

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lanosterol 14-alpha demethylase (Rattus norvegicus) | BDBM50161586 (4-{(2S,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Apparent Ki for rat Lanosterol 14-alpha demethylase | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Homo sapiens (Human)) | BDBM31768 (CHEMBL295698 | Ketoconazole | Nizoral | Panfungol) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Rattus norvegicus) | BDBM50044425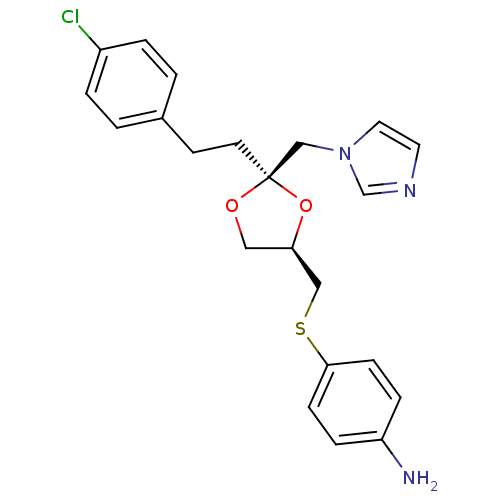 (4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Inhibition of lanosterol 14-alpha-demethylase of rat hepatic microsomes | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Homo sapiens (Human)) | BDBM50044425 (4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Homo sapiens (Human)) | BDBM50044425 (4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 40.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Homo sapiens (Human)) | BDBM31768 (CHEMBL295698 | Ketoconazole | Nizoral | Panfungol) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 64 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Rattus norvegicus) | BDBM31768 (CHEMBL295698 | Ketoconazole | Nizoral | Panfungol) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Inhibition of lanosterol 14-alpha-demethylase of rat hepatic microsomes | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Rattus norvegicus) | BDBM31653 (CHEMBL421109 | imidazole-dioxolane, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 117 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research Curated by ChEMBL | Assay Description Apparent Ki for rat Lanosterol 14-alpha demethylase | J Med Chem 36: 2235-7 (1993) BindingDB Entry DOI: 10.7270/Q2ZW1K0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol 14-alpha demethylase (Rattus norvegicus) | BDBM50213693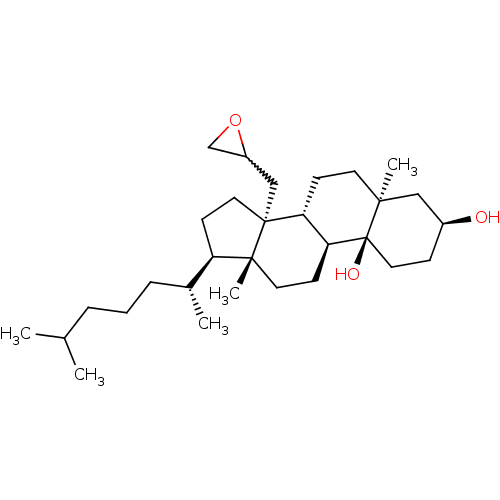 ((3S,5S,8R,9S,10S,13R,14R,17R)-5,13-dimethyl-17-((R...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sinhgad College of Pharmacy Curated by ChEMBL | Assay Description Inhibition of rat liver LDM | Bioorg Med Chem 15: 4674-99 (2007) Article DOI: 10.1016/j.bmc.2007.04.031 BindingDB Entry DOI: 10.7270/Q24J0FZ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||