 Found 31 hits of ic50 for UniProtKB: P00743
Found 31 hits of ic50 for UniProtKB: P00743 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Bos taurus) | BDBM50029498
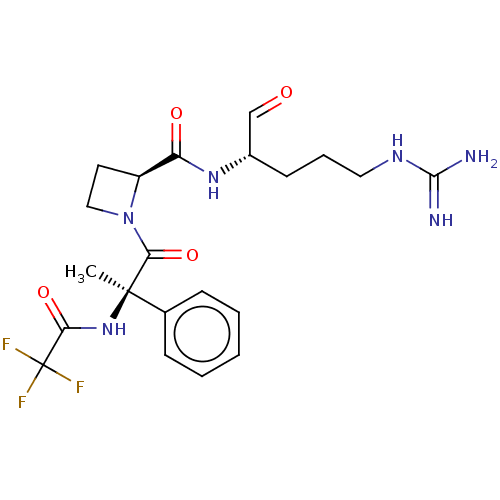
(1N-[2-{2-[4-amino(imino)methylamino-1-formyl-(1S)-...)Show SMILES [#6][C@]([#7]-[#6](=O)C(F)(F)F)([#6](=O)-[#7]-1-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]=O)c1ccccc1 Show InChI InChI=1S/C21H27F3N6O4/c1-20(13-6-3-2-4-7-13,29-17(33)21(22,23)24)18(34)30-11-9-15(30)16(32)28-14(12-31)8-5-10-27-19(25)26/h2-4,6-7,12,14-15H,5,8-11H2,1H3,(H,28,32)(H,29,33)(H4,25,26,27)/t14-,15-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 9.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029507
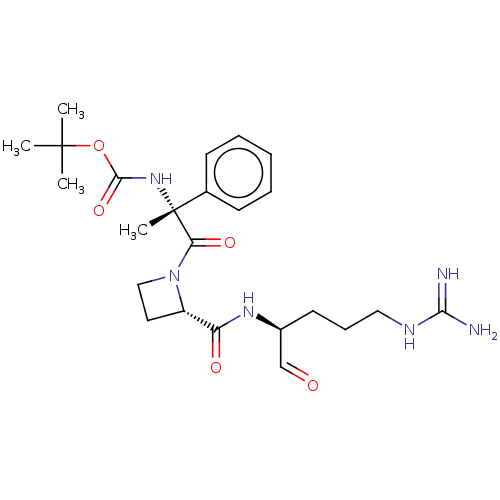
(CHEMBL2370860 | {(R)-2-[2-((S)-1-Formyl-4-guanidin...)Show SMILES [#6]C([#6])([#6])[#8]-[#6](=O)-[#7][C@@]([#6])([#6](=O)-[#7]-1-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]=O)c1ccccc1 Show InChI InChI=1S/C24H36N6O5/c1-23(2,3)35-22(34)29-24(4,16-9-6-5-7-10-16)20(33)30-14-12-18(30)19(32)28-17(15-31)11-8-13-27-21(25)26/h5-7,9-10,15,17-18H,8,11-14H2,1-4H3,(H,28,32)(H,29,34)(H4,25,26,27)/t17-,18-,24+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50323720
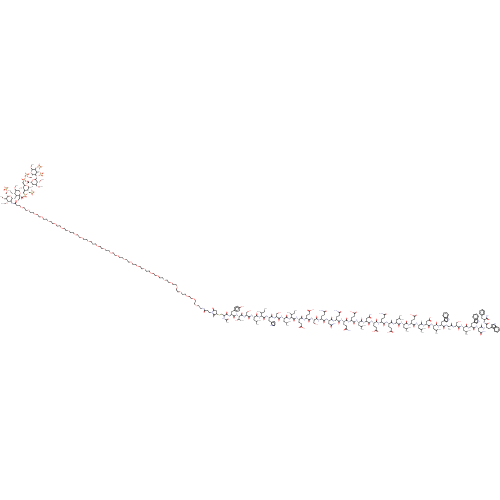
(CHEMBL1213113)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)C(CSC1CC(=O)N(CCC(=O)NCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCC(=O)N[C@@H]2[C@@H](O[C@H]3[C@H](OC)[C@@H](OC)[C@H](O[C@@H]4[C@@H](CS([O-])(=O)=O)O[C@H](O[C@H]5[C@H](OC)[C@@H](OC)[C@H](O[C@H]6[C@H](COS([O-])(=O)=O)O[C@@H](OC)[C@@H](OS([O-])(=O)=O)[C@H]6OS([O-])(=O)=O)O[C@H]5C([O-])=O)[C@H](CS([O-])(=O)=O)[C@H]4CS([O-])(=O)=O)O[C@]3(CC)C([O-])=O)O[C@@H](COS([O-])(=O)=O)[C@H](OC)[C@H]2OC)C1=O)NC(C)=O)[C@@H](C)O)[C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C315H499N53O142S8/c1-32-179(20)253(304(434)341-208(66-76-251(396)397)276(406)336-207(65-75-250(394)395)281(411)361-228(161-370)299(429)340-202(60-70-239(319)379)277(407)355-223(151-240(320)380)292(422)337-201(59-69-238(318)378)274(404)332-199(57-67-236(316)376)272(402)334-205(63-73-248(390)391)279(409)344-212(139-173(8)9)287(417)356-224(152-241(321)381)293(423)339-203(61-71-246(386)387)275(405)333-200(58-68-237(317)377)273(403)335-204(62-72-247(388)389)278(408)343-211(138-172(6)7)284(414)346-210(137-171(4)5)283(413)338-206(64-74-249(392)393)280(410)345-213(140-174(10)11)288(418)358-226(154-252(398)399)295(425)347-214(141-175(12)13)285(415)352-219(147-186-156-326-196-50-41-38-47-191(186)196)282(412)330-181(22)271(401)360-227(160-369)300(430)348-215(142-176(14)15)286(416)353-221(149-188-158-328-198-52-43-40-49-193(188)198)290(420)357-225(153-242(322)382)294(424)354-220(148-187-157-327-197-51-42-39-48-192(187)197)289(419)342-209(270(323)400)145-184-45-36-35-37-46-184)365-296(426)216(143-177(16)17)349-301(431)229(162-371)362-291(421)222(150-189-159-324-170-329-189)359-305(435)254(180(21)33-2)366-297(427)217(144-178(18)19)350-302(432)230(163-372)363-306(436)255(182(23)373)367-298(428)218(146-185-53-55-190(375)56-54-185)351-303(433)231(331-183(24)374)166-511-235-155-245(385)368(307(235)437)80-77-243(383)325-79-84-473-88-92-477-96-100-481-104-108-485-112-116-489-120-124-492-126-128-494-130-132-496-134-136-497-135-133-495-131-129-493-127-125-491-122-118-487-114-110-483-106-102-479-98-94-475-90-86-471-82-44-81-470-85-89-474-93-97-478-101-105-482-109-113-486-117-121-490-123-119-488-115-111-484-107-103-480-99-95-476-91-87-472-83-78-244(384)364-256-260(464-26)258(463-25)232(164-498-515(451,452)453)500-310(256)507-269-264(466-28)267(468-30)313(508-315(269,34-3)314(440)441)503-257-194(167-512(442,443)444)195(168-513(445,446)447)309(502-234(257)169-514(448,449)450)505-262-261(465-27)266(467-29)312(506-265(262)308(438)439)504-259-233(165-499-516(454,455)456)501-311(469-31)268(510-518(460,461)462)263(259)509-517(457,458)459/h35-43,45-56,156-159,170-182,194-195,199-235,253-269,309-313,326-328,369-373,375H,32-34,44,57-155,160-169H2,1-31H3,(H2,316,376)(H2,317,377)(H2,318,378)(H2,319,379)(H2,320,380)(H2,321,381)(H2,322,382)(H2,323,400)(H,324,329)(H,325,383)(H,330,412)(H,331,374)(H,332,404)(H,333,405)(H,334,402)(H,335,403)(H,336,406)(H,337,422)(H,338,413)(H,339,423)(H,340,429)(H,341,434)(H,342,419)(H,343,408)(H,344,409)(H,345,410)(H,346,414)(H,347,425)(H,348,430)(H,349,431)(H,350,432)(H,351,433)(H,352,415)(H,353,416)(H,354,424)(H,355,407)(H,356,417)(H,357,420)(H,358,418)(H,359,435)(H,360,401)(H,361,411)(H,362,421)(H,363,436)(H,364,384)(H,365,426)(H,366,427)(H,367,428)(H,386,387)(H,388,389)(H,390,391)(H,392,393)(H,394,395)(H,396,397)(H,398,399)(H,438,439)(H,440,441)(H,442,443,444)(H,445,446,447)(H,448,449,450)(H,451,452,453)(H,454,455,456)(H,457,458,459)(H,460,461,462)/p-9/t179-,180-,181-,182+,194+,195+,199-,200-,201-,202-,203-,204-,205-,206-,207-,208-,209-,210-,211-,212-,213-,214-,215-,216-,217-,218-,219-,220-,221-,222-,223-,224-,225-,226-,227-,228-,229-,230-,231?,232-,233-,234+,235?,253-,254-,255-,256-,257-,258-,259-,260-,261-,262-,263-,264+,265+,266+,267+,268-,269-,309+,310+,311+,312+,313+,315-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 53 | n/a | n/a | n/a | n/a | n/a | n/a |
Endotis Pharma
Curated by ChEMBL
| Assay Description
Inhibition of bovine coagulation factor 10a by spectrophotometry |
Antimicrob Agents Chemother 54: 134-42 (2009)
Article DOI: 10.1128/AAC.00827-09
BindingDB Entry DOI: 10.7270/Q23N23KQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50323719
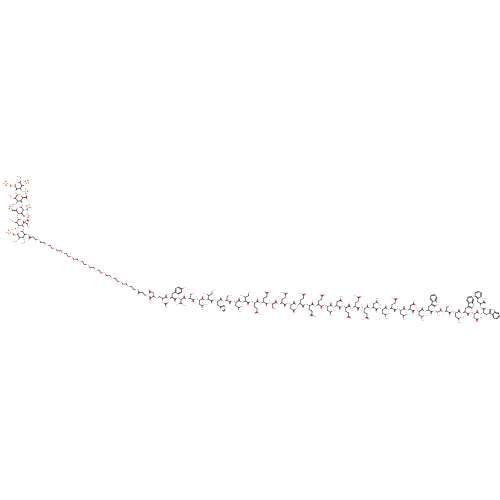
(CHEMBL1213112)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)C(CSC1CC(=O)N(CCC(=O)NCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCC(=O)N[C@@H]2[C@@H](O[C@H]3[C@H](OC)[C@@H](OC)[C@H](O[C@@H]4[C@@H](CS([O-])(=O)=O)O[C@H](O[C@H]5[C@H](OC)[C@@H](OC)[C@H](O[C@H]6[C@H](COS([O-])(=O)=O)O[C@@H](OC)[C@@H](OS([O-])(=O)=O)[C@H]6OS([O-])(=O)=O)O[C@H]5C([O-])=O)[C@H](CS([O-])(=O)=O)[C@H]4CS([O-])(=O)=O)O[C@]3(CC)C([O-])=O)O[C@@H](COS([O-])(=O)=O)[C@H](OC)[C@H]2OC)C1=O)NC(C)=O)[C@@H](C)O)[C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C282H433N53O126S8/c1-32-146(20)220(332-263(393)183(110-144(16)17)316-268(398)196(129-338)329-258(388)189(117-156-126-291-137-296-156)326-272(402)221(147(21)33-2)333-264(394)184(111-145(18)19)317-269(399)197(130-339)330-273(403)222(149(23)340)334-265(395)185(113-152-52-54-157(342)55-53-152)318-270(400)198(298-150(24)341)133-462-202-122-212(352)335(274(202)404)79-76-210(350)292-78-81-438-83-85-440-87-89-442-91-93-444-95-97-446-99-101-448-103-102-447-100-98-445-96-94-443-92-90-441-88-86-439-84-82-437-80-77-211(351)331-223-227(431-26)225(430-25)199(131-449-466(418,419)420)451-277(223)458-236-231(433-28)234(435-30)280(459-282(236,34-3)281(407)408)454-224-161(134-463(409,410)411)162(135-464(412,413)414)276(453-201(224)136-465(415,416)417)456-229-228(432-27)233(434-29)279(457-232(229)275(405)406)455-226-200(132-450-467(421,422)423)452-278(436-31)235(461-469(427,428)429)230(226)460-468(424,425)426)271(401)308-175(65-75-218(363)364)243(373)303-174(64-74-217(361)362)248(378)328-195(128-337)266(396)307-169(59-69-206(286)346)244(374)322-190(118-207(287)347)259(389)304-168(58-68-205(285)345)241(371)299-166(56-66-203(283)343)239(369)301-172(62-72-215(357)358)246(376)311-179(106-140(8)9)254(384)323-191(119-208(288)348)260(390)306-170(60-70-213(353)354)242(372)300-167(57-67-204(284)344)240(370)302-171(61-71-214(355)356)245(375)310-178(105-139(6)7)251(381)313-177(104-138(4)5)250(380)305-173(63-73-216(359)360)247(377)312-180(107-141(10)11)255(385)325-193(121-219(365)366)262(392)314-181(108-142(12)13)252(382)319-186(114-153-123-293-163-49-41-38-46-158(153)163)249(379)297-148(22)238(368)327-194(127-336)267(397)315-182(109-143(14)15)253(383)320-188(116-155-125-295-165-51-43-40-48-160(155)165)257(387)324-192(120-209(289)349)261(391)321-187(115-154-124-294-164-50-42-39-47-159(154)164)256(386)309-176(237(290)367)112-151-44-36-35-37-45-151/h35-55,123-126,137-149,161-162,166-202,220-236,276-280,293-295,336-340,342H,32-34,56-122,127-136H2,1-31H3,(H2,283,343)(H2,284,344)(H2,285,345)(H2,286,346)(H2,287,347)(H2,288,348)(H2,289,349)(H2,290,367)(H,291,296)(H,292,350)(H,297,379)(H,298,341)(H,299,371)(H,300,372)(H,301,369)(H,302,370)(H,303,373)(H,304,389)(H,305,380)(H,306,390)(H,307,396)(H,308,401)(H,309,386)(H,310,375)(H,311,376)(H,312,377)(H,313,381)(H,314,392)(H,315,397)(H,316,398)(H,317,399)(H,318,400)(H,319,382)(H,320,383)(H,321,391)(H,322,374)(H,323,384)(H,324,387)(H,325,385)(H,326,402)(H,327,368)(H,328,378)(H,329,388)(H,330,403)(H,331,351)(H,332,393)(H,333,394)(H,334,395)(H,353,354)(H,355,356)(H,357,358)(H,359,360)(H,361,362)(H,363,364)(H,365,366)(H,405,406)(H,407,408)(H,409,410,411)(H,412,413,414)(H,415,416,417)(H,418,419,420)(H,421,422,423)(H,424,425,426)(H,427,428,429)/p-9/t146-,147-,148-,149+,161+,162+,166-,167-,168-,169-,170-,171-,172-,173-,174-,175-,176-,177-,178-,179-,180-,181-,182-,183-,184-,185-,186-,187-,188-,189-,190-,191-,192-,193-,194-,195-,196-,197-,198?,199-,200-,201+,202?,220-,221-,222-,223-,224-,225-,226-,227-,228-,229-,230-,231+,232+,233+,234+,235-,236-,276+,277+,278+,279+,280+,282-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
Endotis Pharma
Curated by ChEMBL
| Assay Description
Inhibition of bovine coagulation factor 10a by spectrophotometry |
Antimicrob Agents Chemother 54: 134-42 (2009)
Article DOI: 10.1128/AAC.00827-09
BindingDB Entry DOI: 10.7270/Q23N23KQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50323718

(CHEMBL1213080)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)C(CSC1CC(=O)N(CCC(=O)NCCOCCOCCOCCOCCC(=O)N[C@@H]2[C@@H](O[C@H]3[C@H](OC)[C@@H](OC)[C@H](O[C@@H]4[C@@H](CS([O-])(=O)=O)O[C@H](O[C@H]5[C@H](OC)[C@@H](OC)[C@H](O[C@H]6[C@H](COS([O-])(=O)=O)O[C@@H](OC)[C@@H](OS([O-])(=O)=O)[C@H]6OS([O-])(=O)=O)O[C@H]5C([O-])=O)[C@H](CS([O-])(=O)=O)[C@H]4CS([O-])(=O)=O)O[C@]3(CC)C([O-])=O)O[C@@H](COS([O-])(=O)=O)[C@H](OC)[C@H]2OC)C1=O)NC(C)=O)[C@@H](C)O)[C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(C)C)C(=O)N[C@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](C)C(=O)N[C@H](CO)C(=O)N[C@H](CC(C)C)C(=O)N[C@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](CC(N)=O)C(=O)N[C@H](Cc1c[nH]c2ccccc12)C(=O)N[C@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C266H401N53O118S8/c1-32-130(20)204(316-247(377)167(94-128(16)17)300-252(382)180(113-322)313-242(372)173(101-140-110-275-121-280-140)310-256(386)205(131(21)33-2)317-248(378)168(95-129(18)19)301-253(383)181(114-323)314-257(387)206(133(23)324)318-249(379)169(97-136-52-54-141(326)55-53-136)302-254(384)182(282-134(24)325)117-438-186-106-196(336)319(258(186)388)79-76-194(334)276-78-81-422-83-85-424-87-86-423-84-82-421-80-77-195(335)315-207-211(415-26)209(414-25)183(115-425-442(402,403)404)427-261(207)434-220-215(417-28)218(419-30)264(435-266(220,34-3)265(391)392)430-208-145(118-439(393,394)395)146(119-440(396,397)398)260(429-185(208)120-441(399,400)401)432-213-212(416-27)217(418-29)263(433-216(213)259(389)390)431-210-184(116-426-443(405,406)407)428-262(420-31)219(437-445(411,412)413)214(210)436-444(408,409)410)255(385)292-159(65-75-202(347)348)227(357)287-158(64-74-201(345)346)232(362)312-179(112-321)250(380)291-153(59-69-190(270)330)228(358)306-174(102-191(271)331)243(373)288-152(58-68-189(269)329)225(355)283-150(56-66-187(267)327)223(353)285-156(62-72-199(341)342)230(360)295-163(90-124(8)9)238(368)307-175(103-192(272)332)244(374)290-154(60-70-197(337)338)226(356)284-151(57-67-188(268)328)224(354)286-155(61-71-198(339)340)229(359)294-162(89-123(6)7)235(365)297-161(88-122(4)5)234(364)289-157(63-73-200(343)344)231(361)296-164(91-125(10)11)239(369)309-177(105-203(349)350)246(376)298-165(92-126(12)13)236(366)303-170(98-137-107-277-147-49-41-38-46-142(137)147)233(363)281-132(22)222(352)311-178(111-320)251(381)299-166(93-127(14)15)237(367)304-172(100-139-109-279-149-51-43-40-48-144(139)149)241(371)308-176(104-193(273)333)245(375)305-171(99-138-108-278-148-50-42-39-47-143(138)148)240(370)293-160(221(274)351)96-135-44-36-35-37-45-135/h35-55,107-110,121-133,145-146,150-186,204-220,260-264,277-279,320-324,326H,32-34,56-106,111-120H2,1-31H3,(H2,267,327)(H2,268,328)(H2,269,329)(H2,270,330)(H2,271,331)(H2,272,332)(H2,273,333)(H2,274,351)(H,275,280)(H,276,334)(H,281,363)(H,282,325)(H,283,355)(H,284,356)(H,285,353)(H,286,354)(H,287,357)(H,288,373)(H,289,364)(H,290,374)(H,291,380)(H,292,385)(H,293,370)(H,294,359)(H,295,360)(H,296,361)(H,297,365)(H,298,376)(H,299,381)(H,300,382)(H,301,383)(H,302,384)(H,303,366)(H,304,367)(H,305,375)(H,306,358)(H,307,368)(H,308,371)(H,309,369)(H,310,386)(H,311,352)(H,312,362)(H,313,372)(H,314,387)(H,315,335)(H,316,377)(H,317,378)(H,318,379)(H,337,338)(H,339,340)(H,341,342)(H,343,344)(H,345,346)(H,347,348)(H,349,350)(H,389,390)(H,391,392)(H,393,394,395)(H,396,397,398)(H,399,400,401)(H,402,403,404)(H,405,406,407)(H,408,409,410)(H,411,412,413)/p-9/t130-,131-,132+,133+,145+,146+,150-,151-,152-,153-,154-,155-,156-,157-,158-,159-,160+,161-,162-,163-,164+,165+,166+,167-,168-,169-,170+,171+,172+,173-,174-,175-,176+,177+,178+,179-,180-,181-,182?,183-,184-,185+,186?,204-,205-,206-,207-,208-,209-,210-,211-,212-,213-,214-,215+,216+,217+,218+,219-,220-,260+,261+,262+,263+,264+,266-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 93 | n/a | n/a | n/a | n/a | n/a | n/a |
Endotis Pharma
Curated by ChEMBL
| Assay Description
Inhibition of bovine coagulation factor 10a by spectrophotometry |
Antimicrob Agents Chemother 54: 134-42 (2009)
Article DOI: 10.1128/AAC.00827-09
BindingDB Entry DOI: 10.7270/Q23N23KQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50280655
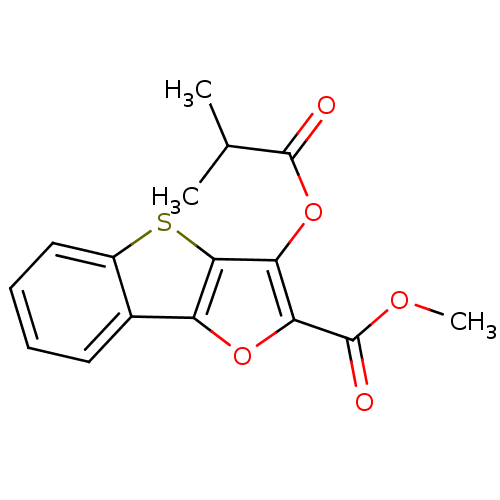
(3-Isobutyryloxy-benzo[4,5]thieno[3,2-b]furan-2-car...)Show InChI InChI=1S/C16H14O5S/c1-8(2)15(17)21-12-13(16(18)19-3)20-11-9-6-4-5-7-10(9)22-14(11)12/h4-8H,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro concentration of compound required to inhibit 50% of Factor Xa |
Bioorg Med Chem Lett 2: 1025-1028 (1992)
Article DOI: 10.1016/S0960-894X(00)80611-3
BindingDB Entry DOI: 10.7270/Q2X066XV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029506
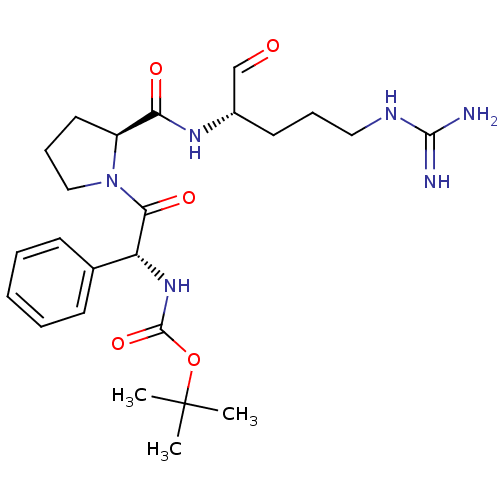
(CHEMBL318998 | tert-butyloxy carbonyl-D-ethylpheny...)Show SMILES CC(C)(C)OC(=O)N[C@@H](C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O)c1ccccc1 Show InChI InChI=1S/C24H36N6O5/c1-24(2,3)35-23(34)29-19(16-9-5-4-6-10-16)21(33)30-14-8-12-18(30)20(32)28-17(15-31)11-7-13-27-22(25)26/h4-6,9-10,15,17-19H,7-8,11-14H2,1-3H3,(H,28,32)(H,29,34)(H4,25,26,27)/t17-,18-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029510
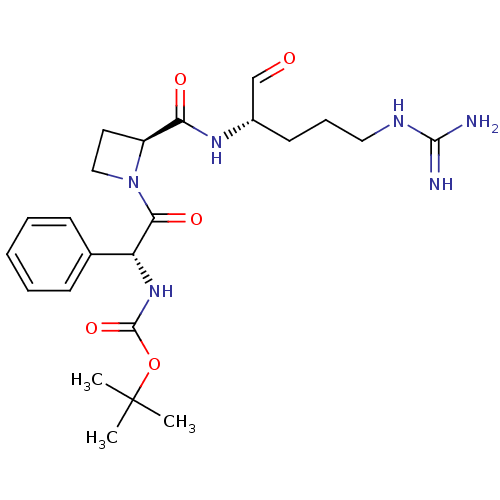
(1N-[2-{2-[4-amino(imino)methylamino-1-formyl-(1S)-...)Show SMILES CC(C)(C)OC(=O)N[C@@H](C(=O)N1CC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O)c1ccccc1 Show InChI InChI=1S/C23H34N6O5/c1-23(2,3)34-22(33)28-18(15-8-5-4-6-9-15)20(32)29-13-11-17(29)19(31)27-16(14-30)10-7-12-26-21(24)25/h4-6,8-9,14,16-18H,7,10-13H2,1-3H3,(H,27,31)(H,28,33)(H4,24,25,26)/t16-,17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM12658
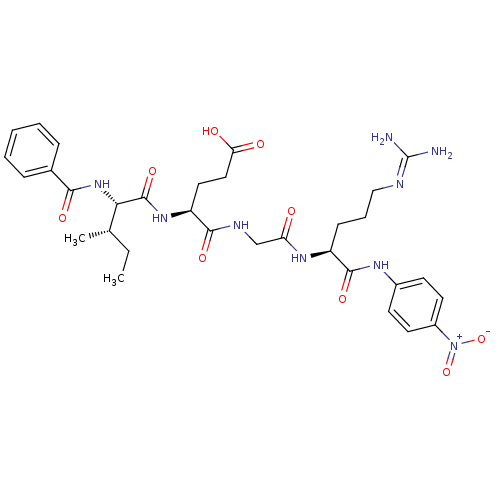
(4-[({[(1S)-4-carbamimidamido-1-[(4-nitrophenyl)car...)Show SMILES [#6]-[#6]-[#6@H](-[#6])-[#6@H](-[#7]-[#6](=O)-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-c1ccc(cc1)-[#7+](-[#8-])=O |r| Show InChI InChI=1S/C32H43N9O9/c1-3-19(2)27(40-28(45)20-8-5-4-6-9-20)31(48)39-24(15-16-26(43)44)29(46)36-18-25(42)38-23(10-7-17-35-32(33)34)30(47)37-21-11-13-22(14-12-21)41(49)50/h4-6,8-9,11-14,19,23-24,27H,3,7,10,15-18H2,1-2H3,(H,36,46)(H,37,47)(H,38,42)(H,39,48)(H,40,45)(H,43,44)(H4,33,34,35)/t19-,23-,24-,27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
COR Therapeutics, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine coagulation factor X |
Bioorg Med Chem Lett 10: 13-6 (2000)
BindingDB Entry DOI: 10.7270/Q2CR5SM3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029501
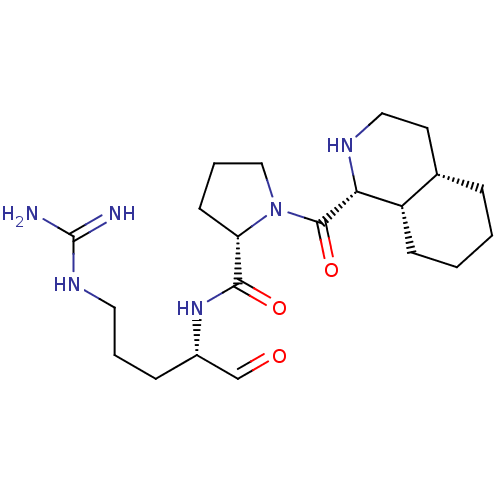
(5N-[4-amino(imino)methylamino-1-formyl-(1S)-butyl]...)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1NCC[C@@H]2CCCC[C@H]12)C=O Show InChI InChI=1S/C21H36N6O3/c22-21(23)25-10-3-6-15(13-28)26-19(29)17-8-4-12-27(17)20(30)18-16-7-2-1-5-14(16)9-11-24-18/h13-18,24H,1-12H2,(H,26,29)(H4,22,23,25)/t14-,15-,16-,17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 550 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029500
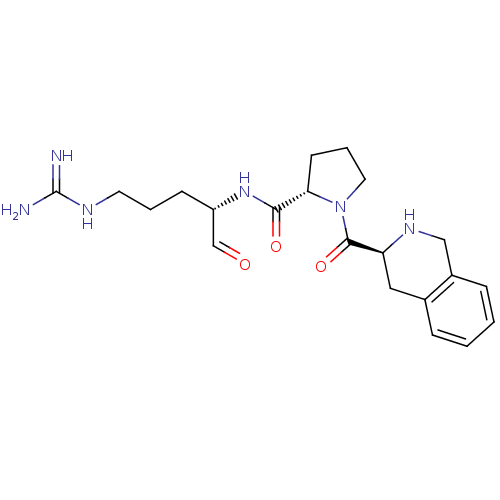
((S)-1-((S)-1,2,3,4-Tetrahydro-isoquinoline-3-carbo...)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1Cc2ccccc2CN1)C=O Show InChI InChI=1S/C21H30N6O3/c22-21(23)24-9-3-7-16(13-28)26-19(29)18-8-4-10-27(18)20(30)17-11-14-5-1-2-6-15(14)12-25-17/h1-2,5-6,13,16-18,25H,3-4,7-12H2,(H,26,29)(H4,22,23,24)/t16-,17-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029499
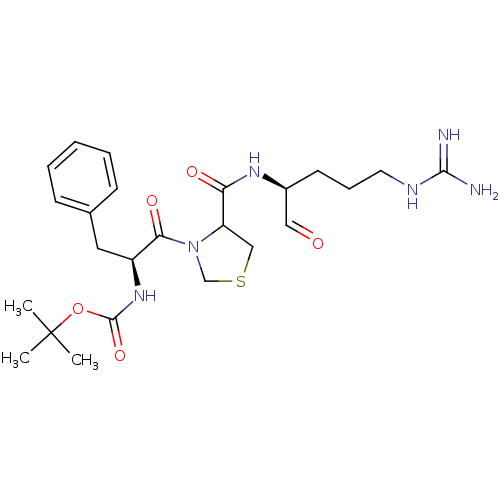
(CHEMBL141834 | tert-butyloxy carbonyl-D-Phe-thiazo...)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)C(=O)N1CSCC1C(=O)N[C@@H](CCCNC(N)=N)C=O Show InChI InChI=1S/C24H36N6O5S/c1-24(2,3)35-23(34)29-18(12-16-8-5-4-6-9-16)21(33)30-15-36-14-19(30)20(32)28-17(13-31)10-7-11-27-22(25)26/h4-6,8-9,13,17-19H,7,10-12,14-15H2,1-3H3,(H,28,32)(H,29,34)(H4,25,26,27)/t17-,18-,19?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 860 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029515
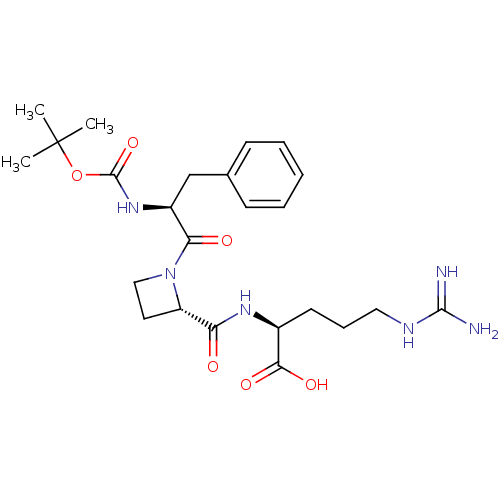
(CHEMBL141584 | tert-butyloxy carbonyl-D-Phe-azetid...)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)C(=O)N1CC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(O)=O Show InChI InChI=1S/C24H36N6O6/c1-24(2,3)36-23(35)29-17(14-15-8-5-4-6-9-15)20(32)30-13-11-18(30)19(31)28-16(21(33)34)10-7-12-27-22(25)26/h4-6,8-9,16-18H,7,10-14H2,1-3H3,(H,28,31)(H,29,35)(H,33,34)(H4,25,26,27)/t16-,17-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029511
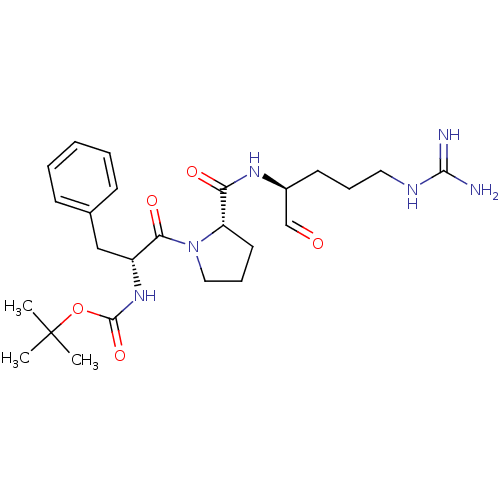
(CHEMBL104472 | tert-butyloxy carbonyl-D-Phe-pro-Ar...)Show SMILES CC(C)(C)OC(=O)N[C@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O Show InChI InChI=1S/C25H38N6O5/c1-25(2,3)36-24(35)30-19(15-17-9-5-4-6-10-17)22(34)31-14-8-12-20(31)21(33)29-18(16-32)11-7-13-28-23(26)27/h4-6,9-10,16,18-20H,7-8,11-15H2,1-3H3,(H,29,33)(H,30,35)(H4,26,27,28)/t18-,19+,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50280654
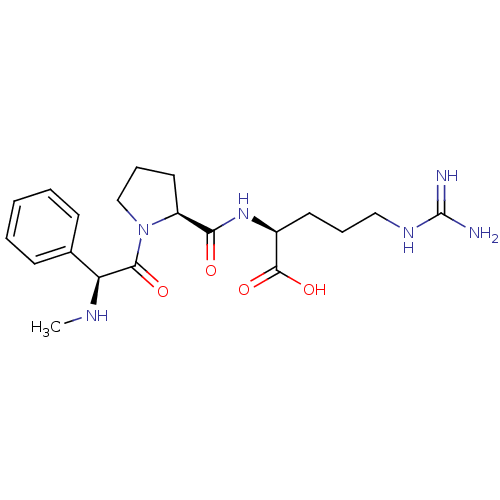
((S)-5-Guanidino-2-{[(S)-1-((S)-2-methylamino-2-phe...)Show SMILES CN[C@H](C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)c1ccccc1 Show InChI InChI=1S/C20H30N6O4/c1-23-16(13-7-3-2-4-8-13)18(28)26-12-6-10-15(26)17(27)25-14(19(29)30)9-5-11-24-20(21)22/h2-4,7-8,14-16,23H,5-6,9-12H2,1H3,(H,25,27)(H,29,30)(H4,21,22,24)/t14-,15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro concentration of compound required to inhibit 50% of factor Xa |
Bioorg Med Chem Lett 2: 1025-1028 (1992)
Article DOI: 10.1016/S0960-894X(00)80611-3
BindingDB Entry DOI: 10.7270/Q2X066XV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50083998

(CHEMBL32044 | [(S)-4-Guanidino-1-({[4-guanidino-1-...)Show SMILES NC(=N)NCCCC(NC(=O)CNC(=O)[C@H](CCC[N-]C(N)=[NH2+])NC(=O)OCc1ccccc1)C(=O)Nc1ccc(cc1)[N+]([O-])=O Show InChI InChI=1S/C28H39N11O7/c29-26(30)33-14-4-8-21(38-28(43)46-17-18-6-2-1-3-7-18)24(41)35-16-23(40)37-22(9-5-15-34-27(31)32)25(42)36-19-10-12-20(13-11-19)39(44)45/h1-3,6-7,10-13,21-22H,4-5,8-9,14-17H2,(H12,29,30,31,32,33,34,35,36,37,38,40,41,42,43)/t21-,22?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
COR Therapeutics, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against bovine coagulation factor X |
Bioorg Med Chem Lett 10: 13-6 (2000)
BindingDB Entry DOI: 10.7270/Q2CR5SM3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029513

((S)-1-((4aS,8aS)-Decahydro-isoquinoline-3-carbonyl...)Show SMILES [H][C@]12CCCC[C@@]1([H])C[C@H](NC2)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O Show InChI InChI=1S/C21H36N6O3/c22-21(23)24-9-3-7-16(13-28)26-19(29)18-8-4-10-27(18)20(30)17-11-14-5-1-2-6-15(14)12-25-17/h13-18,25H,1-12H2,(H,26,29)(H4,22,23,24)/t14-,15+,16-,17-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50228863

((S)-1-((R)-2-Methylamino-3-phenyl-propionyl)-pyrro...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]=O Show InChI InChI=1S/C21H32N6O3/c1-24-17(13-15-7-3-2-4-8-15)20(30)27-12-6-10-18(27)19(29)26-16(14-28)9-5-11-25-21(22)23/h2-4,7-8,14,16-18,24H,5-6,9-13H2,1H3,(H,26,29)(H4,22,23,25)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 7.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029512

(CHEMBL139910 | tert-butyloxy carbonyl-D-Phe-DL-Pro...)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)C(=O)N1C(CCC1(C)C)C(=O)N[C@@H](CCCNC(N)=N)C=O Show InChI InChI=1S/C27H42N6O5/c1-26(2,3)38-25(37)32-20(16-18-10-7-6-8-11-18)23(36)33-21(13-14-27(33,4)5)22(35)31-19(17-34)12-9-15-30-24(28)29/h6-8,10-11,17,19-21H,9,12-16H2,1-5H3,(H,31,35)(H,32,37)(H4,28,29,30)/t19-,20-,21?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029504
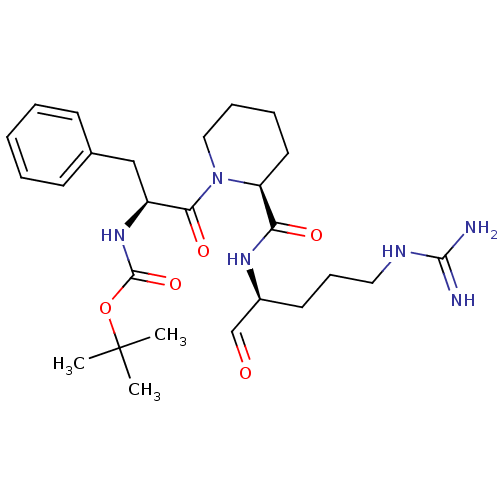
(CHEMBL139490 | {(S)-1-Benzyl-2-[(S)-2-((S)-1-formy...)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)C(=O)N1CCCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O Show InChI InChI=1S/C26H40N6O5/c1-26(2,3)37-25(36)31-20(16-18-10-5-4-6-11-18)23(35)32-15-8-7-13-21(32)22(34)30-19(17-33)12-9-14-29-24(27)28/h4-6,10-11,17,19-21H,7-9,12-16H2,1-3H3,(H,30,34)(H,31,36)(H4,27,28,29)/t19-,20-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50194743
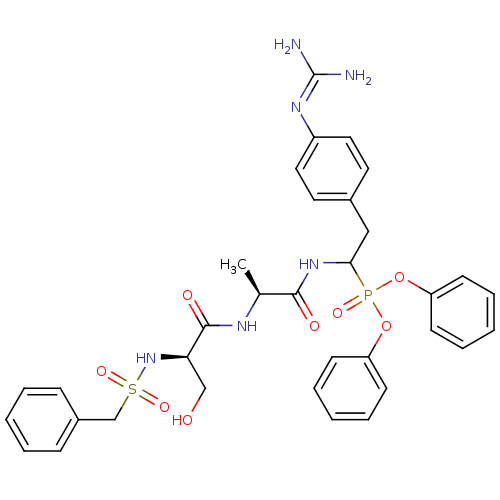
(CHEMBL214814 | diphenyl 1-[(N-alpha-toluenesulfony...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H39N6O8PS/c1-24(37-33(43)30(22-41)40-50(45,46)23-26-11-5-2-6-12-26)32(42)39-31(21-25-17-19-27(20-18-25)38-34(35)36)49(44,47-28-13-7-3-8-14-28)48-29-15-9-4-10-16-29/h2-20,24,30-31,40-41H,21-23H2,1H3,(H,37,43)(H,39,42)(H4,35,36,38)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of bovine F10a |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50194738
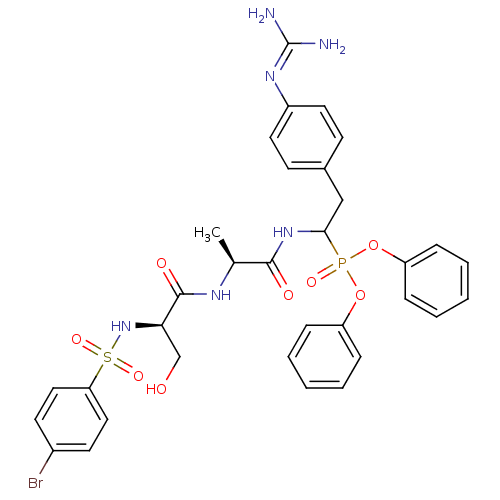
(CHEMBL386249 | diphenyl 1-[(N-p-bromobenzenesulfon...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1ccc(Br)cc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C33H36BrN6O8PS/c1-22(37-32(43)29(21-41)40-50(45,46)28-18-14-24(34)15-19-28)31(42)39-30(20-23-12-16-25(17-13-23)38-33(35)36)49(44,47-26-8-4-2-5-9-26)48-27-10-6-3-7-11-27/h2-19,22,29-30,40-41H,20-21H2,1H3,(H,37,43)(H,39,42)(H4,35,36,38)/t22-,29+,30?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of bovine F10a |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029508
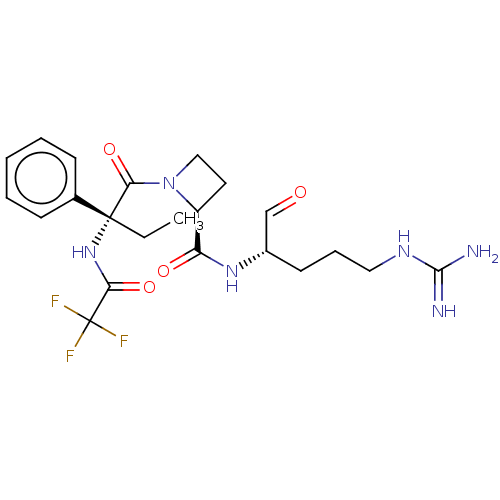
(1N-[1-{2-[4-amino(imino)methylamino-1-formyl-(1S)-...)Show SMILES [#6]-[#6][C@]([#7]-[#6](=O)C(F)(F)F)([#6](=O)-[#7]-1-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6]=O)c1ccccc1 Show InChI InChI=1S/C22H29F3N6O4/c1-2-21(14-7-4-3-5-8-14,30-18(34)22(23,24)25)19(35)31-12-10-16(31)17(33)29-15(13-32)9-6-11-28-20(26)27/h3-5,7-8,13,15-16H,2,6,9-12H2,1H3,(H,29,33)(H,30,34)(H4,26,27,28)/t15-,16-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50145688

(CHEMBL311067 | benzyl (R)-1-((S)-1-(1-(diphenoxyph...)Show SMILES C[C@H](NC(=O)[C@@H](CO)NC(=O)OCc1ccccc1)C(=O)NC(CCCNC(N)=N)P(=O)(Oc1ccccc1)Oc1ccccc1 Show InChI InChI=1S/C31H39N6O8P/c1-22(35-29(40)26(20-38)36-31(41)43-21-23-12-5-2-6-13-23)28(39)37-27(18-11-19-34-30(32)33)46(42,44-24-14-7-3-8-15-24)45-25-16-9-4-10-17-25/h2-10,12-17,22,26-27,38H,11,18-21H2,1H3,(H,35,40)(H,36,41)(H,37,39)(H4,32,33,34)/t22-,26+,27?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of bovine F10a |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029514
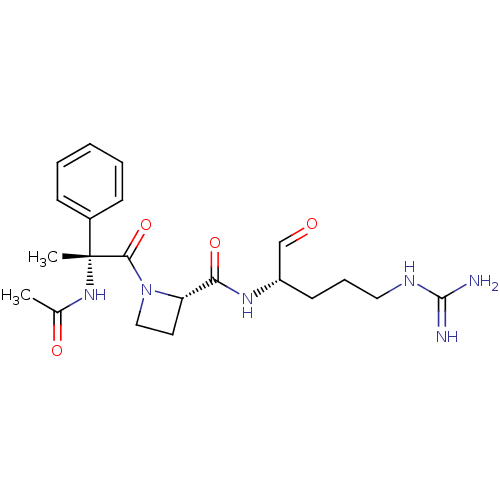
((S)-1-((R)-2-Acetylamino-2-phenyl-propionyl)-azeti...)Show SMILES CC(=O)N[C@@](C)(C(=O)N1CC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O)c1ccccc1 Show InChI InChI=1S/C21H30N6O4/c1-14(29)26-21(2,15-7-4-3-5-8-15)19(31)27-12-10-17(27)18(30)25-16(13-28)9-6-11-24-20(22)23/h3-5,7-8,13,16-17H,6,9-12H2,1-2H3,(H,25,30)(H,26,29)(H4,22,23,24)/t16-,17-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50194744

(CHEMBL385897 | diphenyl 1-[(N-naphthalenesulfonyl-...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1cccc2ccccc12)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C37H39N6O8PS/c1-25(40-36(46)32(24-44)43-53(48,49)33-18-10-12-27-11-8-9-17-31(27)33)35(45)42-34(23-26-19-21-28(22-20-26)41-37(38)39)52(47,50-29-13-4-2-5-14-29)51-30-15-6-3-7-16-30/h2-22,25,32,34,43-44H,23-24H2,1H3,(H,40,46)(H,42,45)(H4,38,39,41)/t25-,32+,34?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of bovine F10a |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50194734

(CHEMBL386275 | diphenyl 1-[(N-p-methoxybenzenesulf...)Show SMILES [#6]-[#8]-c1ccc(cc1)S(=O)(=O)[#7]-[#6@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H39N6O9PS/c1-23(37-33(43)30(22-41)40-51(45,46)29-19-17-26(47-2)18-20-29)32(42)39-31(21-24-13-15-25(16-14-24)38-34(35)36)50(44,48-27-9-5-3-6-10-27)49-28-11-7-4-8-12-28/h3-20,23,30-31,40-41H,21-22H2,1-2H3,(H,37,43)(H,39,42)(H4,35,36,38)/t23-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of bovine F10a |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029502
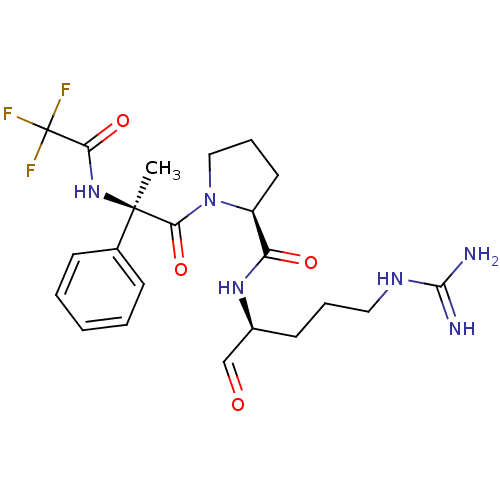
(1N-[2-[5-[4-amino(imino)methylamino-1-formyl-(1S)-...)Show SMILES C[C@](NC(=O)C(F)(F)F)(C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C=O)c1ccccc1 Show InChI InChI=1S/C22H29F3N6O4/c1-21(14-7-3-2-4-8-14,30-18(34)22(23,24)25)19(35)31-12-6-10-16(31)17(33)29-15(13-32)9-5-11-28-20(26)27/h2-4,7-8,13,15-16H,5-6,9-12H2,1H3,(H,29,33)(H,30,34)(H4,26,27,28)/t15-,16-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50194739

(CHEMBL213216 | diphenyl 1-[(N-2-thienyl-D-seryl)-L...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1cccs1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C32H35N6O7PS/c1-21(35-30(41)26(20-39)37-31(42)27-13-8-18-47-27)29(40)38-28(19-22-14-16-23(17-15-22)36-32(33)34)46(43,44-24-9-4-2-5-10-24)45-25-11-6-3-7-12-25/h2-18,21,26,28,39H,19-20H2,1H3,(H,35,41)(H,37,42)(H,38,40)(H4,33,34,36)/t21-,26+,28?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of bovine F10a at 250 uM |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029503

((S)-1-((S)-1,2,3,4-Tetrahydro-isoquinoline-3-carbo...)Show SMILES C[C@@](CCCNC(N)=N)(NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1Cc2ccccc2CN1)C=O Show InChI InChI=1S/C22H32N6O3/c1-22(14-29,9-5-10-25-21(23)24)27-19(30)18-8-4-11-28(18)20(31)17-12-15-6-2-3-7-16(15)13-26-17/h2-3,6-7,14,17-18,26H,4-5,8-13H2,1H3,(H,27,30)(H4,23,24,25)/t17-,18-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Bos taurus) | BDBM50029505
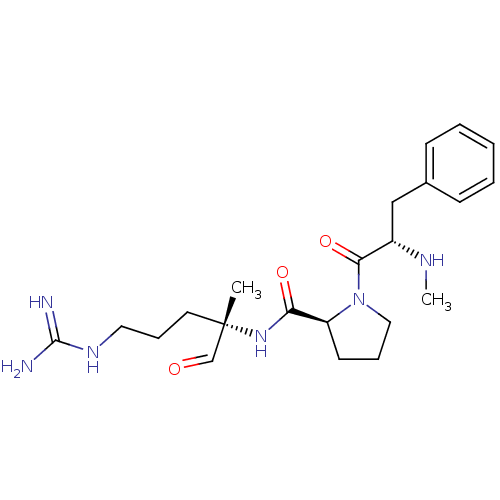
(5N-[4-amino(imino)methylamino-1-formyl-1-methyl-(1...)Show SMILES CN[C@@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@](C)(CCCNC(N)=N)C=O Show InChI InChI=1S/C22H34N6O3/c1-22(15-29,11-7-12-26-21(23)24)27-19(30)18-10-6-13-28(18)20(31)17(25-2)14-16-8-4-3-5-9-16/h3-5,8-9,15,17-18,25H,6-7,10-14H2,1-2H3,(H,27,30)(H4,23,24,26)/t17-,18-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.80E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
In vitro Enzyme Inhibitory activity measured against Coagulation factor X |
J Med Chem 38: 4446-53 (1995)
BindingDB Entry DOI: 10.7270/Q2RB73MV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data