 Found 126 hits of ic50 data for polymerid = 3035
Found 126 hits of ic50 data for polymerid = 3035 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26349

((2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-amino-3-(4-hydroxyp...)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |wU:83.86,72.75,12.20,29.34,37.50,51.58,59.62,wD:4.4,23.26,(23.93,-21.81,;22.46,-22.29,;22.15,-23.79,;21.33,-21.26,;21.65,-19.76,;20.51,-18.74,;19.04,-19.2,;18.73,-20.72,;17.9,-18.17,;16.44,-18.65,;15.29,-17.62,;15.61,-16.12,;13.83,-18.1,;12.69,-17.06,;13.01,-15.57,;11.86,-14.54,;12.18,-13.03,;13.65,-12.55,;14.79,-13.59,;14.47,-15.08,;13.51,-19.6,;12.05,-20.08,;10.9,-19.05,;11.73,-21.57,;12.87,-22.61,;14.34,-22.12,;10.27,-22.05,;9.94,-23.55,;11.09,-24.58,;8.49,-24.03,;7.34,-23,;7.67,-21.49,;6.52,-20.46,;9.12,-21.02,;8.16,-25.53,;6.7,-26.01,;5.56,-24.98,;6.38,-27.51,;4.92,-27.99,;4.6,-29.49,;5.63,-30.63,;4.86,-31.97,;3.35,-31.65,;2.11,-32.56,;.71,-31.92,;.55,-30.4,;1.79,-29.49,;3.2,-30.12,;7.53,-28.54,;8.98,-28.07,;9.31,-26.56,;10.13,-29.1,;9.81,-30.6,;8.35,-31.08,;8.02,-32.58,;7.2,-30.05,;11.59,-28.62,;12.74,-29.65,;12.42,-31.16,;14.21,-29.18,;14.52,-27.67,;15.34,-30.2,;16.8,-29.73,;17.12,-28.22,;18.58,-27.75,;19.72,-28.77,;21.19,-28.3,;19.41,-30.28,;17.94,-30.75,;23.11,-19.28,;23.42,-17.77,;24.24,-20.31,;25.71,-19.83,;26.03,-18.32,;27.49,-17.85,;27.81,-16.34,;29.28,-15.87,;29.59,-14.36,;28.45,-13.34,;31.06,-13.9,;26.85,-20.86,;26.53,-22.37,;28.32,-20.39,;29.46,-21.42,;30.93,-20.94,;32.07,-21.97,;33.53,-21.49,;34.68,-22.52,;34.35,-24.03,;32.89,-24.5,;31.75,-23.47,;29.14,-22.92,;27.68,-23.4,;30.28,-23.95,)| Show InChI InChI=1S/C63H83N17O14/c1-34(2)24-45(58(90)74-43(18-11-23-70-63(68)69)57(89)75-44(54(67)86)26-35-12-5-3-6-13-35)73-53(85)32-72-56(88)46(27-36-14-7-4-8-15-36)77-62(94)50(33-81)80-61(93)49(30-52(66)84)79-59(91)47(28-38-31-71-42-17-10-9-16-40(38)42)78-60(92)48(29-51(65)83)76-55(87)41(64)25-37-19-21-39(82)22-20-37/h3-10,12-17,19-22,31,34,41,43-50,71,81-82H,11,18,23-30,32-33,64H2,1-2H3,(H2,65,83)(H2,66,84)(H2,67,86)(H,72,88)(H,73,85)(H,74,90)(H,75,89)(H,76,87)(H,77,94)(H,78,92)(H,79,91)(H,80,93)(H4,68,69,70)/t41-,43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0820 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392401
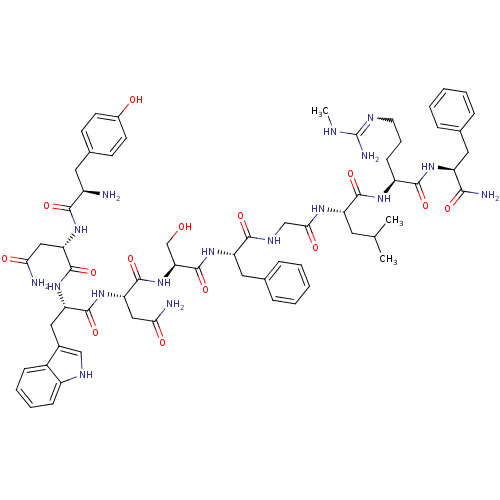
(CHEMBL2151642)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C64H85N17O14/c1-35(2)25-46(59(91)75-44(19-12-24-71-64(69)70-3)58(90)76-45(55(68)87)27-36-13-6-4-7-14-36)74-54(86)33-73-57(89)47(28-37-15-8-5-9-16-37)78-63(95)51(34-82)81-62(94)50(31-53(67)85)80-60(92)48(29-39-32-72-43-18-11-10-17-41(39)43)79-61(93)49(30-52(66)84)77-56(88)42(65)26-38-20-22-40(83)23-21-38/h4-11,13-18,20-23,32,35,42,44-51,72,82-83H,12,19,24-31,33-34,65H2,1-3H3,(H2,66,84)(H2,67,85)(H2,68,87)(H,73,89)(H,74,86)(H,75,91)(H,76,90)(H,77,88)(H,78,95)(H,79,93)(H,80,92)(H,81,94)(H3,69,70,71)/t42-,44+,45+,46+,47+,48+,49+,50+,51+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26349

((2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-amino-3-(4-hydroxyp...)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |wU:83.86,72.75,12.20,29.34,37.50,51.58,59.62,wD:4.4,23.26,(23.93,-21.81,;22.46,-22.29,;22.15,-23.79,;21.33,-21.26,;21.65,-19.76,;20.51,-18.74,;19.04,-19.2,;18.73,-20.72,;17.9,-18.17,;16.44,-18.65,;15.29,-17.62,;15.61,-16.12,;13.83,-18.1,;12.69,-17.06,;13.01,-15.57,;11.86,-14.54,;12.18,-13.03,;13.65,-12.55,;14.79,-13.59,;14.47,-15.08,;13.51,-19.6,;12.05,-20.08,;10.9,-19.05,;11.73,-21.57,;12.87,-22.61,;14.34,-22.12,;10.27,-22.05,;9.94,-23.55,;11.09,-24.58,;8.49,-24.03,;7.34,-23,;7.67,-21.49,;6.52,-20.46,;9.12,-21.02,;8.16,-25.53,;6.7,-26.01,;5.56,-24.98,;6.38,-27.51,;4.92,-27.99,;4.6,-29.49,;5.63,-30.63,;4.86,-31.97,;3.35,-31.65,;2.11,-32.56,;.71,-31.92,;.55,-30.4,;1.79,-29.49,;3.2,-30.12,;7.53,-28.54,;8.98,-28.07,;9.31,-26.56,;10.13,-29.1,;9.81,-30.6,;8.35,-31.08,;8.02,-32.58,;7.2,-30.05,;11.59,-28.62,;12.74,-29.65,;12.42,-31.16,;14.21,-29.18,;14.52,-27.67,;15.34,-30.2,;16.8,-29.73,;17.12,-28.22,;18.58,-27.75,;19.72,-28.77,;21.19,-28.3,;19.41,-30.28,;17.94,-30.75,;23.11,-19.28,;23.42,-17.77,;24.24,-20.31,;25.71,-19.83,;26.03,-18.32,;27.49,-17.85,;27.81,-16.34,;29.28,-15.87,;29.59,-14.36,;28.45,-13.34,;31.06,-13.9,;26.85,-20.86,;26.53,-22.37,;28.32,-20.39,;29.46,-21.42,;30.93,-20.94,;32.07,-21.97,;33.53,-21.49,;34.68,-22.52,;34.35,-24.03,;32.89,-24.5,;31.75,-23.47,;29.14,-22.92,;27.68,-23.4,;30.28,-23.95,)| Show InChI InChI=1S/C63H83N17O14/c1-34(2)24-45(58(90)74-43(18-11-23-70-63(68)69)57(89)75-44(54(67)86)26-35-12-5-3-6-13-35)73-53(85)32-72-56(88)46(27-36-14-7-4-8-15-36)77-62(94)50(33-81)80-61(93)49(30-52(66)84)79-59(91)47(28-38-31-71-42-17-10-9-16-40(38)42)78-60(92)48(29-51(65)83)76-55(87)41(64)25-37-19-21-39(82)22-20-37/h3-10,12-17,19-22,31,34,41,43-50,71,81-82H,11,18,23-30,32-33,64H2,1-2H3,(H2,65,83)(H2,66,84)(H2,67,86)(H,72,88)(H,73,85)(H,74,90)(H,75,89)(H,76,87)(H,77,94)(H,78,92)(H,79,91)(H,80,93)(H4,68,69,70)/t41-,43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26349

((2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-amino-3-(4-hydroxyp...)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |wU:83.86,72.75,12.20,29.34,37.50,51.58,59.62,wD:4.4,23.26,(23.93,-21.81,;22.46,-22.29,;22.15,-23.79,;21.33,-21.26,;21.65,-19.76,;20.51,-18.74,;19.04,-19.2,;18.73,-20.72,;17.9,-18.17,;16.44,-18.65,;15.29,-17.62,;15.61,-16.12,;13.83,-18.1,;12.69,-17.06,;13.01,-15.57,;11.86,-14.54,;12.18,-13.03,;13.65,-12.55,;14.79,-13.59,;14.47,-15.08,;13.51,-19.6,;12.05,-20.08,;10.9,-19.05,;11.73,-21.57,;12.87,-22.61,;14.34,-22.12,;10.27,-22.05,;9.94,-23.55,;11.09,-24.58,;8.49,-24.03,;7.34,-23,;7.67,-21.49,;6.52,-20.46,;9.12,-21.02,;8.16,-25.53,;6.7,-26.01,;5.56,-24.98,;6.38,-27.51,;4.92,-27.99,;4.6,-29.49,;5.63,-30.63,;4.86,-31.97,;3.35,-31.65,;2.11,-32.56,;.71,-31.92,;.55,-30.4,;1.79,-29.49,;3.2,-30.12,;7.53,-28.54,;8.98,-28.07,;9.31,-26.56,;10.13,-29.1,;9.81,-30.6,;8.35,-31.08,;8.02,-32.58,;7.2,-30.05,;11.59,-28.62,;12.74,-29.65,;12.42,-31.16,;14.21,-29.18,;14.52,-27.67,;15.34,-30.2,;16.8,-29.73,;17.12,-28.22,;18.58,-27.75,;19.72,-28.77,;21.19,-28.3,;19.41,-30.28,;17.94,-30.75,;23.11,-19.28,;23.42,-17.77,;24.24,-20.31,;25.71,-19.83,;26.03,-18.32,;27.49,-17.85,;27.81,-16.34,;29.28,-15.87,;29.59,-14.36,;28.45,-13.34,;31.06,-13.9,;26.85,-20.86,;26.53,-22.37,;28.32,-20.39,;29.46,-21.42,;30.93,-20.94,;32.07,-21.97,;33.53,-21.49,;34.68,-22.52,;34.35,-24.03,;32.89,-24.5,;31.75,-23.47,;29.14,-22.92,;27.68,-23.4,;30.28,-23.95,)| Show InChI InChI=1S/C63H83N17O14/c1-34(2)24-45(58(90)74-43(18-11-23-70-63(68)69)57(89)75-44(54(67)86)26-35-12-5-3-6-13-35)73-53(85)32-72-56(88)46(27-36-14-7-4-8-15-36)77-62(94)50(33-81)80-61(93)49(30-52(66)84)79-59(91)47(28-38-31-71-42-17-10-9-16-40(38)42)78-60(92)48(29-51(65)83)76-55(87)41(64)25-37-19-21-39(82)22-20-37/h3-10,12-17,19-22,31,34,41,43-50,71,81-82H,11,18,23-30,32-33,64H2,1-2H3,(H2,65,83)(H2,66,84)(H2,67,86)(H,72,88)(H,73,85)(H,74,90)(H,75,89)(H,76,87)(H,77,94)(H,78,92)(H,79,91)(H,80,93)(H4,68,69,70)/t41-,43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]kisspeptin-15 from GPR54 |
ACS Med Chem Lett 2: 53-57 (2011)
Article DOI: 10.1021/ml1002053
BindingDB Entry DOI: 10.7270/Q2K07586 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26339
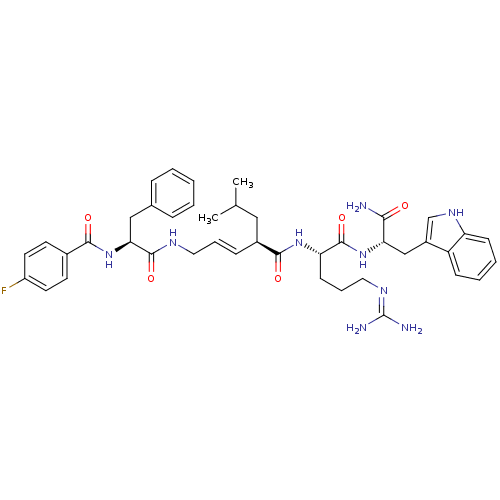
((2R,3E)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carba...)Show SMILES CC(C)C[C@H](\C=C\CNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:32.33,wD:11.19,4.4,43.44,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C42H52FN9O5/c1-26(2)22-29(12-8-20-47-40(56)36(23-27-10-4-3-5-11-27)52-38(54)28-16-18-31(43)19-17-28)39(55)50-34(15-9-21-48-42(45)46)41(57)51-35(37(44)53)24-30-25-49-33-14-7-6-13-32(30)33/h3-8,10-14,16-19,25-26,29,34-36,49H,9,15,20-24H2,1-2H3,(H2,44,53)(H,47,56)(H,50,55)(H,51,57)(H,52,54)(H4,45,46,48)/b12-8+/t29-,34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26339

((2R,3E)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carba...)Show SMILES CC(C)C[C@H](\C=C\CNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:32.33,wD:11.19,4.4,43.44,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C42H52FN9O5/c1-26(2)22-29(12-8-20-47-40(56)36(23-27-10-4-3-5-11-27)52-38(54)28-16-18-31(43)19-17-28)39(55)50-34(15-9-21-48-42(45)46)41(57)51-35(37(44)53)24-30-25-49-33-14-7-6-13-32(30)33/h3-8,10-14,16-19,25-26,29,34-36,49H,9,15,20-24H2,1-2H3,(H2,44,53)(H,47,56)(H,50,55)(H,51,57)(H,52,54)(H4,45,46,48)/b12-8+/t29-,34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]kisspeptin-15 from GPR54 |
ACS Med Chem Lett 2: 53-57 (2011)
Article DOI: 10.1021/ml1002053
BindingDB Entry DOI: 10.7270/Q2K07586 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM107307
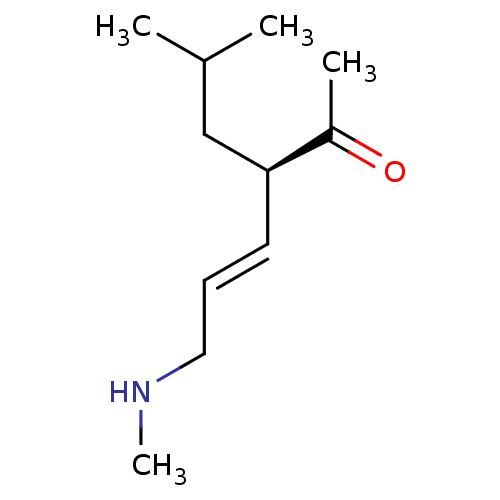
(US8592379, 19)Show InChI InChI=1S/C11H21NO/c1-9(2)8-11(10(3)13)6-5-7-12-4/h5-6,9,11-12H,7-8H2,1-4H3/b6-5+/t11-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University; Takeda Pharmaceutical Company Limited
US Patent
| Assay Description
Binding inhibition assay using human GPR54. |
US Patent US8592379 (2013)
BindingDB Entry DOI: 10.7270/Q2N0156N |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392412
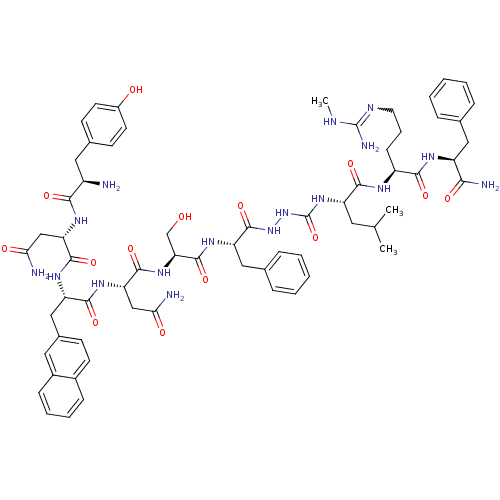
(CHEMBL2151653)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1ccc2ccccc2c1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C65H85N17O14/c1-36(2)27-47(58(90)73-45(19-12-26-72-64(70)71-3)57(89)74-46(55(69)87)30-37-13-6-4-7-14-37)80-65(96)82-81-63(95)49(31-38-15-8-5-9-16-38)77-62(94)52(35-83)79-61(93)51(34-54(68)86)78-59(91)48(32-40-20-23-41-17-10-11-18-42(41)28-40)76-60(92)50(33-53(67)85)75-56(88)44(66)29-39-21-24-43(84)25-22-39/h4-11,13-18,20-25,28,36,44-52,83-84H,12,19,26-27,29-35,66H2,1-3H3,(H2,67,85)(H2,68,86)(H2,69,87)(H,73,90)(H,74,89)(H,75,88)(H,76,92)(H,77,94)(H,78,91)(H,79,93)(H,81,95)(H3,70,71,72)(H2,80,82,96)/t44-,45+,46+,47+,48+,49+,50+,51+,52+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392405
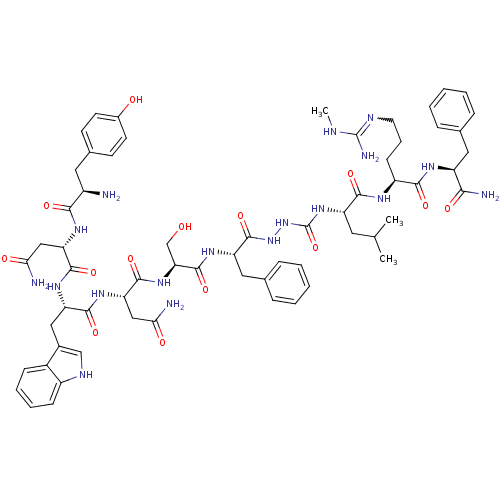
(CHEMBL2151646)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C63H84N18O14/c1-34(2)25-45(56(89)72-43(19-12-24-70-62(68)69-3)55(88)73-44(53(67)86)27-35-13-6-4-7-14-35)79-63(95)81-80-61(94)46(28-36-15-8-5-9-16-36)75-60(93)50(33-82)78-59(92)49(31-52(66)85)77-57(90)47(29-38-32-71-42-18-11-10-17-40(38)42)76-58(91)48(30-51(65)84)74-54(87)41(64)26-37-20-22-39(83)23-21-37/h4-11,13-18,20-23,32,34,41,43-50,71,82-83H,12,19,24-31,33,64H2,1-3H3,(H2,65,84)(H2,66,85)(H2,67,86)(H,72,89)(H,73,88)(H,74,87)(H,75,93)(H,76,91)(H,77,90)(H,78,92)(H,80,94)(H3,68,69,70)(H2,79,81,95)/t41-,43+,44+,45+,46+,47+,48+,49+,50+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392413
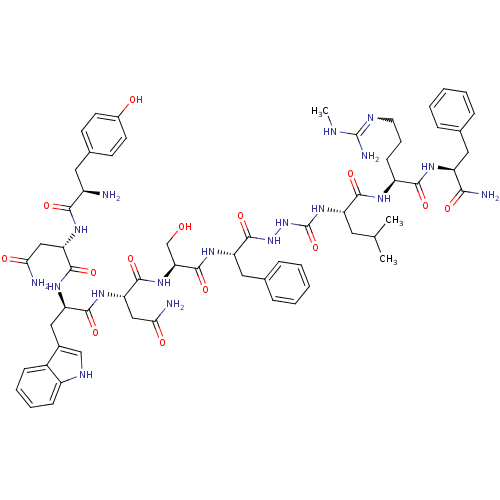
(CHEMBL2151654)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C63H84N18O14/c1-34(2)25-45(56(89)72-43(19-12-24-70-62(68)69-3)55(88)73-44(53(67)86)27-35-13-6-4-7-14-35)79-63(95)81-80-61(94)46(28-36-15-8-5-9-16-36)75-60(93)50(33-82)78-59(92)49(31-52(66)85)77-57(90)47(29-38-32-71-42-18-11-10-17-40(38)42)76-58(91)48(30-51(65)84)74-54(87)41(64)26-37-20-22-39(83)23-21-37/h4-11,13-18,20-23,32,34,41,43-50,71,82-83H,12,19,24-31,33,64H2,1-3H3,(H2,65,84)(H2,66,85)(H2,67,86)(H,72,89)(H,73,88)(H,74,87)(H,75,93)(H,76,91)(H,77,90)(H,78,92)(H,80,94)(H3,68,69,70)(H2,79,81,95)/t41-,43+,44+,45+,46+,47-,48+,49+,50+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392409
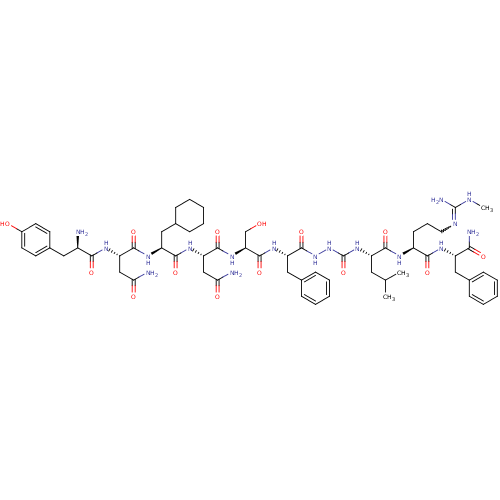
(CHEMBL2151650)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C61H89N17O14/c1-34(2)26-43(54(86)69-41(20-13-25-68-60(66)67-3)53(85)70-42(51(65)83)28-35-14-7-4-8-15-35)76-61(92)78-77-59(91)45(30-37-18-11-6-12-19-37)73-58(90)48(33-79)75-57(89)47(32-50(64)82)74-55(87)44(29-36-16-9-5-10-17-36)72-56(88)46(31-49(63)81)71-52(84)40(62)27-38-21-23-39(80)24-22-38/h4,6-8,11-12,14-15,18-19,21-24,34,36,40-48,79-80H,5,9-10,13,16-17,20,25-33,62H2,1-3H3,(H2,63,81)(H2,64,82)(H2,65,83)(H,69,86)(H,70,85)(H,71,84)(H,72,88)(H,73,90)(H,74,87)(H,75,89)(H,77,91)(H3,66,67,68)(H2,76,78,92)/t40-,41+,42+,43+,44+,45+,46+,47+,48+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26347
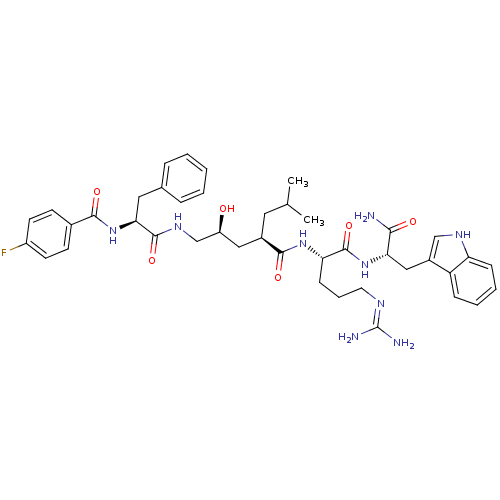
((2R,4S)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carba...)Show SMILES CC(C)C[C@H](C[C@H](O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:33.34,wD:12.20,4.31,44.45,6.6,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;6.76,.89,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C42H54FN9O6/c1-25(2)19-28(21-31(53)24-49-40(57)36(20-26-9-4-3-5-10-26)52-38(55)27-14-16-30(43)17-15-27)39(56)50-34(13-8-18-47-42(45)46)41(58)51-35(37(44)54)22-29-23-48-33-12-7-6-11-32(29)33/h3-7,9-12,14-17,23,25,28,31,34-36,48,53H,8,13,18-22,24H2,1-2H3,(H2,44,54)(H,49,57)(H,50,56)(H,51,58)(H,52,55)(H4,45,46,47)/t28-,31+,34+,35+,36+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.240 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM107314
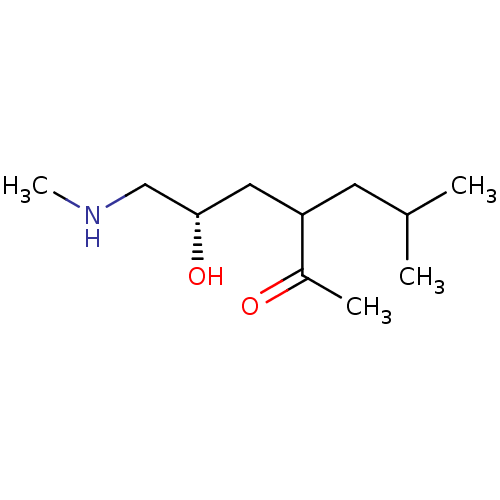
(US8592379, 36 | US8592379, 37)Show InChI InChI=1S/C11H23NO2/c1-8(2)5-10(9(3)13)6-11(14)7-12-4/h8,10-12,14H,5-7H2,1-4H3/t10?,11-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University; Takeda Pharmaceutical Company Limited
US Patent
| Assay Description
Binding inhibition assay using human GPR54. |
US Patent US8592379 (2013)
BindingDB Entry DOI: 10.7270/Q2N0156N |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392414

(CHEMBL2151655)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](Cc1ccncc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C60H82N18O14/c1-33(2)25-42(53(86)69-40(15-10-22-68-59(65)66-3)52(85)70-41(50(64)83)27-34-11-6-4-7-12-34)76-60(92)78-77-58(91)44(28-35-13-8-5-9-14-35)73-57(90)47(32-79)75-56(89)46(31-49(63)82)74-54(87)43(29-37-20-23-67-24-21-37)72-55(88)45(30-48(62)81)71-51(84)39(61)26-36-16-18-38(80)19-17-36/h4-9,11-14,16-21,23-24,33,39-47,79-80H,10,15,22,25-32,61H2,1-3H3,(H2,62,81)(H2,63,82)(H2,64,83)(H,69,86)(H,70,85)(H,71,84)(H,72,88)(H,73,90)(H,74,87)(H,75,89)(H,77,91)(H3,65,66,68)(H2,76,78,92)/t39-,40+,41+,42+,43-,44+,45+,46+,47+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392411
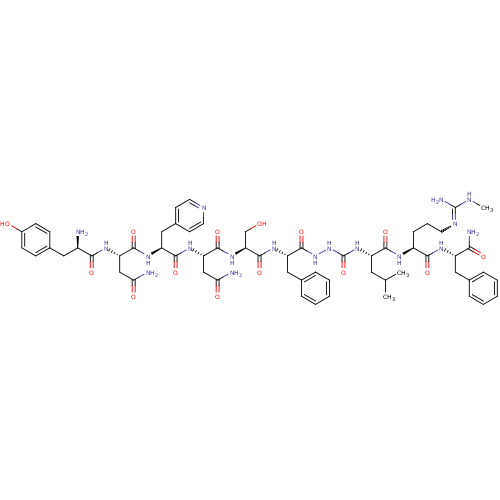
(CHEMBL2151652)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1ccncc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C60H82N18O14/c1-33(2)25-42(53(86)69-40(15-10-22-68-59(65)66-3)52(85)70-41(50(64)83)27-34-11-6-4-7-12-34)76-60(92)78-77-58(91)44(28-35-13-8-5-9-14-35)73-57(90)47(32-79)75-56(89)46(31-49(63)82)74-54(87)43(29-37-20-23-67-24-21-37)72-55(88)45(30-48(62)81)71-51(84)39(61)26-36-16-18-38(80)19-17-36/h4-9,11-14,16-21,23-24,33,39-47,79-80H,10,15,22,25-32,61H2,1-3H3,(H2,62,81)(H2,63,82)(H2,64,83)(H,69,86)(H,70,85)(H,71,84)(H,72,88)(H,73,90)(H,74,87)(H,75,89)(H,77,91)(H3,65,66,68)(H2,76,78,92)/t39-,40+,41+,42+,43+,44+,45+,46+,47+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392408
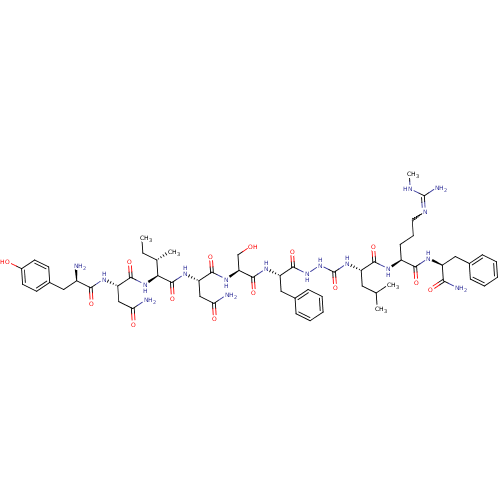
(CHEMBL2151649)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc1ccccc1)C(=O)NNC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)NC)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:70.71| Show InChI InChI=1S/C58H85N17O14/c1-6-32(4)47(73-53(85)43(29-46(61)79)68-49(81)37(59)25-35-19-21-36(77)22-20-35)56(88)70-42(28-45(60)78)52(84)71-44(30-76)54(86)69-41(27-34-16-11-8-12-17-34)55(87)74-75-58(89)72-40(24-31(2)3)51(83)66-38(18-13-23-65-57(63)64-5)50(82)67-39(48(62)80)26-33-14-9-7-10-15-33/h7-12,14-17,19-22,31-32,37-44,47,76-77H,6,13,18,23-30,59H2,1-5H3,(H2,60,78)(H2,61,79)(H2,62,80)(H,66,83)(H,67,82)(H,68,81)(H,69,86)(H,70,88)(H,71,84)(H,73,85)(H,74,87)(H3,63,64,65)(H2,72,75,89)/t32-,37+,38-,39-,40-,41-,42-,43-,44-,47-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.430 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392406
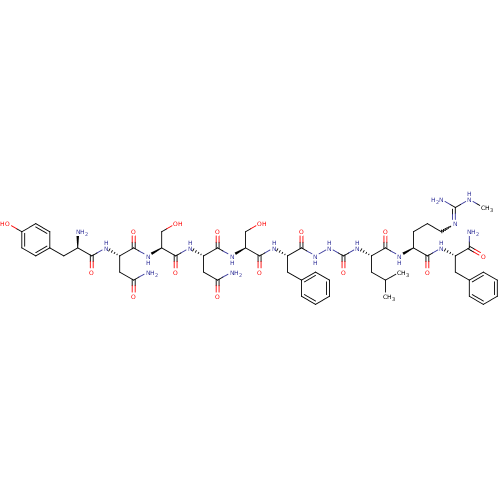
(CHEMBL2151647)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C55H79N17O15/c1-29(2)21-37(48(81)63-35(15-10-20-62-54(60)61-3)47(80)64-36(45(59)78)23-30-11-6-4-7-12-30)70-55(87)72-71-53(86)38(24-31-13-8-5-9-14-31)66-51(84)41(27-73)69-50(83)40(26-44(58)77)67-52(85)42(28-74)68-49(82)39(25-43(57)76)65-46(79)34(56)22-32-16-18-33(75)19-17-32/h4-9,11-14,16-19,29,34-42,73-75H,10,15,20-28,56H2,1-3H3,(H2,57,76)(H2,58,77)(H2,59,78)(H,63,81)(H,64,80)(H,65,79)(H,66,84)(H,67,85)(H,68,82)(H,69,83)(H,71,86)(H3,60,61,62)(H2,70,72,87)/t34-,35+,36+,37+,38+,39+,40+,41+,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392404

(CHEMBL2151645)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)CC(=O)N[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C64H85N17O14/c1-35(2)25-46(59(91)74-44(19-12-24-71-64(69)70-3)58(90)75-45(56(68)88)27-36-13-6-4-7-14-36)73-54(86)32-55(87)80-53(28-37-15-8-5-9-16-37)81-63(95)50(34-82)79-62(94)49(31-52(67)85)78-60(92)47(29-39-33-72-43-18-11-10-17-41(39)43)77-61(93)48(30-51(66)84)76-57(89)42(65)26-38-20-22-40(83)23-21-38/h4-11,13-18,20-23,33,35,42,44-50,53,72,82-83H,12,19,24-32,34,65H2,1-3H3,(H2,66,84)(H2,67,85)(H2,68,88)(H,73,86)(H,74,91)(H,75,90)(H,76,89)(H,77,93)(H,78,92)(H,79,94)(H,80,87)(H,81,95)(H3,69,70,71)/t42-,44+,45+,46+,47+,48+,49+,50+,53+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.510 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392403
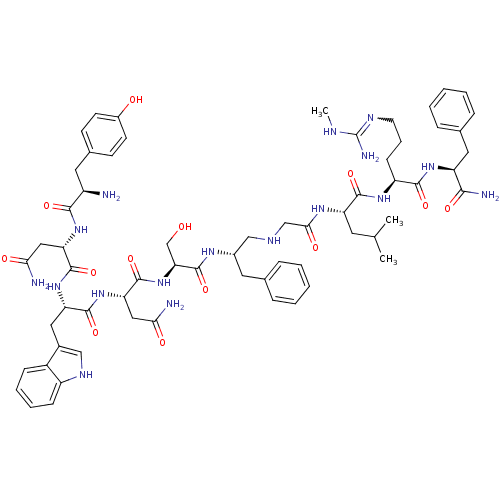
(CHEMBL2151644)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)CNC[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C64H87N17O13/c1-36(2)25-48(59(90)76-46(19-12-24-72-64(69)70-3)58(89)77-47(56(68)87)28-38-15-8-5-9-16-38)75-55(86)34-71-33-41(26-37-13-6-4-7-14-37)74-63(94)52(35-82)81-62(93)51(31-54(67)85)80-60(91)49(29-40-32-73-45-18-11-10-17-43(40)45)79-61(92)50(30-53(66)84)78-57(88)44(65)27-39-20-22-42(83)23-21-39/h4-11,13-18,20-23,32,36,41,44,46-52,71,73,82-83H,12,19,24-31,33-35,65H2,1-3H3,(H2,66,84)(H2,67,85)(H2,68,87)(H,74,94)(H,75,86)(H,76,90)(H,77,89)(H,78,88)(H,79,92)(H,80,91)(H,81,93)(H3,69,70,72)/t41-,44+,46-,47-,48-,49-,50-,51-,52-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.610 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392410
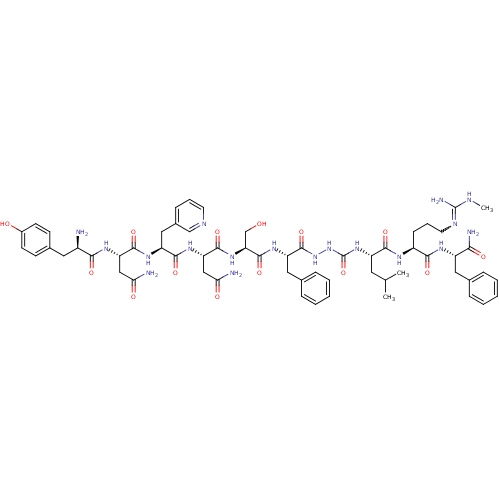
(CHEMBL2151651)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1cccnc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C60H82N18O14/c1-33(2)24-42(53(86)69-40(17-11-23-68-59(65)66-3)52(85)70-41(50(64)83)26-34-12-6-4-7-13-34)76-60(92)78-77-58(91)44(27-35-14-8-5-9-15-35)73-57(90)47(32-79)75-56(89)46(30-49(63)82)74-54(87)43(28-37-16-10-22-67-31-37)72-55(88)45(29-48(62)81)71-51(84)39(61)25-36-18-20-38(80)21-19-36/h4-10,12-16,18-22,31,33,39-47,79-80H,11,17,23-30,32,61H2,1-3H3,(H2,62,81)(H2,63,82)(H2,64,83)(H,69,86)(H,70,85)(H,71,84)(H,72,88)(H,73,90)(H,74,87)(H,75,89)(H,77,91)(H3,65,66,68)(H2,76,78,92)/t39-,40+,41+,42+,43+,44+,45+,46+,47+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM107306
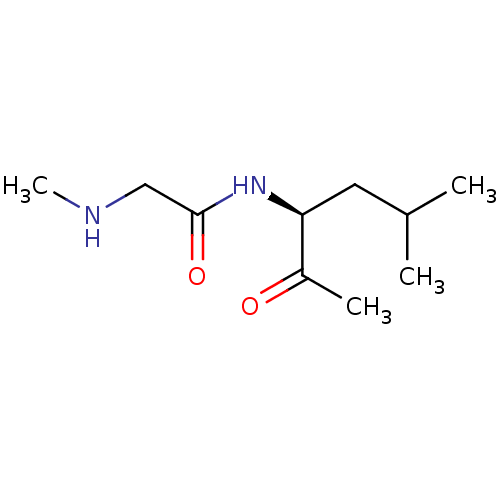
(US8592379, 1)Show InChI InChI=1S/C10H20N2O2/c1-7(2)5-9(8(3)13)12-10(14)6-11-4/h7,9,11H,5-6H2,1-4H3,(H,12,14)/t9-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University; Takeda Pharmaceutical Company Limited
US Patent
| Assay Description
Binding inhibition assay using human GPR54. |
US Patent US8592379 (2013)
BindingDB Entry DOI: 10.7270/Q2N0156N |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26338
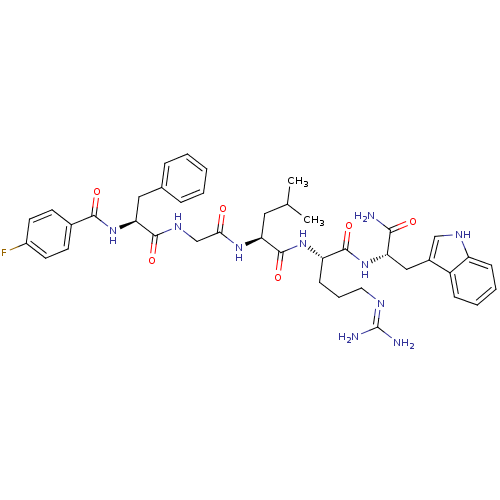
((2S)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carbamoy...)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:33.34,wD:12.20,4.4,44.45,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;6.76,.89,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C41H51FN10O6/c1-24(2)19-33(49-35(53)23-48-38(56)34(20-25-9-4-3-5-10-25)52-37(55)26-14-16-28(42)17-15-26)40(58)50-31(13-8-18-46-41(44)45)39(57)51-32(36(43)54)21-27-22-47-30-12-7-6-11-29(27)30/h3-7,9-12,14-17,22,24,31-34,47H,8,13,18-21,23H2,1-2H3,(H2,43,54)(H,48,56)(H,49,53)(H,50,58)(H,51,57)(H,52,55)(H4,44,45,46)/t31-,32-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26338

((2S)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carbamoy...)Show SMILES CC(C)C[C@H](NC(=O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:33.34,wD:12.20,4.4,44.45,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;6.76,.89,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C41H51FN10O6/c1-24(2)19-33(49-35(53)23-48-38(56)34(20-25-9-4-3-5-10-25)52-37(55)26-14-16-28(42)17-15-26)40(58)50-31(13-8-18-46-41(44)45)39(57)51-32(36(43)54)21-27-22-47-30-12-7-6-11-29(27)30/h3-7,9-12,14-17,22,24,31-34,47H,8,13,18-21,23H2,1-2H3,(H2,43,54)(H,48,56)(H,49,53)(H,50,58)(H,51,57)(H,52,55)(H4,44,45,46)/t31-,32-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]kisspeptin-15 from GPR54 |
ACS Med Chem Lett 2: 53-57 (2011)
Article DOI: 10.1021/ml1002053
BindingDB Entry DOI: 10.7270/Q2K07586 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392407
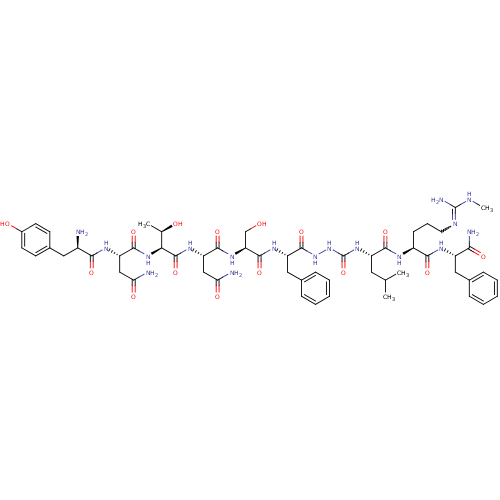
(CHEMBL2151648)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)[C@@H](C)O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C56H81N17O15/c1-29(2)22-38(49(82)64-36(16-11-21-63-55(61)62-4)48(81)65-37(46(60)79)24-31-12-7-5-8-13-31)70-56(88)73-72-53(86)39(25-32-14-9-6-10-15-32)67-52(85)42(28-74)69-50(83)40(26-43(58)77)68-54(87)45(30(3)75)71-51(84)41(27-44(59)78)66-47(80)35(57)23-33-17-19-34(76)20-18-33/h5-10,12-15,17-20,29-30,35-42,45,74-76H,11,16,21-28,57H2,1-4H3,(H2,58,77)(H2,59,78)(H2,60,79)(H,64,82)(H,65,81)(H,66,80)(H,67,85)(H,68,87)(H,69,83)(H,71,84)(H,72,86)(H3,61,62,63)(H2,70,73,88)/t30-,35-,36+,37+,38+,39+,40+,41+,42+,45+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.860 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM107308
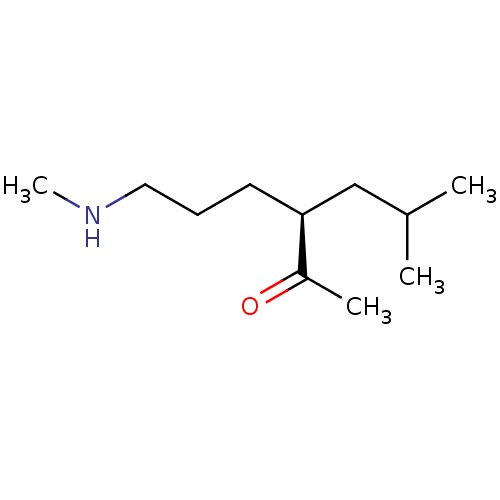
(US8592379, 20)Show InChI InChI=1S/C11H23NO/c1-9(2)8-11(10(3)13)6-5-7-12-4/h9,11-12H,5-8H2,1-4H3/t11-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University; Takeda Pharmaceutical Company Limited
US Patent
| Assay Description
Binding inhibition assay using human GPR54. |
US Patent US8592379 (2013)
BindingDB Entry DOI: 10.7270/Q2N0156N |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26340
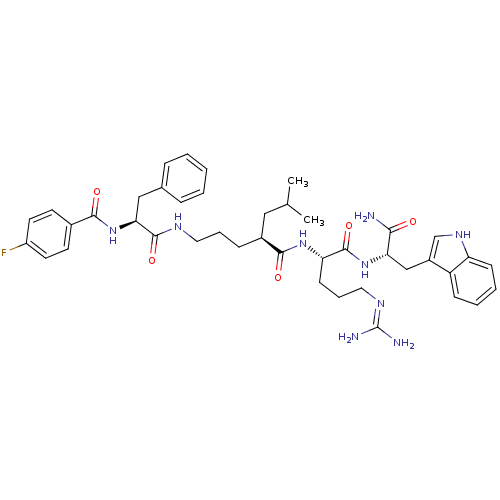
((2S)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carbamoy...)Show SMILES CC(C)C[C@H](CCCNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:32.33,wD:11.19,4.4,43.44,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C42H54FN9O5/c1-26(2)22-29(12-8-20-47-40(56)36(23-27-10-4-3-5-11-27)52-38(54)28-16-18-31(43)19-17-28)39(55)50-34(15-9-21-48-42(45)46)41(57)51-35(37(44)53)24-30-25-49-33-14-7-6-13-32(30)33/h3-7,10-11,13-14,16-19,25-26,29,34-36,49H,8-9,12,15,20-24H2,1-2H3,(H2,44,53)(H,47,56)(H,50,55)(H,51,57)(H,52,54)(H4,45,46,48)/t29-,34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392402
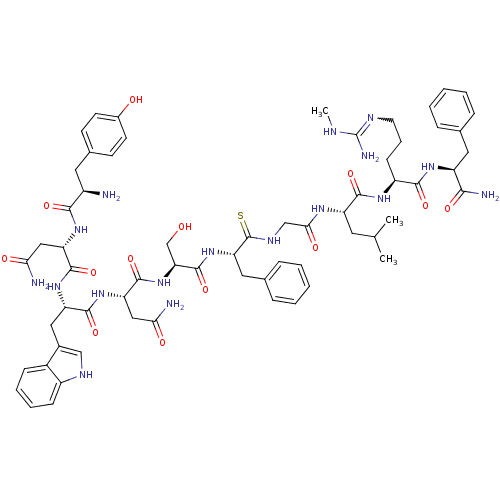
(CHEMBL2151643)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)CNC(=S)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C64H85N17O13S/c1-35(2)25-46(58(90)75-44(19-12-24-71-64(69)70-3)57(89)76-45(55(68)87)27-36-13-6-4-7-14-36)74-54(86)33-73-63(95)50(28-37-15-8-5-9-16-37)80-62(94)51(34-82)81-61(93)49(31-53(67)85)79-59(91)47(29-39-32-72-43-18-11-10-17-41(39)43)78-60(92)48(30-52(66)84)77-56(88)42(65)26-38-20-22-40(83)23-21-38/h4-11,13-18,20-23,32,35,42,44-51,72,82-83H,12,19,24-31,33-34,65H2,1-3H3,(H2,66,84)(H2,67,85)(H2,68,87)(H,73,95)(H,74,86)(H,75,90)(H,76,89)(H,77,88)(H,78,92)(H,79,91)(H,80,94)(H,81,93)(H3,69,70,71)/t42-,44+,45+,46+,47+,48+,49+,50+,51+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to human KISS1R |
Bioorg Med Chem Lett 22: 6391-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.069
BindingDB Entry DOI: 10.7270/Q2ZP477R |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322299
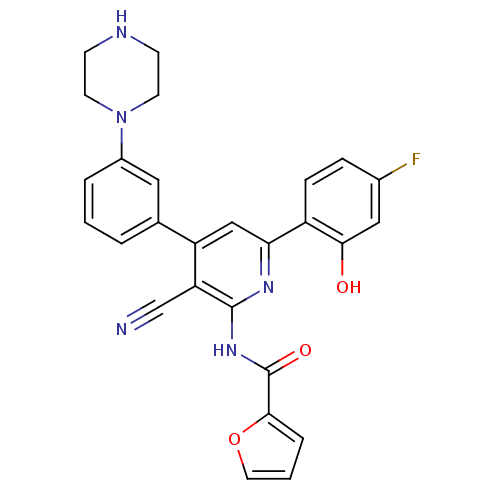
(CHEMBL1173643 | N-[3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES Oc1cc(F)ccc1-c1cc(-c2cccc(c2)N2CCNCC2)c(C#N)c(NC(=O)c2ccco2)n1 Show InChI InChI=1S/C27H22FN5O3/c28-18-6-7-20(24(34)14-18)23-15-21(17-3-1-4-19(13-17)33-10-8-30-9-11-33)22(16-29)26(31-23)32-27(35)25-5-2-12-36-25/h1-7,12-15,30,34H,8-11H2,(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50320821
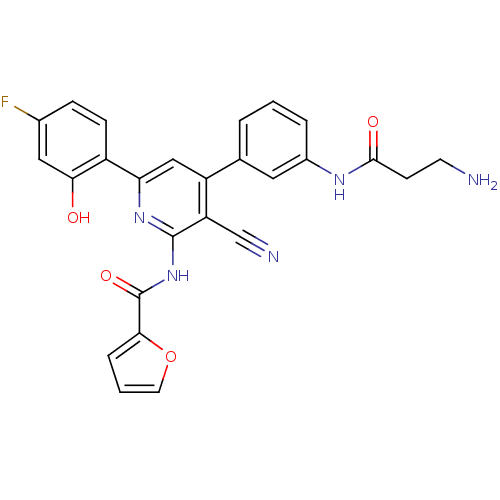
(CHEMBL1163809 | N-(4-(3-(3-aminopropanamido)phenyl...)Show SMILES NCCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C26H20FN5O4/c27-16-6-7-18(22(33)12-16)21-13-19(15-3-1-4-17(11-15)30-24(34)8-9-28)20(14-29)25(31-21)32-26(35)23-5-2-10-36-23/h1-7,10-13,33H,8-9,28H2,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin(40-54) from human GPR54 receptor expressed in CHO cells after 60 mins by scintillation counting |
Bioorg Med Chem 18: 3841-59 (2010)
Article DOI: 10.1016/j.bmc.2010.04.036
BindingDB Entry DOI: 10.7270/Q22807S6 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50320821

(CHEMBL1163809 | N-(4-(3-(3-aminopropanamido)phenyl...)Show SMILES NCCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C26H20FN5O4/c27-16-6-7-18(22(33)12-16)21-13-19(15-3-1-4-17(11-15)30-24(34)8-9-28)20(14-29)25(31-21)32-26(35)23-5-2-10-36-23/h1-7,10-13,33H,8-9,28H2,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322292
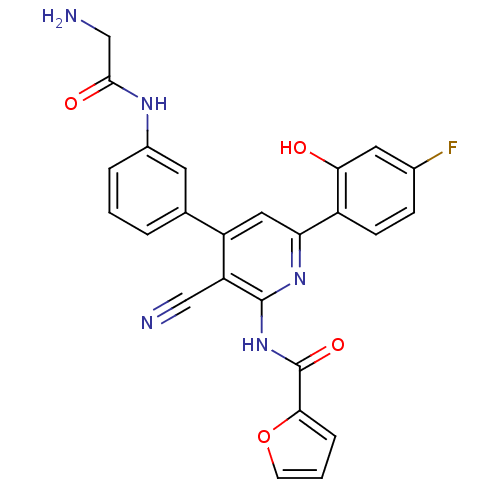
(CHEMBL1171484 | N-{3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES NCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C25H18FN5O4/c26-15-6-7-17(21(32)10-15)20-11-18(14-3-1-4-16(9-14)29-23(33)13-28)19(12-27)24(30-20)31-25(34)22-5-2-8-35-22/h1-11,32H,13,28H2,(H,29,33)(H,30,31,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM107309
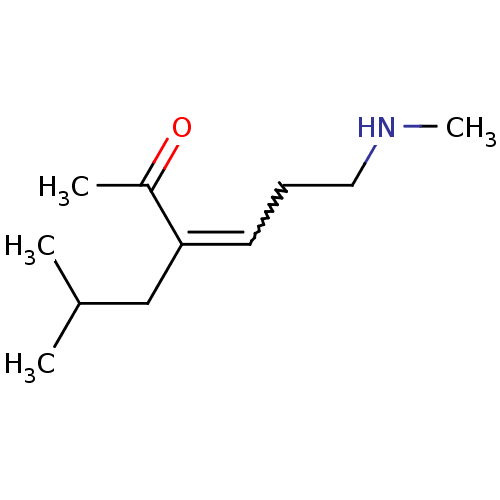
(US8592379, 21)Show InChI InChI=1S/C11H21NO/c1-9(2)8-11(10(3)13)6-5-7-12-4/h6,9,12H,5,7-8H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University; Takeda Pharmaceutical Company Limited
US Patent
| Assay Description
Binding inhibition assay using human GPR54. |
US Patent US8592379 (2013)
BindingDB Entry DOI: 10.7270/Q2N0156N |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26341
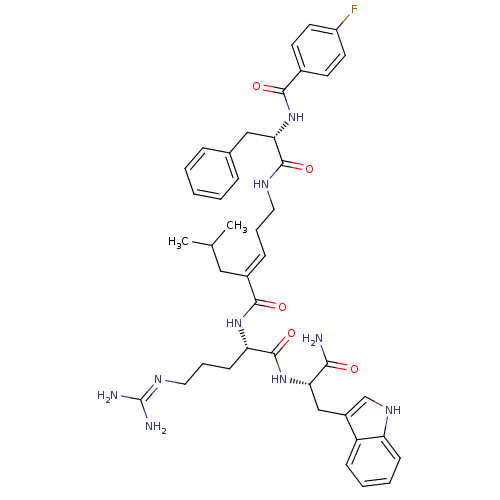
((2E)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carbamoy...)Show SMILES CC(C)C\C(=C/CCNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:32.33,wD:11.19,43.44,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C42H52FN9O5/c1-26(2)22-29(12-8-20-47-40(56)36(23-27-10-4-3-5-11-27)52-38(54)28-16-18-31(43)19-17-28)39(55)50-34(15-9-21-48-42(45)46)41(57)51-35(37(44)53)24-30-25-49-33-14-7-6-13-32(30)33/h3-7,10-14,16-19,25-26,34-36,49H,8-9,15,20-24H2,1-2H3,(H2,44,53)(H,47,56)(H,50,55)(H,51,57)(H,52,54)(H4,45,46,48)/b29-12+/t34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322307
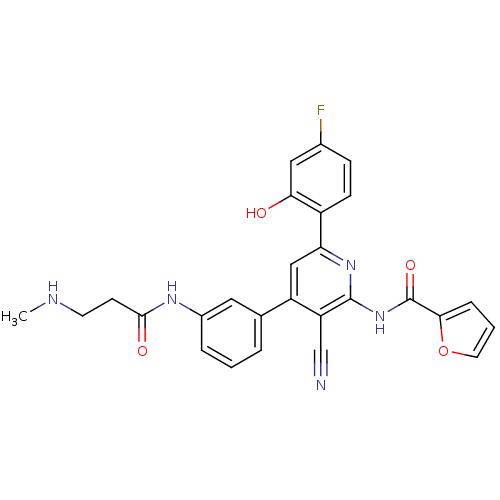
(CHEMBL1169901 | N-[3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES CNCCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C27H22FN5O4/c1-30-10-9-25(35)31-18-5-2-4-16(12-18)20-14-22(19-8-7-17(28)13-23(19)34)32-26(21(20)15-29)33-27(36)24-6-3-11-37-24/h2-8,11-14,30,34H,9-10H2,1H3,(H,31,35)(H,32,33,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM107312
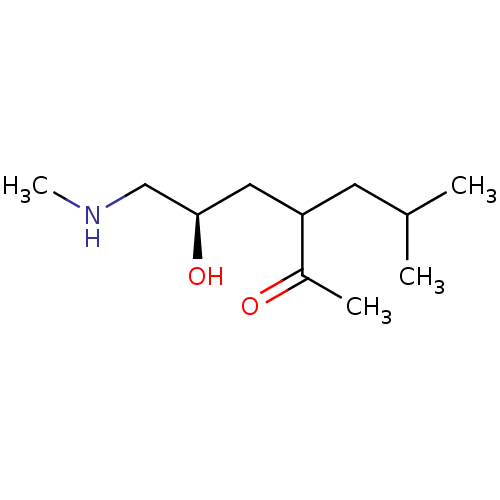
(US8592379, 24a/24b | US8592379, 35)Show InChI InChI=1S/C11H23NO2/c1-8(2)5-10(9(3)13)6-11(14)7-12-4/h8,10-12,14H,5-7H2,1-4H3/t10?,11-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto University; Takeda Pharmaceutical Company Limited
US Patent
| Assay Description
Binding inhibition assay using human GPR54. |
US Patent US8592379 (2013)
BindingDB Entry DOI: 10.7270/Q2N0156N |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM26345
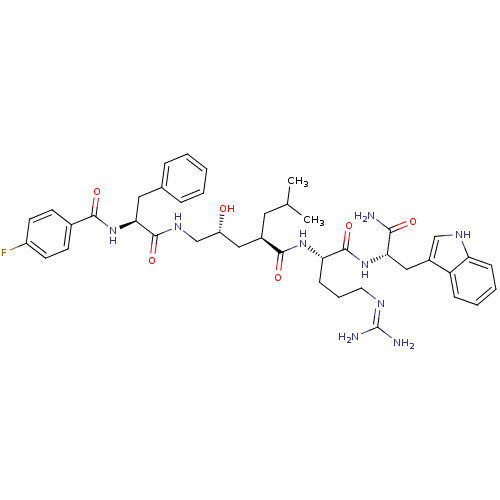
((2R,4R)-N-[(1S)-4-carbamimidamido-1-{[(1S)-1-carba...)Show SMILES CC(C)C[C@H](C[C@@H](O)CNC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccc(F)cc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(N)=O |r,wU:33.34,6.6,wD:12.20,4.31,44.45,(10.76,-1.42,;10.76,.12,;12.1,.89,;9.43,.89,;9.43,2.43,;8.09,3.2,;6.76,2.43,;6.76,.89,;5.43,3.2,;4.09,2.43,;2.76,3.2,;2.76,4.74,;1.43,2.43,;1.43,.89,;.66,-.45,;-.87,-.66,;-1.45,-2.09,;-.5,-3.3,;1.02,-3.09,;1.6,-1.66,;.09,3.2,;-1.24,2.43,;-1.24,.89,;-2.57,3.2,;-2.57,4.74,;-3.91,5.51,;-5.24,4.74,;-6.58,5.51,;-5.24,3.2,;-3.91,2.43,;10.76,3.2,;10.76,4.74,;12.1,2.43,;13.43,3.2,;13.43,4.74,;12.1,5.51,;12.1,7.05,;10.76,7.82,;10.76,9.36,;12.1,10.13,;9.43,10.13,;14.76,2.43,;14.76,.89,;16.1,3.2,;17.43,2.43,;17.43,.89,;18.76,.12,;18.15,-1.3,;19.3,-2.32,;20.63,-1.53,;22.1,-2,;23.23,-.96,;22.9,.55,;21.43,1.01,;20.3,-.03,;18.76,3.2,;20.1,2.43,;18.76,4.74,)| Show InChI InChI=1S/C42H54FN9O6/c1-25(2)19-28(21-31(53)24-49-40(57)36(20-26-9-4-3-5-10-26)52-38(55)27-14-16-30(43)17-15-27)39(56)50-34(13-8-18-47-42(45)46)41(58)51-35(37(44)54)22-29-23-48-33-12-7-6-11-32(29)33/h3-7,9-12,14-17,23,25,28,31,34-36,48,53H,8,13,18-22,24H2,1-2H3,(H2,44,54)(H,49,57)(H,50,56)(H,51,58)(H,52,55)(H4,45,46,47)/t28-,31-,34+,35+,36+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Kyoto University
| Assay Description
Membranes were incubated with [125I] kisspeptin-15 and increasing concentrations of test compound in binding buffer. The reaction mixtures were dilut... |
J Med Chem 51: 7645-9 (2008)
Article DOI: 10.1021/jm800930w
BindingDB Entry DOI: 10.7270/Q2ZS2TTP |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322300
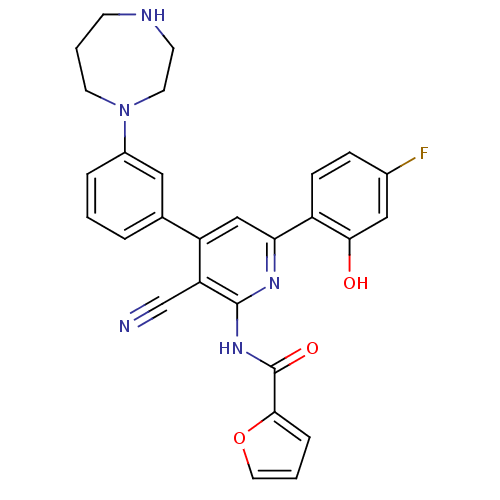
(CHEMBL1173644 | N-{3-Cyano-4-[3-(1,4-diazepan-1-yl...)Show SMILES Oc1cc(F)ccc1-c1cc(-c2cccc(c2)N2CCCNCC2)c(C#N)c(NC(=O)c2ccco2)n1 Show InChI InChI=1S/C28H24FN5O3/c29-19-7-8-21(25(35)15-19)24-16-22(18-4-1-5-20(14-18)34-11-3-9-31-10-12-34)23(17-30)27(32-24)33-28(36)26-6-2-13-37-26/h1-2,4-8,13-16,31,35H,3,9-12H2,(H,32,33,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50320810
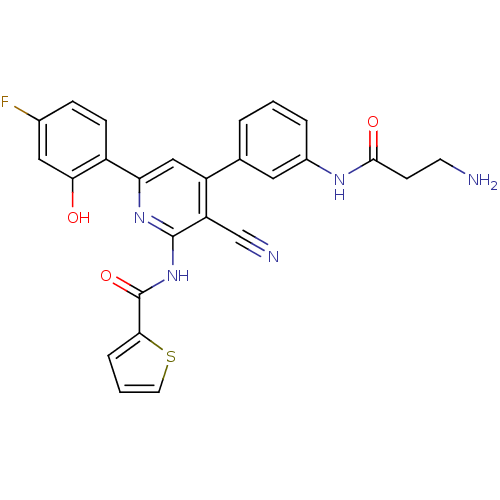
(CHEMBL1165222 | N-(4-(3-(3-aminopropanamido)phenyl...)Show SMILES NCCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2cccs2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C26H20FN5O3S/c27-16-6-7-18(22(33)12-16)21-13-19(15-3-1-4-17(11-15)30-24(34)8-9-28)20(14-29)25(31-21)32-26(35)23-5-2-10-36-23/h1-7,10-13,33H,8-9,28H2,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin(40-54) from human GPR54 receptor expressed in CHO cells after 60 mins by scintillation counting |
Bioorg Med Chem 18: 3841-59 (2010)
Article DOI: 10.1016/j.bmc.2010.04.036
BindingDB Entry DOI: 10.7270/Q22807S6 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50320810

(CHEMBL1165222 | N-(4-(3-(3-aminopropanamido)phenyl...)Show SMILES NCCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2cccs2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C26H20FN5O3S/c27-16-6-7-18(22(33)12-16)21-13-19(15-3-1-4-17(11-15)30-24(34)8-9-28)20(14-29)25(31-21)32-26(35)23-5-2-10-36-23/h1-7,10-13,33H,8-9,28H2,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322309
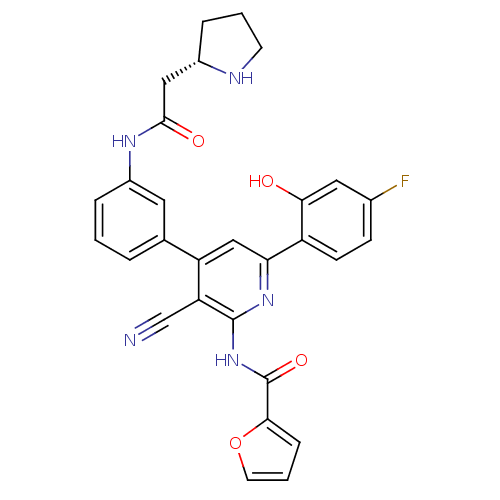
(CHEMBL1170903 | N-[3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES Oc1cc(F)ccc1-c1cc(-c2cccc(NC(=O)C[C@@H]3CCCN3)c2)c(C#N)c(NC(=O)c2ccco2)n1 |r| Show InChI InChI=1S/C29H24FN5O4/c30-18-8-9-21(25(36)13-18)24-15-22(23(16-31)28(34-24)35-29(38)26-7-3-11-39-26)17-4-1-5-20(12-17)33-27(37)14-19-6-2-10-32-19/h1,3-5,7-9,11-13,15,19,32,36H,2,6,10,14H2,(H,33,37)(H,34,35,38)/t19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50320822
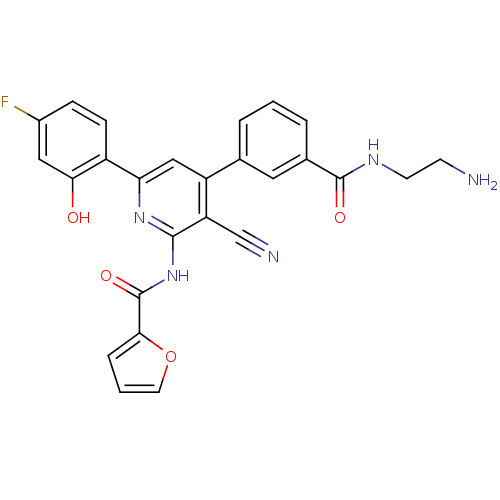
(CHEMBL1163736 | N-[4-{3-[(2-Aminoethyl)carbamoyl]p...)Show SMILES NCCNC(=O)c1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C26H20FN5O4/c27-17-6-7-18(22(33)12-17)21-13-19(15-3-1-4-16(11-15)25(34)30-9-8-28)20(14-29)24(31-21)32-26(35)23-5-2-10-36-23/h1-7,10-13,33H,8-9,28H2,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin(40-54) from human GPR54 receptor expressed in CHO cells after 60 mins by scintillation counting |
Bioorg Med Chem 18: 3841-59 (2010)
Article DOI: 10.1016/j.bmc.2010.04.036
BindingDB Entry DOI: 10.7270/Q22807S6 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322313
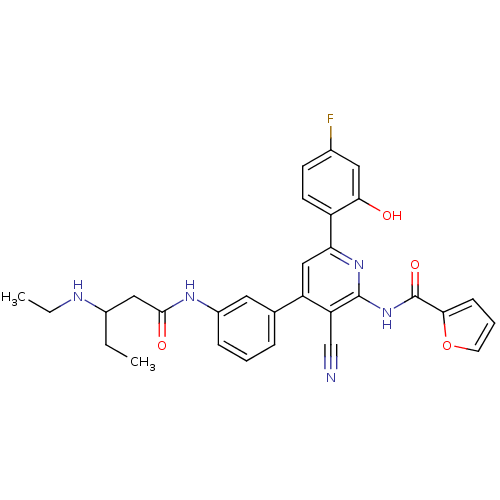
(CHEMBL1170896 | rac-N-[3-Cyano-4-(3-{[3-(ethylamin...)Show SMILES CCNC(CC)CC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C30H28FN5O4/c1-3-20(33-4-2)15-28(38)34-21-8-5-7-18(13-21)23-16-25(22-11-10-19(31)14-26(22)37)35-29(24(23)17-32)36-30(39)27-9-6-12-40-27/h5-14,16,20,33,37H,3-4,15H2,1-2H3,(H,34,38)(H,35,36,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322293
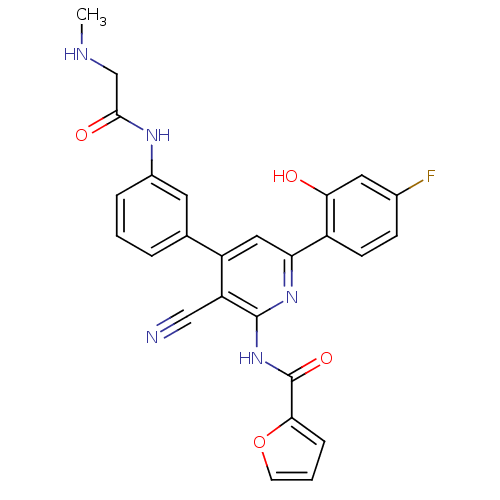
(CHEMBL1170911 | N-[3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES CNCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C26H20FN5O4/c1-29-14-24(34)30-17-5-2-4-15(10-17)19-12-21(18-8-7-16(27)11-22(18)33)31-25(20(19)13-28)32-26(35)23-6-3-9-36-23/h2-12,29,33H,14H2,1H3,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50442961
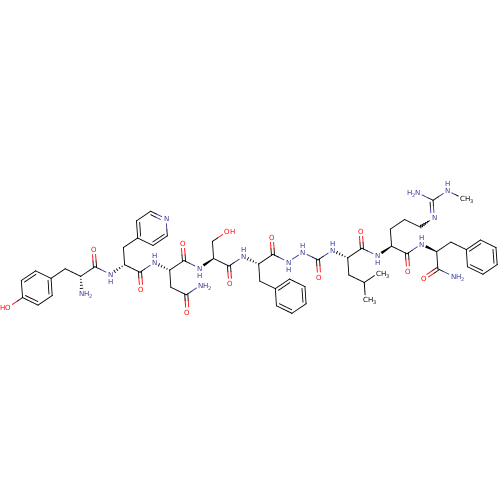
(CHEMBL3086284)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](Cc1ccncc1)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C56H76N16O12/c1-32(2)25-41(50(79)64-39(15-10-22-63-55(60)61-3)49(78)65-40(47(59)76)27-33-11-6-4-7-12-33)70-56(84)72-71-54(83)43(28-34-13-8-5-9-14-34)67-53(82)45(31-73)69-52(81)44(30-46(58)75)68-51(80)42(29-36-20-23-62-24-21-36)66-48(77)38(57)26-35-16-18-37(74)19-17-35/h4-9,11-14,16-21,23-24,32,38-45,73-74H,10,15,22,25-31,57H2,1-3H3,(H2,58,75)(H2,59,76)(H,64,79)(H,65,78)(H,66,77)(H,67,82)(H,68,80)(H,69,81)(H,71,83)(H3,60,61,63)(H2,70,72,84)/t38-,39+,40+,41+,42-,43+,44+,45+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Inhibition of human KISS1R expressed in CHO cells assessed as inhibition of KISS1R-mediated cell growth after 4 days by cell counting kit-8 assay |
J Med Chem 56: 8298-307 (2013)
Article DOI: 10.1021/jm401056w
BindingDB Entry DOI: 10.7270/Q25M675S |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50392405

(CHEMBL2151646)Show SMILES CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,w:4.4| Show InChI InChI=1S/C63H84N18O14/c1-34(2)25-45(56(89)72-43(19-12-24-70-62(68)69-3)55(88)73-44(53(67)86)27-35-13-6-4-7-14-35)79-63(95)81-80-61(94)46(28-36-15-8-5-9-16-36)75-60(93)50(33-82)78-59(92)49(31-52(66)85)77-57(90)47(29-38-32-71-42-18-11-10-17-40(38)42)76-58(91)48(30-51(65)84)74-54(87)41(64)26-37-20-22-39(83)23-21-37/h4-11,13-18,20-23,32,34,41,43-50,71,82-83H,12,19,24-31,33,64H2,1-3H3,(H2,65,84)(H2,66,85)(H2,67,86)(H,72,89)(H,73,88)(H,74,87)(H,75,93)(H,76,91)(H,77,90)(H,78,92)(H,80,94)(H3,68,69,70)(H2,79,81,95)/t41-,43+,44+,45+,46+,47+,48+,49+,50+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd
Curated by ChEMBL
| Assay Description
Inhibition of human KISS1R expressed in CHO cells assessed as inhibition of KISS1R-mediated cell growth after 4 days by cell counting kit-8 assay |
J Med Chem 56: 8298-307 (2013)
Article DOI: 10.1021/jm401056w
BindingDB Entry DOI: 10.7270/Q25M675S |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322319
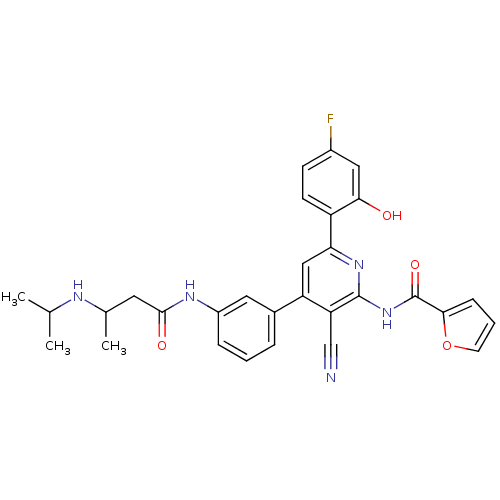
(CHEMBL1170692 | rac-N-{3-Cyano-6-(4-fluoro-2-hydro...)Show SMILES CC(C)NC(C)CC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C30H28FN5O4/c1-17(2)33-18(3)12-28(38)34-21-7-4-6-19(13-21)23-15-25(22-10-9-20(31)14-26(22)37)35-29(24(23)16-32)36-30(39)27-8-5-11-40-27/h4-11,13-15,17-18,33,37H,12H2,1-3H3,(H,34,38)(H,35,36,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322303
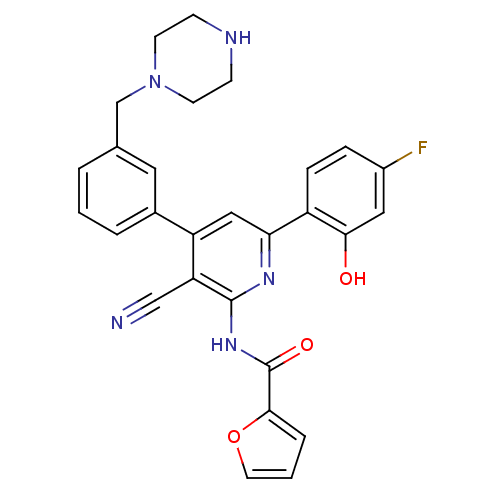
(CHEMBL1170870 | N-{3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES Oc1cc(F)ccc1-c1cc(-c2cccc(CN3CCNCC3)c2)c(C#N)c(NC(=O)c2ccco2)n1 Show InChI InChI=1S/C28H24FN5O3/c29-20-6-7-21(25(35)14-20)24-15-22(19-4-1-3-18(13-19)17-34-10-8-31-9-11-34)23(16-30)27(32-24)33-28(36)26-5-2-12-37-26/h1-7,12-15,31,35H,8-11,17H2,(H,32,33,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50320811
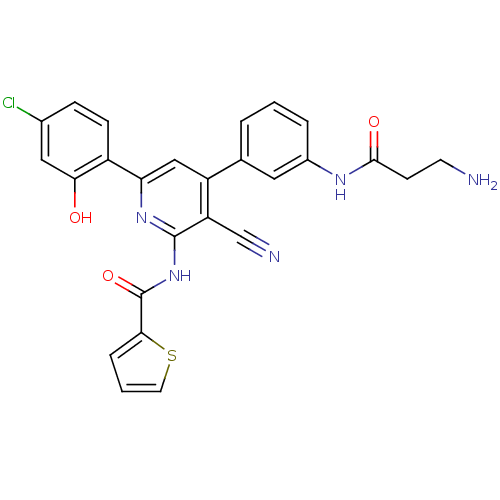
(CHEMBL1163896 | N-{4-[3-(beta-Alanylamino)phenyl]-...)Show SMILES NCCC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2cccs2)c1C#N)-c1ccc(Cl)cc1O Show InChI InChI=1S/C26H20ClN5O3S/c27-16-6-7-18(22(33)12-16)21-13-19(15-3-1-4-17(11-15)30-24(34)8-9-28)20(14-29)25(31-21)32-26(35)23-5-2-10-36-23/h1-7,10-13,33H,8-9,28H2,(H,30,34)(H,31,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin(40-54) from human GPR54 receptor expressed in CHO cells after 60 mins by scintillation counting |
Bioorg Med Chem 18: 3841-59 (2010)
Article DOI: 10.1016/j.bmc.2010.04.036
BindingDB Entry DOI: 10.7270/Q22807S6 |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322310
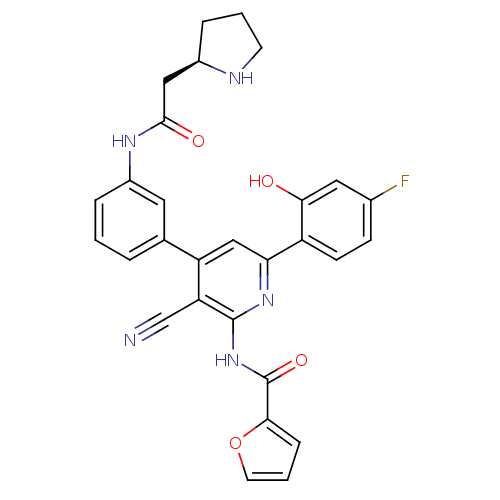
(CHEMBL1170904 | N-[3-Cyano-6-(4-fluoro-2-hydroxyph...)Show SMILES Oc1cc(F)ccc1-c1cc(-c2cccc(NC(=O)C[C@H]3CCCN3)c2)c(C#N)c(NC(=O)c2ccco2)n1 |r| Show InChI InChI=1S/C29H24FN5O4/c30-18-8-9-21(25(36)13-18)24-15-22(23(16-31)28(34-24)35-29(38)26-7-3-11-39-26)17-4-1-5-20(12-17)33-27(37)14-19-6-2-10-32-19/h1,3-5,7-9,11-13,15,19,32,36H,2,6,10,14H2,(H,33,37)(H,34,35,38)/t19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |
KiSS-1 receptor
(Homo sapiens (Human)) | BDBM50322318
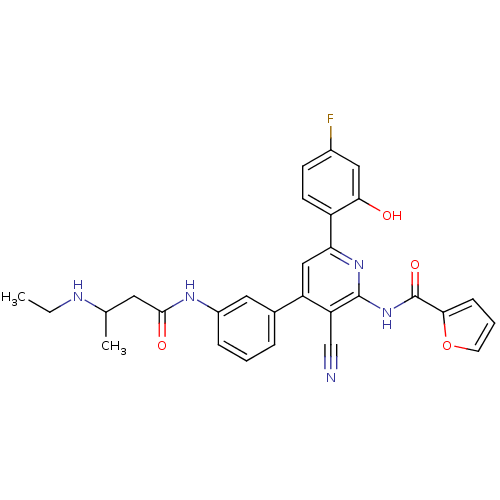
(CHEMBL1170685 | rac-N-[3-Cyano-4-(3-{[3-(ethylamin...)Show SMILES CCNC(C)CC(=O)Nc1cccc(c1)-c1cc(nc(NC(=O)c2ccco2)c1C#N)-c1ccc(F)cc1O Show InChI InChI=1S/C29H26FN5O4/c1-3-32-17(2)12-27(37)33-20-7-4-6-18(13-20)22-15-24(21-10-9-19(30)14-25(21)36)34-28(23(22)16-31)35-29(38)26-8-5-11-39-26/h4-11,13-15,17,32,36H,3,12H2,1-2H3,(H,33,37)(H,34,35,38) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd
Curated by ChEMBL
| Assay Description
Displacement of [125I]metastin from human GPR54 receptor expressed in CHO cells |
Bioorg Med Chem 18: 5157-71 (2010)
Article DOI: 10.1016/j.bmc.2010.05.061
BindingDB Entry DOI: 10.7270/Q21R6RHM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

















































