

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409468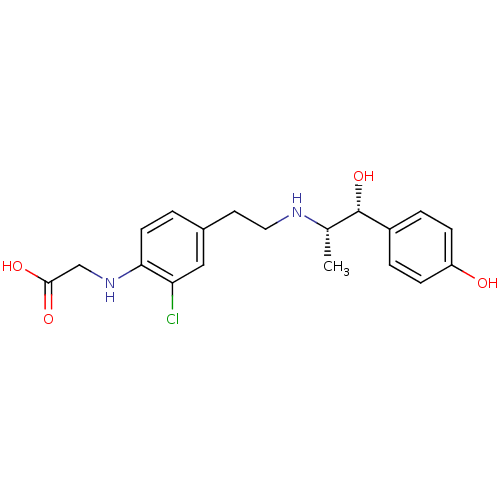 (CHEMBL32670) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 0.933 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50002133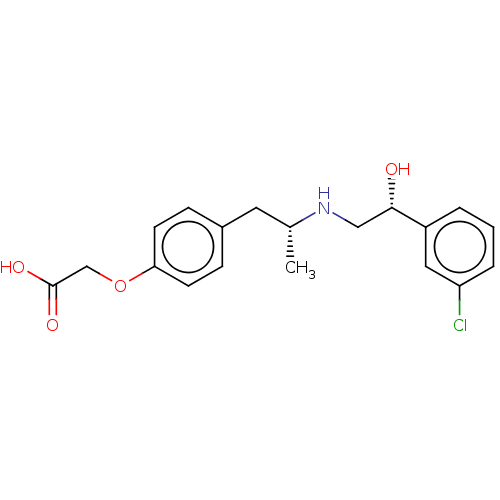 ((+/-)-2-(4-((R)-2-((R)-2-(3-chlorophenyl)-2-hydrox...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 2.19 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50002134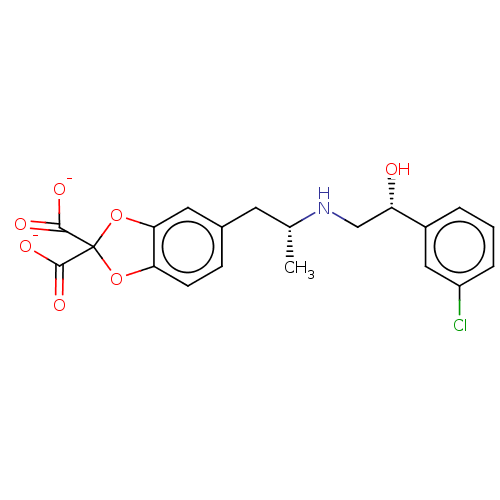 (5-((2R)-2-{[(2R)-2-(3-chlorophenyl)-2-hydroxyethyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
American Cyanamid Company Curated by ChEMBL | Assay Description Compound was evaluated in vitro for beta-adrenergic activity against beta-3 adrenergic receptor in rat epididymal fat pads by stimulation of glycerol... | J Med Chem 35: 3081-4 (1992) BindingDB Entry DOI: 10.7270/Q2PR7TWN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50379086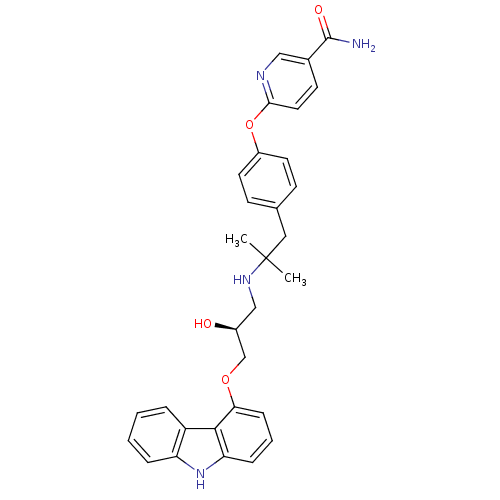 (CHEMBL2012521 | CHEMBL2012522 | LY-377604) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Agonist activity at rat adrenergic beta3 receptor expressed in DHB-11 CHO cells assessed as cAMP accumulation after 20 mins by scintillation proximit... | ACS Med Chem Lett 2: 583-586 (2011) Article DOI: 10.1021/ml200071k BindingDB Entry DOI: 10.7270/Q20R9QDP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409471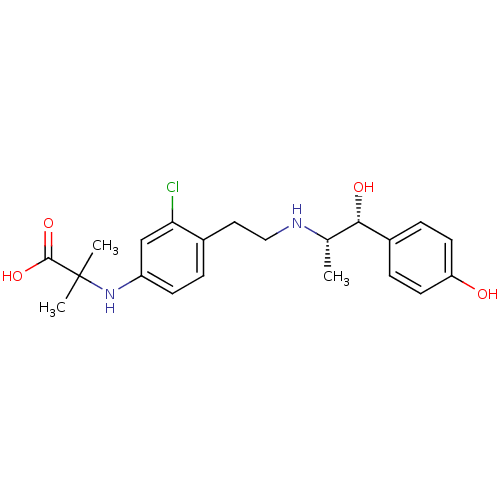 (CHEMBL32586) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 5.62 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50224021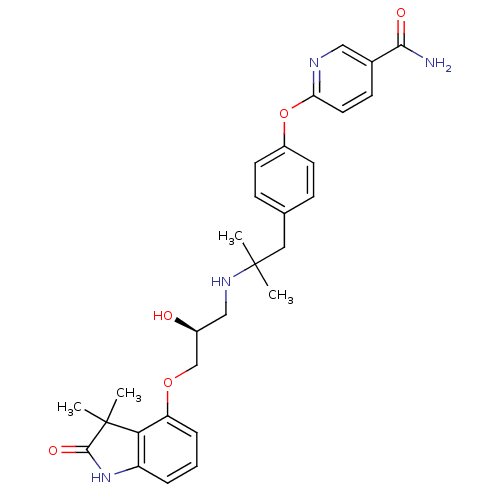 ((S)-6-(4-(2-(3-(3,3-dimethyl-2-oxoindolin-4-yloxy)...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 6.5 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta-3 adrenergic receptor | Bioorg Med Chem Lett 17: 6270-3 (2007) Article DOI: 10.1016/j.bmcl.2007.09.011 BindingDB Entry DOI: 10.7270/Q22V2FV6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50193805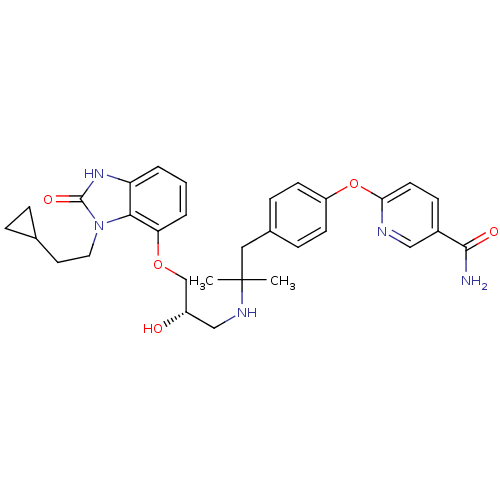 ((S)-6-(4-(2-(3-(3-(2-cyclopropylethyl)-2-oxo-2,3-d...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 6.80 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta-3 adrenergic receptor expressed in CHO cells assessed as cAMP levels | Bioorg Med Chem Lett 16: 5691-4 (2006) Article DOI: 10.1016/j.bmcl.2006.08.010 BindingDB Entry DOI: 10.7270/Q2251HVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409479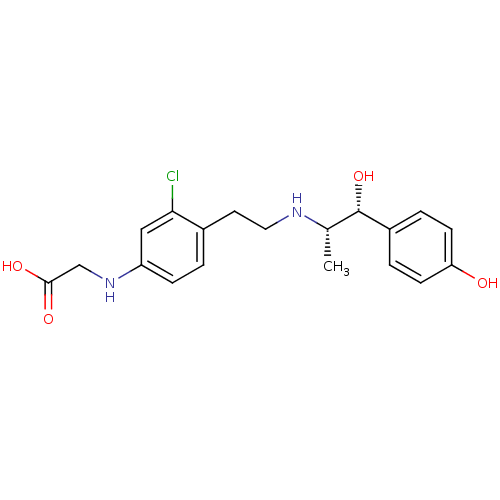 (CHEMBL32933) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 7.24 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50002133 ((+/-)-2-(4-((R)-2-((R)-2-(3-chlorophenyl)-2-hydrox...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 8.40 | n/a | n/a | n/a | n/a |
American Cyanamid Company Curated by ChEMBL | Assay Description Compound was evaluated in vitro for beta-adrenergic activity against beta-3 adrenergic receptor in rat epididymal fat pads by stimulation of glycerol... | J Med Chem 35: 3081-4 (1992) BindingDB Entry DOI: 10.7270/Q2PR7TWN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409483 (CHEMBL32115) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 11.0 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409484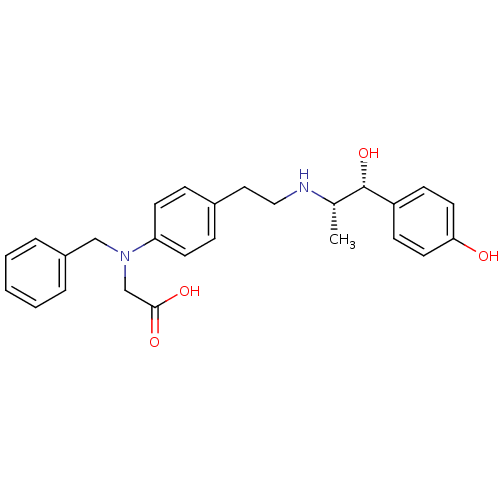 (CHEMBL32470) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 12.9 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50218164 ((R)-2-(2-(4-(2-methylthiazol-4-yl)phenoxy)ethylami...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 13 | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Agonist activity at rat adrenergic beta-3 receptor assessed as cAMP production | Bioorg Med Chem Lett 17: 5245-50 (2007) Article DOI: 10.1016/j.bmcl.2007.06.072 BindingDB Entry DOI: 10.7270/Q2MK6CMZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM25392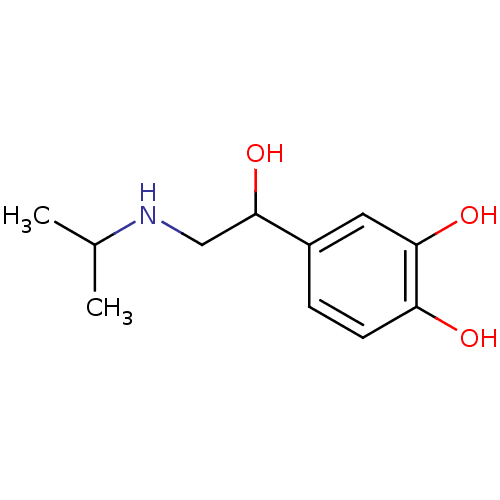 (4-[1-hydroxy-2-(isopropylamino)ethyl]pyrocatechol;...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 13 | n/a | n/a | n/a | n/a |
American Cyanamid Company Curated by ChEMBL | Assay Description Compound was evaluated in vitro for beta adrenergic receptor activity against beta-3 adrenergic receptor in rat epididymal fat pads by stimulation of... | J Med Chem 35: 3081-4 (1992) BindingDB Entry DOI: 10.7270/Q2PR7TWN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409472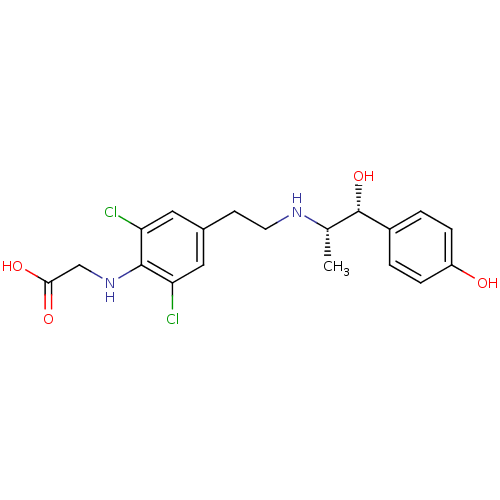 (CHEMBL32391) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 14.1 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50002132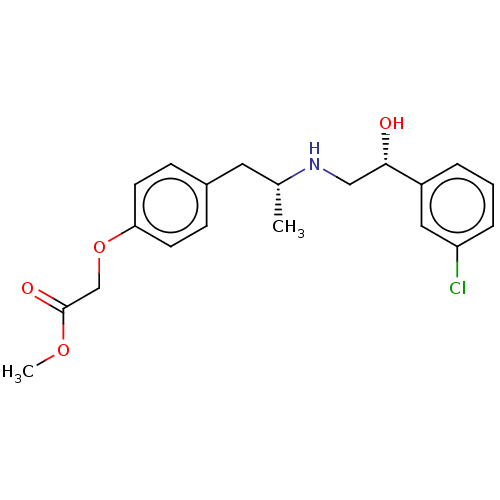 ((4-{(R)-2-[(R)-2-(3-Chloro-phenyl)-2-hydroxy-ethyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 16 | n/a | n/a | n/a | n/a |
American Cyanamid Company Curated by ChEMBL | Assay Description Compound was evaluated in vitro for beta-adrenergic activity against beta-3 adrenergic receptor in rat epididymal fat pads by stimulation of glycerol... | J Med Chem 35: 3081-4 (1992) BindingDB Entry DOI: 10.7270/Q2PR7TWN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409482 (CHEMBL35459) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 22.9 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409470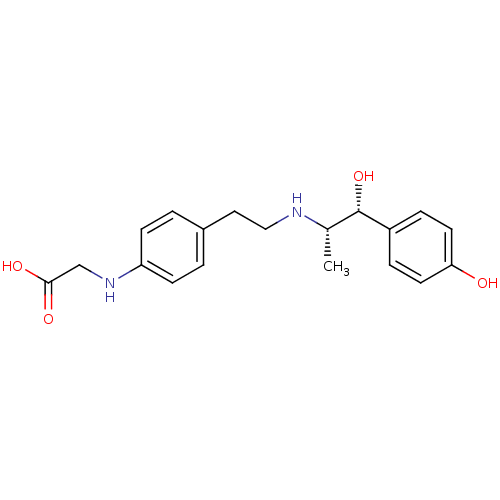 (CHEMBL32532) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 24.0 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50193803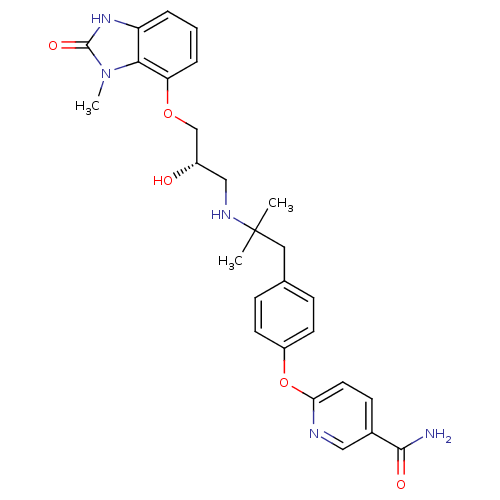 ((S)-6-(4-(2-(2-hydroxy-3-(3-methyl-2-oxo-2,3-dihyd...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta-3 adrenergic receptor expressed in CHO cells assessed as cAMP levels | Bioorg Med Chem Lett 16: 5691-4 (2006) Article DOI: 10.1016/j.bmcl.2006.08.010 BindingDB Entry DOI: 10.7270/Q2251HVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50002134 (5-((2R)-2-{[(2R)-2-(3-chlorophenyl)-2-hydroxyethyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 25.1 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50218165 ((R)-1-(pyridin-3-yl)-2-(2-(4-(thiazol-4-yl)phenoxy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 26 | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Agonist activity at rat adrenergic beta-3 receptor assessed as cAMP production | Bioorg Med Chem Lett 17: 5245-50 (2007) Article DOI: 10.1016/j.bmcl.2007.06.072 BindingDB Entry DOI: 10.7270/Q2MK6CMZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50193804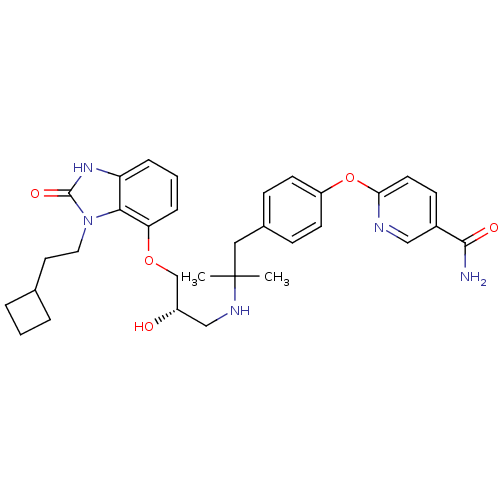 ((S)-6-(4-(2-(3-(3-(2-cyclobutylethyl)-2-oxo-2,3-di...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 29 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta-3 adrenergic receptor expressed in CHO cells assessed as cAMP levels | Bioorg Med Chem Lett 16: 5691-4 (2006) Article DOI: 10.1016/j.bmcl.2006.08.010 BindingDB Entry DOI: 10.7270/Q2251HVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409480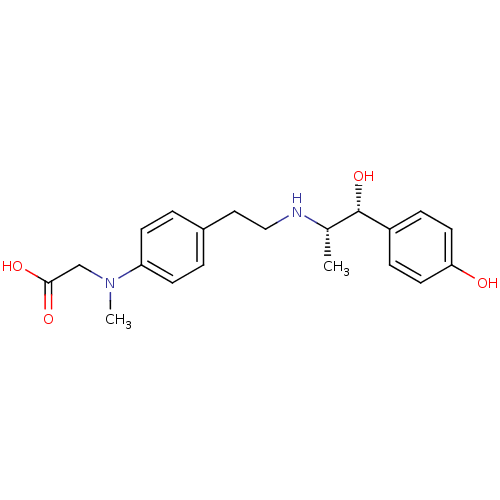 (CHEMBL33360) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 52.5 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409469 (CHEMBL284520) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 61.7 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409473 (CHEMBL33399) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 81.3 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50146154 (KRP-114V | MK-4618 | Vibegron) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 86 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM25392 (4-[1-hydroxy-2-(isopropylamino)ethyl]pyrocatechol;...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 87.1 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. EC50, the mean concentration required to produce 50% relaxation of detrusor before the addition in th... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50146154 (KRP-114V | MK-4618 | Vibegron) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 122 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presence ... | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409474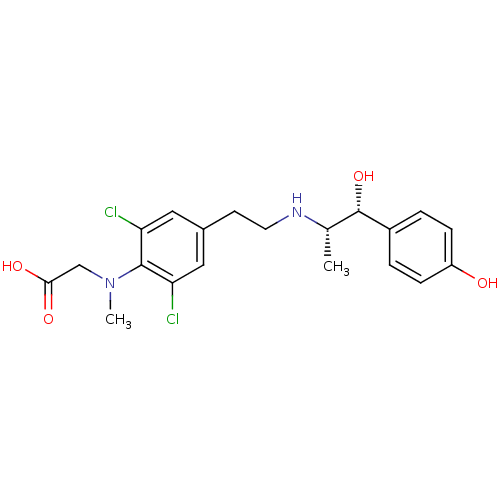 (CHEMBL417137) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 158 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM97162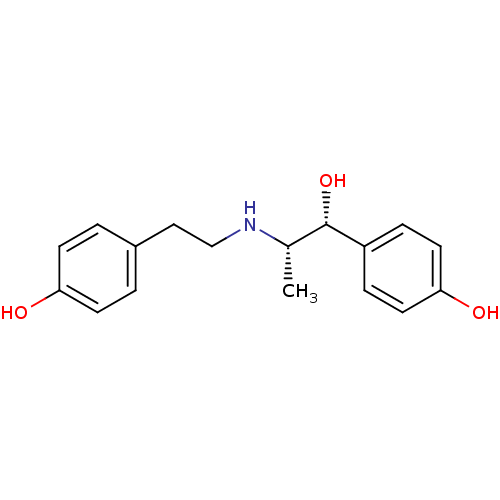 (MLS002153782 | RITODRINE | RITODRINE HYDROCHLORIDE...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase DrugBank KEGG PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 191 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50146160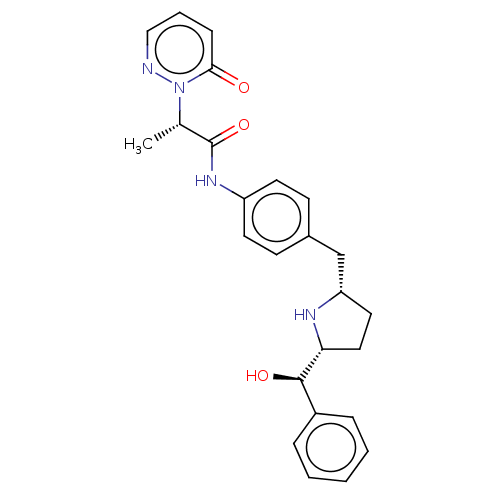 (CHEMBL3765335) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 245 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presence ... | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409478 (CHEMBL285888) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 457 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50146160 (CHEMBL3765335) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 531 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50108088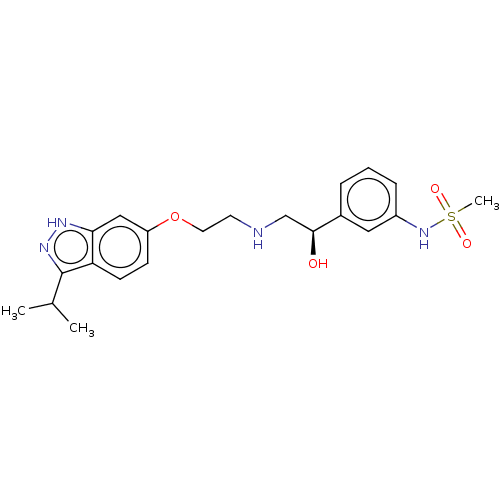 (CHEMBL3601305) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 535 | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Agonist activity at rat beta3-adrenergic receptor | J Med Chem 58: 6048-57 (2015) Article DOI: 10.1021/acs.jmedchem.5b00638 BindingDB Entry DOI: 10.7270/Q2NG4SDR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409475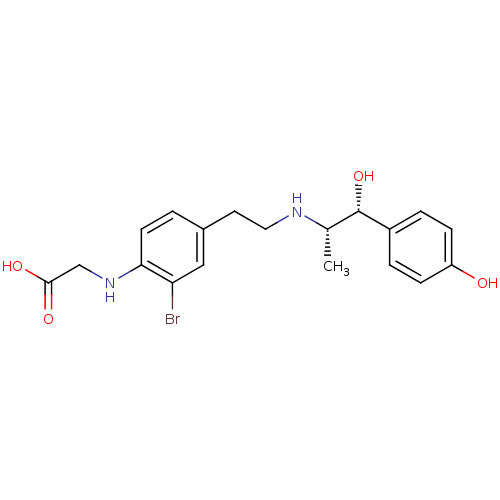 (CHEMBL32939) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 575 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50146156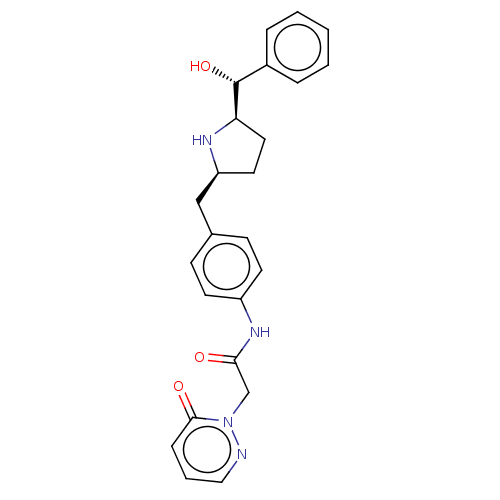 (CHEMBL3764088) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 615 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409481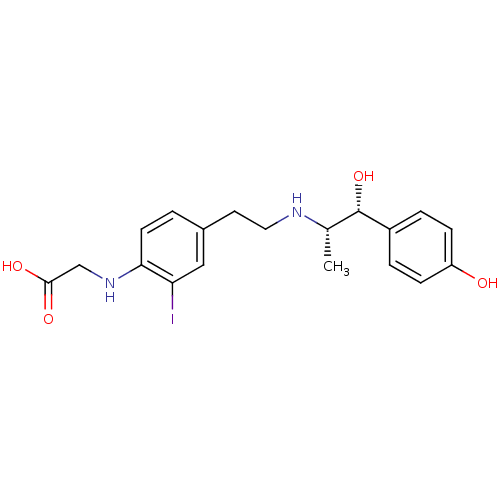 (CHEMBL35738) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | n/a | n/a | 813 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50409476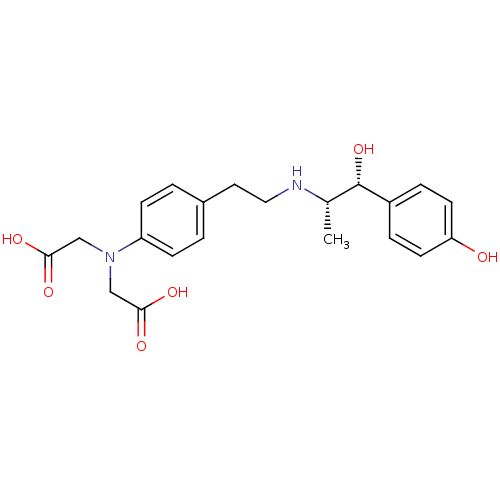 (CHEMBL289670) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 832 | n/a | n/a | n/a | n/a |
Kissei Pharmaceutical Company Ltd. Curated by ChEMBL | Assay Description Agonistic activity towards beta-3 adrenoceptor. Mean concentration required to produce 50% relaxation of detrusor before the addition in the ferret d... | J Med Chem 44: 1436-45 (2001) BindingDB Entry DOI: 10.7270/Q2Q52QVH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50146156 (CHEMBL3764088) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.06E+3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay in presence ... | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50029051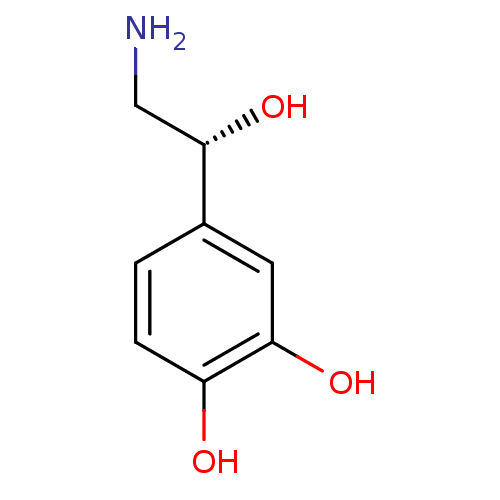 ((-)-arterenol | (-)-noradrenaline | (-)-norepineph...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.58E+3 | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals Curated by ChEMBL | Assay Description Effective dose for contraction of reserpine-pretreated rat vas deferens | J Med Chem 38: 3681-716 (1995) BindingDB Entry DOI: 10.7270/Q2SB46C4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50092645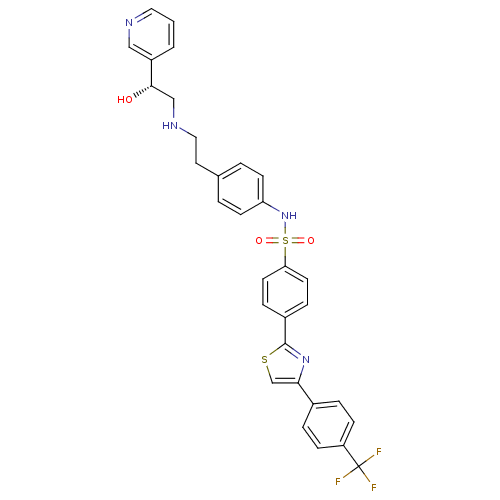 ((R)-N-(4-(2-(2-hydroxy-2-(pyridin-3-yl)ethylamino)...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Agonist activity at rat beta3 adrenergic receptor expressed in CHO cells assessed as accumulation of cAMP after 30 mins by TR-FRET assay | J Med Chem 59: 609-23 (2016) Article DOI: 10.1021/acs.jmedchem.5b01372 BindingDB Entry DOI: 10.7270/Q2M047B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50407518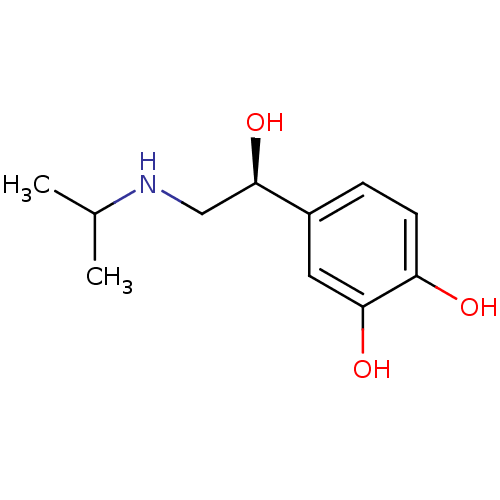 (ISOPROTERENOL | L-ISOPRENALINE | LEVISOPRENALINE) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 6.30E+4 | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals Curated by ChEMBL | Assay Description Activity for beta 3-adrenoceptor was assessed from effect on stimulation of lipolysis in rat adipocytes. | J Med Chem 38: 3681-716 (1995) BindingDB Entry DOI: 10.7270/Q2SB46C4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-3 adrenergic receptor (Rattus norvegicus) | BDBM50029051 ((-)-arterenol | (-)-noradrenaline | (-)-norepineph...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals Curated by ChEMBL | Assay Description Activity for beta 3-adrenoceptor was assessed from effect on stimulation of lipolysis in rat adipocytes. | J Med Chem 38: 3681-716 (1995) BindingDB Entry DOI: 10.7270/Q2SB46C4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||