description
To check the B-Raf cell activity inhibitory capability of the compounds
of the present invention, the following experiments were conducted in
A375P... |
US Patent US9216981 (2015)
BindingDB Entry DOI: 10.7270/Q2BC3XCR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50428286

(DABRAFENIB | GSK2118436A)Show SMILES CC(C)(C)c1nc(c(s1)-c1ccnc(N)n1)-c1cccc(NS(=O)(=O)c2c(F)cccc2F)c1F Show InChI InChI=1S/C23H20F3N5O2S2/c1-23(2,3)21-30-18(19(34-21)16-10-11-28-22(27)29-16)12-6-4-9-15(17(12)26)31-35(32,33)20-13(24)7-5-8-14(20)25/h4-11,31H,1-3H3,(H2,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 529 | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild type B-Raf in human MIAPaCa2 cells assessed as reduction in ERK phosphorylation preincubated for 1 hr by Western blot method |
Bioorg Med Chem 24: 2215-34 (2016)
Article DOI: 10.1016/j.bmc.2016.03.055
BindingDB Entry DOI: 10.7270/Q21C1ZS0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50452153

(CHEMBL4211935)Show SMILES CC(=O)Nc1cn2cc(ccc2n1)-c1cc(NC(=O)c2ccccc2)ccc1C Show InChI InChI=1S/C23H20N4O2/c1-15-8-10-19(25-23(29)17-6-4-3-5-7-17)12-20(15)18-9-11-22-26-21(24-16(2)28)14-27(22)13-18/h3-14H,1-2H3,(H,24,28)(H,25,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 540 | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of B-Raf V600E mutant in human A375 cells assessed as reduction in ERK phosphorylation |
Bioorg Med Chem Lett 27: 5221-5224 (2017)
Article DOI: 10.1016/j.bmcl.2017.10.047
BindingDB Entry DOI: 10.7270/Q2SB4892 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data